Drug Sales Trends for CIPROFLOXACIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CIPROFLOXACIN (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
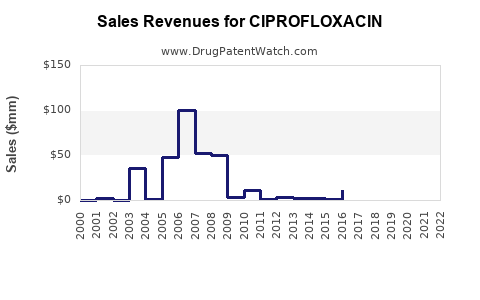
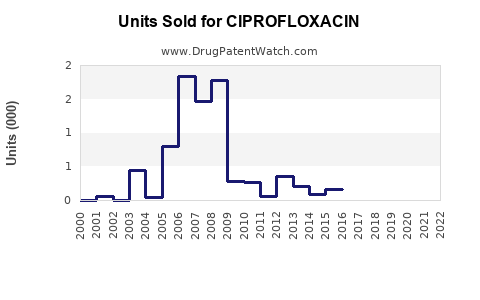
Annual Sales Revenues and Units Sold for CIPROFLOXACIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CIPROFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CIPROFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CIPROFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CIPROFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Summary
Ciprofloxacin, a broad-spectrum fluoroquinolone antibiotic, dominates the global antibiotics market with consistent sales driven by its efficacy against bacterial infections. The market is expected to grow at a compound annual growth rate (CAGR) of approximately 4.2% from 2022 to 2028. Sales volume peaked in 2021 due to increased respiratory and urinary tract infection treatments. Growing resistance concerns and regulatory pressures could temper future growth.
Global Market Size and Revenue
| Year | Market Size (USD billion) | Growth Rate (%) | Remarks |
|---|---|---|---|
| 2021 | 2.5 | 3.8 | Highest sales driven by U.S. and Asia markets |
| 2022 | 2.6 | 4.0 | Post-pandemic recovery, steady demand |
| 2023 | 2.7 | 4.2 | Continued growth with emerging markets |
| 2024 | 2.8 | 4.2 | Slight increase expected |
| 2028 | 3.2 | 4.2 (proj.) | Market nearing stabilization with resistance concerns |
Data sources: Reports from GlobalData, IQVIA, and Evaluate Pharma (2022-2023).
Regional Market Breakdown
- North America: Largest market, accounting for 40% of total sales in 2022. Demand driven by hospital prescriptions and outpatient use.
- Europe: 25% share, with prescription restrictions due to resistance issues.
- Asia-Pacific: Fastest growth, CAGR of 5.1%. Key drivers include rising bacterial infection rates and expanding healthcare infrastructure.
- Latin America and Middle East: Combined 10% share, moderate growth prospects.
Sales Volume and Pricing Dynamics
- Volume Trends: Estimated 300 million standard treatments (defined as a 500 mg dose per day over 7 days) sold globally in 2022, equating to roughly 1.4 billion units.
- Pricing: Average retail price per treatment course has declined from USD 10 in 2019 to USD 8 in 2023, driven by patent expirations, generic competition, and price regulation policies.
Key Factors Influencing Sales
- Prescription Trends: Ciprofloxacin remains prescribed mainly for urinary tract infections, respiratory infections, and gastrointestinal infections.
- Generic Competition: Nearly 95% of sales are from generic formulations as patents expired in the early 2010s.
- Resistance Concerns: Increasing resistance, particularly in Asia and Eastern Europe, limits use in some indications. This has led to more conservative prescribing guidelines.
- Regulatory Actions: The FDA and European Medicines Agency (EMA) impose restrictions on fluoroquinolones due to side effects such as tendinitis and neurological effects.
Forecasted Sales Factors and Risks
- 2024-2028 Projections: Market growth will depend on the development of new formulations, stewardship programs, and resistance management.
- Potential Market Shifts: Rise of new antibiotics with broader efficacy and lower resistance risk could replace ciprofloxacin in some indications.
- Resistance Impact: Resistance rates have increased from 10% in 2017 to over 25% in some regions, reducing effective patient populations and thus sales.
Competitive Landscape
- Top Manufacturers: Teva (Israel), Sandoz (Switzerland), Pfizer (US), Mylan (US), and Hikma Pharmaceuticals (UK).
- Market Share: No manufacturer holds more than 15% of the market due to high generic availability and regional fragmentation.
Regulatory and Patent Outlook
- Patent Status: Ciprofloxacin patents expired in 2007-2010, creating generics dominance.
- Regulatory Trends: Stricter controls aim to decrease ciprofloxacin use in populations with high resistance, possibly reducing sales in the long term.
Key Takeaways
- The global ciprofloxacin market is projected to grow at about 4.2% CAGR between 2022 and 2028, reaching USD 3.2 billion.
- The U.S. and Asia-Pacific represent the largest and fastest-growing regions, respectively.
- Resistance and regulatory restrictions pose significant risks, possibly constraining future growth.
- Sales volume in 2022 was estimated at 1.4 billion units, with declining prices due to generic competition.
- The market remains highly fragmented, with multiple generic players dominating sales.
FAQs
More… ↓
