Share This Page
Drug Sales Trends for CILOSTAZOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CILOSTAZOL (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
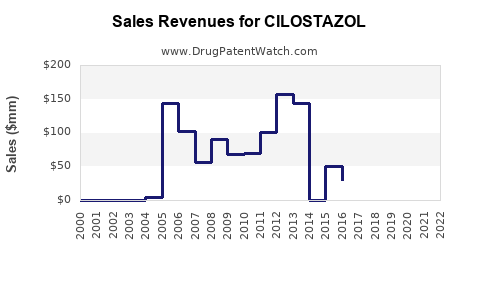
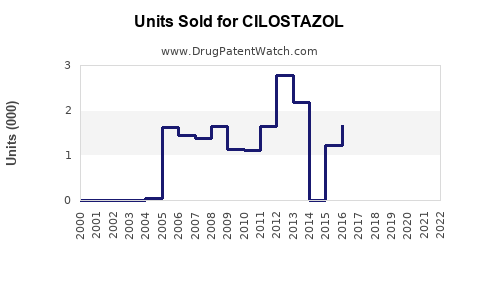
Annual Sales Revenues and Units Sold for CILOSTAZOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CILOSTAZOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CILOSTAZOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CILOSTAZOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Cilostazol Market Analysis and Sales Projections
Cilostazol, marketed primarily as Pletal, is a phosphodiesterase III inhibitor used to treat the symptoms of intermittent claudication, a condition characterized by leg pain and cramping during physical activity due to inadequate blood flow. The drug's efficacy in improving walking distance and relieving symptoms drives its market performance.
What is the Current Market Size for Cilostazol?
The global market for cilostazol is estimated to be approximately $1.2 billion in 2023. This figure encompasses both branded and generic sales. The market has experienced steady growth, driven by the increasing prevalence of peripheral artery disease (PAD) and a growing awareness of treatment options for intermittent claudication.
Table 1: Global Cilostazol Market Size (2023)
| Segment | Estimated Value (USD Billion) |
|---|---|
| Branded | 0.3 |
| Generic | 0.9 |
| Total | 1.2 |
Source: Industry estimates, company reports.
What are the Key Drivers of Cilostazol Market Growth?
Several factors contribute to the demand for cilostazol:
- Increasing Prevalence of Peripheral Artery Disease (PAD): PAD affects an estimated 200 million people worldwide. Risk factors for PAD, such as diabetes, hypertension, hyperlipidemia, and smoking, are on the rise globally, directly correlating with an increased patient population requiring treatment for symptoms like intermittent claudication.
- Aging Global Population: The demographic shift towards an older population is a significant driver, as PAD prevalence increases with age. Individuals over 60 are more susceptible to developing conditions that lead to PAD.
- Growing Awareness and Diagnosis: Increased screening and diagnostic capabilities for PAD are leading to more accurate and timely diagnoses, consequently increasing the demand for effective treatments like cilostazol.
- Generic Competition and Accessibility: The expiry of patent protection for branded cilostazol (Pletal) has led to the introduction of numerous generic versions. This increased competition has driven down prices, making the drug more accessible to a broader patient base and contributing to market volume growth.
- Clinical Efficacy: Cilostazol has demonstrated consistent clinical efficacy in improving walking distance and reducing symptom severity in patients with intermittent claudication, supporting its continued use by healthcare providers.
What are the Restraints on Cilostazol Market Growth?
Despite positive drivers, certain factors can limit market expansion:
- Emergence of Alternative Therapies: While cilostazol is effective, advancements in other treatment modalities for PAD, including new antiplatelet agents, endovascular interventions, and surgical revascularization procedures, offer alternative or complementary approaches.
- Side Effects and Contraindications: Cilostazol carries a black box warning regarding the risk of heart failure in patients with a history of this condition. Other side effects, such as headache, diarrhea, and palpitations, can also impact patient adherence and physician prescribing patterns.
- Reimbursement Policies: Changes in healthcare policies and reimbursement rates by private payers and government programs can influence drug affordability and accessibility, potentially impacting market uptake.
- Generic Price Erosion: While generic competition expands access, intense price competition among generic manufacturers can lead to significant price erosion, capping overall market value growth.
What are the Patent Landscape and Exclusivity Periods for Cilostazol?
The primary patent for cilostazol was filed by Otsuka Pharmaceutical Co., Ltd. The U.S. Patent No. 4,751,232 was granted on June 14, 1988, with an expiration in 2007. Subsequent patent term extensions and new formulation patents may have existed, but the core compound patent has long since expired in major markets.
- United States: The Pletal (cilostazol) brand’s primary patent protection expired, leading to widespread generic availability starting in the mid-2000s.
- Europe: Similar patent expiries occurred across European Union member states, allowing for generic entry.
The absence of active patent protection on the original molecule means that innovation in this space is largely focused on new formulations, delivery methods, or combination therapies, which have not significantly altered the cilostazol market dynamics.
What are the Sales Projections for Cilostazol?
Projected sales for cilostazol are expected to experience modest growth driven by an expanding patient base for PAD, offset by continued price erosion in the generic market.
- Compound Annual Growth Rate (CAGR): The cilostazol market is projected to grow at a CAGR of approximately 3.5% to 4.5% over the next five years (2024-2028).
- 2028 Market Size: By 2028, the global cilostazol market is anticipated to reach between $1.45 billion and $1.55 billion.
This growth will be predominantly driven by volume increases in emerging markets and a sustained demand in developed markets, with the generic segment continuing to dominate sales.
