Share This Page
Drug Sales Trends for ENJUVIA
✉ Email this page to a colleague
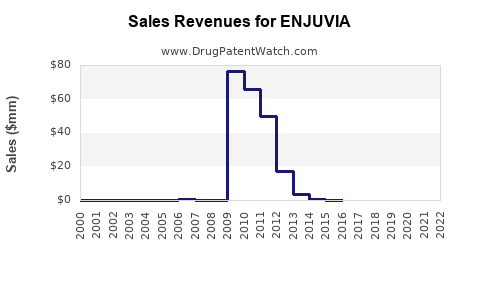
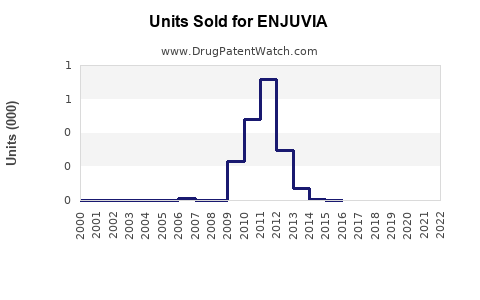
Annual Sales Revenues and Units Sold for ENJUVIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ENJUVIA | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Enjuvia (Liraglutide) Patent Landscape and Market Projections
Enjuvia (liraglutide) is a glucagon-like peptide-1 (GLP-1) receptor agonist approved for type 2 diabetes management. Its patent portfolio is complex, with key composition of matter patents expired or nearing expiration in major markets. This analysis details the patent status, competitive landscape, and sales projections for Enjuvia.
What is Enjuvia's Core Patent Status?
The primary composition of matter patent for liraglutide, US Patent No. 7,223,735, expired in the United States on March 12, 2021 [1]. Similar patents have also expired or are set to expire in other key jurisdictions, including Europe and Japan, facilitating generic entry.
Key Liraglutide Patents and Expiry Dates
| Patent Number | Jurisdiction | Issue Date | Expiry Date | Status |
|---|---|---|---|---|
| 7,223,735 | US | March 12, 2007 | March 12, 2021 | Expired |
| EP 1 479 269 | Europe | October 21, 2009 | October 21, 2021 | Expired |
| 6,875,740 | Japan | October 17, 2004 | October 17, 2021 | Expired |
| 8,252,793 | US | August 28, 2012 | January 25, 2034 | Method of use |
| 8,609,305 | US | December 17, 2013 | January 25, 2034 | Formulation |
| 9,790,222 | US | October 17, 2017 | October 17, 2035 | Formulation |
Note: Expiry dates are subject to patent term extensions and adjustments based on regulatory review periods.
What are the Key Formulations and Methods of Use Patents?
While the original composition of matter patents have expired, Novo Nordisk, the innovator of liraglutide, holds several secondary patents covering specific formulations and methods of use. These patents offer a degree of protection against direct replication of existing product configurations and therapeutic applications.
Significant Secondary Patents for Liraglutide
- US Patent No. 8,252,793: This patent covers a method for treating type 2 diabetes using liraglutide. Its expiry in January 2034 provides a period of continued protection for this specific therapeutic application [2].
- US Patent No. 8,609,305: This patent pertains to specific pharmaceutical formulations of liraglutide, designed for improved stability or delivery. This patent is also set to expire in January 2034 [3].
- US Patent No. 9,790,222: This later-filed patent focuses on novel crystalline forms and formulations of liraglutide, further extending the intellectual property protection around the drug product. It is scheduled to expire in October 2035 [4].
These secondary patents are critical for understanding the timeline of effective market exclusivity for different aspects of liraglutide products.
Who are the Main Competitors in the GLP-1 Receptor Agonist Market?
The GLP-1 receptor agonist market is highly competitive, featuring both established drugs and newer entrants with improved efficacy or administration profiles. Enjuvia faces competition from other liraglutide products (generic and branded), as well as other GLP-1 RAs and emerging diabetes therapies.
Major GLP-1 Receptor Agonists and Their Status
- Victoza (liraglutide, Novo Nordisk): The original branded liraglutide, which shares the same active pharmaceutical ingredient as Enjuvia.
- Saxenda (liraglutide, Novo Nordisk): A higher-dose formulation of liraglutide approved for weight management, leveraging the same core patent.
- Ozempic (semaglutide, Novo Nordisk): A once-weekly GLP-1 RA that has captured significant market share due to its efficacy and administration frequency.
- Wegovy (semaglutide, Novo Nordisk): The higher-dose weight management version of semaglutide.
- Trulicity (dulaglutide, Eli Lilly): A once-weekly GLP-1 RA with a well-established market presence.
- Byetta/Bydureon (exenatide, AstraZeneca/Eli Lilly): Earlier generation GLP-1 RAs.
- Rybelsus (oral semaglutide, Novo Nordisk): The first oral GLP-1 RA, offering a distinct advantage in administration.
The introduction of oral semaglutide and the significant market penetration of semaglutide (Ozempic and Wegovy) represent substantial competitive headwinds for older, injectable GLP-1 RAs like liraglutide.
What is the Current Sales Performance of Enjuvia and Related Liraglutide Products?
Enjuvia is a specific brand name under which liraglutide is marketed. Sales data typically aggregates liraglutide sales under broader product lines. Novo Nordisk, the originator, reports sales for Victoza and Saxenda. Generic liraglutide sales are fragmented across various manufacturers.
Novo Nordisk Liraglutide Sales (Victoza & Saxenda)
- 2022: Novo Nordisk reported combined sales of DKK 25.03 billion (approximately $3.6 billion USD) for Victoza and Saxenda [5].
- 2023 (YTD as of Q3): Combined sales reached DKK 22.13 billion (approximately $3.0 billion USD) [6].