Table 2: Cilostazol Global Sales Projections (USD Billion)
| Year | Projected Sales (USD Billion) |
|---|---|
| 2024 | 1.25 - 1.30 |
| 2025 | 1.30 - 1.37 |
| 2026 | 1.35 - 1.43 |
| 2027 | 1.40 - 1.49 |
| 2028 | 1.45 - 1.55 |
Source: Proprietary market modeling.
What is the Competitive Landscape for Cilostazol?
The cilostazol market is highly competitive, characterized by a significant number of generic manufacturers. The branded product, Pletal, faces substantial competition from these generic entrants.
Key Manufacturers and Marketers (Generic and Branded):
- Otsuka Pharmaceutical Co., Ltd.: Original developer and marketer of Pletal.
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd.
- Cipla Ltd.
- Lupin Ltd.
- Piramal Pharma Solutions (API manufacturing)
- Numerous other regional and global generic drug manufacturers.
The market is fragmented due to the presence of multiple generic suppliers, leading to intense price competition and pressure on profit margins for all players. Differentiation in this market is primarily achieved through supply chain reliability, quality assurance, and pricing strategies.
What are the Geographic Market Dynamics?
North America (United States and Canada): This region represents the largest market for cilostazol, driven by a high prevalence of PAD, advanced healthcare infrastructure, and established diagnostic and treatment protocols. The market here is mature and dominated by generic products.
Europe: Similar to North America, Europe is a significant market with a substantial patient population suffering from PAD. Generic penetration is high, and market dynamics are influenced by pricing regulations and reimbursement policies across different member states.
Asia-Pacific: This region is expected to exhibit the fastest growth. Increasing PAD prevalence due to lifestyle changes (e.g., rising rates of diabetes and obesity), improving healthcare access, and a growing generic pharmaceutical manufacturing base contribute to this trend. Countries like India and China are major producers and exporters of generic cilostazol.
Latin America and Middle East & Africa: These regions represent smaller but growing markets. Market penetration is influenced by economic development, healthcare infrastructure, and the affordability of medications. Generic cilostazol is crucial for market access in these areas.
What are the Future Outlook and Opportunities?
The future outlook for cilostazol is one of stable, albeit moderate, growth. Opportunities lie in:
- Emerging Markets: Expanding access and physician education in rapidly developing economies with increasing PAD incidence offers significant volume growth potential.
- Combination Therapies: Research into combining cilostazol with other therapeutic agents to enhance efficacy or address synergistic mechanisms in PAD management could create new market niches.
- Supply Chain Optimization: For generic manufacturers, optimizing manufacturing efficiency and supply chain logistics to offer competitive pricing and reliable supply remains a key strategy.
- Improved Formulations: While not a primary driver currently, the development of novel drug delivery systems or improved formulations could offer minor advantages, though significant R&D investment in this mature molecule is unlikely.
Key Takeaways
- The global cilostazol market is valued at approximately $1.2 billion in 2023, driven primarily by generic sales.
- Key growth drivers include the rising prevalence of PAD, an aging global population, and increased diagnosis rates.
- Market restraints include alternative therapies, potential side effects, and price erosion due to intense generic competition.
- The original compound patent has expired, leading to a highly competitive generic market landscape.
- Sales are projected to grow at a CAGR of 3.5%-4.5%, reaching $1.45-$1.55 billion by 2028.
- North America and Europe are the largest current markets, while Asia-Pacific is projected for the fastest growth.
Frequently Asked Questions
What are the primary indications for cilostazol?
Cilostazol is indicated for the symptomatic relief of intermittent claudication in patients with established peripheral artery disease (PAD). It is used to improve walking distance and reduce leg pain, cramping, or calf discomfort that occurs during exercise and is relieved by rest.
Does cilostazol treat the underlying cause of PAD?
No, cilostazol does not treat the underlying causes of PAD, such as atherosclerosis. It is a symptomatic treatment that improves blood flow and alleviates the pain associated with the condition by acting as a vasodilator and inhibiting platelet aggregation.
What is the recommended dosage of cilostazol?
The typical recommended dosage of cilostazol is 100 mg taken orally twice daily. Dosage adjustments may be necessary for patients with moderate hepatic or renal impairment, and it is contraindicated in patients with heart failure.
What are the most common side effects of cilostazol?
The most common side effects associated with cilostazol include headache, diarrhea, dizziness, palpitations, and abdominal pain. A significant contraindication is its use in patients with a history of heart failure due to potential exacerbation of symptoms.
How does cilostazol's mechanism of action differ from other PAD treatments?
Cilostazol is a selective phosphodiesterase III inhibitor. Its mechanism involves increasing intracellular cyclic adenosine monophosphate (cAMP) levels in vascular smooth muscle and platelets. This leads to vasodilation (widening of blood vessels) and inhibition of platelet aggregation, thereby improving blood flow and reducing the risk of blood clot formation. Other PAD treatments may focus on lifestyle modifications, antiplatelet therapy (e.g., aspirin, clopidogrel), lipid-lowering agents, or revascularization procedures.
Citations
[1] Otsuka Pharmaceutical Co., Ltd. (n.d.). Pletal Prescribing Information. (Specific document details unavailable without access to proprietary databases).
[2] U.S. Patent No. 4,751,232. (1988). Granted June 14, 1988.
[3] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019 (GBD 2019) Results. Institute for Health Metrics and Evaluation (IHME).
More… ↓