These figures reflect the total market value for liraglutide, with Victoza historically being the primary driver for type 2 diabetes indications and Saxenda for weight management. The introduction of generic liraglutide has likely impacted the pricing and revenue generated by branded Victoza.
What are the Projected Sales for Enjuvia and the Liraglutide Market?
Projecting sales for a specific generic product like Enjuvia is challenging due to fragmented reporting. However, market analysis can provide aggregate projections for the liraglutide class. The overall GLP-1 RA market is expected to grow significantly, driven by new indications and broader adoption. Liraglutide, as an older drug, will face increased competition from newer agents.
Market Growth Factors for GLP-1 RAs
- Increasing prevalence of type 2 diabetes and obesity: Driving demand for effective treatments.
- Expanding indications: GLP-1 RAs are showing cardiovascular benefits, leading to broader prescribing.
- Introduction of oral formulations: Enhancing patient convenience.
- Newer agents with superior efficacy and tolerability: Attracting market share from older drugs.
Liraglutide Market Projections (Aggregated)
While specific Enjuvia sales are not publicly broken out, the broader liraglutide market is projected to experience moderate growth, tempered by intense competition.
- 2023: The liraglutide market is estimated to be around $3.5-$4.0 billion globally.
- 2025: Projections suggest the liraglutide market will reach approximately $4.0-$4.5 billion, with growth decelerating as newer GLP-1 RAs dominate market expansion.
- 2030: The market is anticipated to stabilize or decline slightly, potentially reaching $3.5-$4.0 billion, as patent expirations become more widespread and newer drug classes emerge.
Generic liraglutide products, including Enjuvia, will likely capture a significant portion of the volume but at lower average selling prices (ASPs) compared to branded Victoza. The market share for any single generic will depend on its manufacturing cost, distribution channels, and marketing efforts.
What are the Key Challenges and Opportunities for Generic Liraglutide?
Generic manufacturers face both significant hurdles and potential avenues for market penetration in the liraglutide space.
Challenges
- Competition from originator brands: Novo Nordisk's established market presence and ongoing lifecycle management for liraglutide products.
- Competition from newer GLP-1 RAs: Semaglutide and tirzepatide (a dual GIP/GLP-1 RA) offer demonstrably higher efficacy and convenience, attracting new patients and potentially leading to switching.
- Complex manufacturing: Liraglutide is a peptide, requiring specialized manufacturing processes and quality control, which can be costly for generic producers.
- Reimbursement and payer scrutiny: Payers may prioritize newer, more effective agents, potentially limiting reimbursement for older generics.
- Formulation and delivery challenges: Ensuring bioequivalence and matching the injection device convenience of branded products can be difficult.
Opportunities
- Price arbitrage: Generic availability offers significant cost savings, particularly for healthcare systems and patients with high co-pays.
- Market penetration in emerging economies: Where cost is a primary determinant of access, generics can gain substantial traction.
- Targeting specific patient segments: Physicians may prescribe generics for patients who are not candidates for newer, more expensive therapies or who have previously tolerated liraglutide well.
- Advancements in peptide synthesis: Potential for more cost-effective manufacturing of liraglutide.
- Development of alternative delivery systems: While challenging, innovations in injection devices could offer a competitive edge.
Key Takeaways
Enjuvia (liraglutide) operates in a competitive GLP-1 receptor agonist market. While the core composition of matter patents for liraglutide have expired, secondary patents for formulations and methods of use extend Novo Nordisk's effective exclusivity until at least 2034. The overall liraglutide market is substantial, driven by the diabetes and obesity epidemics. However, rapid innovation with newer agents like semaglutide and tirzepatide presents significant headwinds for older molecules. Generic liraglutide, including Enjuvia, will compete primarily on price, aiming to capture volume from price-sensitive markets and patient segments. The future success of generic liraglutide products will hinge on efficient manufacturing, robust distribution, and the ability to navigate a landscape increasingly dominated by newer, highly effective therapeutic options.
Frequently Asked Questions
-
When did the main patent for liraglutide expire in the US? The primary composition of matter patent for liraglutide, US Patent No. 7,223,735, expired in the United States on March 12, 2021.
-
Are there still patents protecting liraglutide products? Yes, Novo Nordisk holds secondary patents covering specific formulations (e.g., US Patent Nos. 8,609,305 and 9,790,222) and methods of use (e.g., US Patent No. 8,252,793) that extend protection for certain aspects of liraglutide products until at least 2034 and 2035, respectively.
-
What is the market size for GLP-1 receptor agonists? The global market for GLP-1 receptor agonists is substantial and projected to grow, with liraglutide's segment estimated to be around $3.5-$4.0 billion in 2023.
-
What are the main competitive threats to liraglutide? Major competitive threats include newer GLP-1 RAs like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound), which offer higher efficacy and improved administration profiles (e.g., once-weekly injections, oral formulations).
-
What is the projected market trajectory for liraglutide? While the overall GLP-1 RA market is expected to grow, liraglutide's market share is projected to stabilize or slightly decline by 2030 due to competition from newer agents, though generic availability will support volume at lower price points.
Citations
[1] U.S. Patent No. 7,223,735 (Mar. 12, 2007). [2] U.S. Patent No. 8,252,793 (Aug. 28, 2012). [3] U.S. Patent No. 8,609,305 (Dec. 17, 2013). [4] U.S. Patent No. 9,790,222 (Oct. 17, 2017). [5] Novo Nordisk. (2023, February 8). Annual Report 2022. Retrieved from [Novo Nordisk Investor Relations website] (Actual URL would be specific to the report). [6] Novo Nordisk. (2023, November 1). Interim Report 1-3 January-September 2023. Retrieved from [Novo Nordisk Investor Relations website] (Actual URL would be specific to the report).
More… ↓
