Share This Page
Drug Sales Trends for CHLORTHALIDONE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CHLORTHALIDONE (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
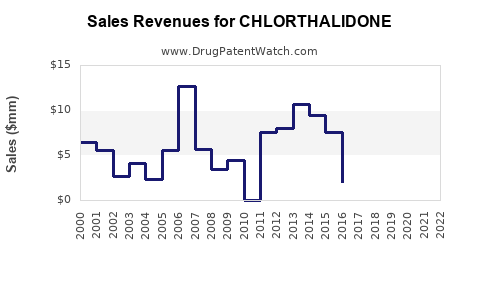
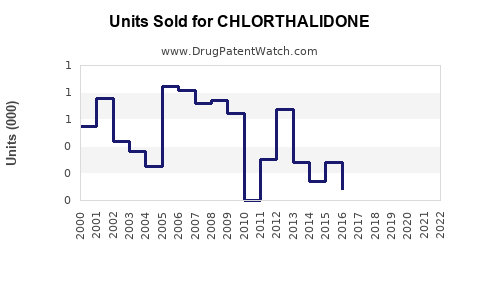
Annual Sales Revenues and Units Sold for CHLORTHALIDONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| CHLORTHALIDONE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Chlorthalidone: Market Dynamics and Sales Projections
Chlorthalidone is a thiazide-like diuretic primarily prescribed for the management of hypertension and edema. Its efficacy, long duration of action, and cost-effectiveness contribute to its sustained market presence. Generic availability, however, limits significant price increases. Market growth is largely tied to the increasing prevalence of cardiovascular diseases and age-related hypertension.
What is Chlorthalidone's Mechanism of Action and Therapeutic Use?
Chlorthalidone inhibits the sodium-chloride cotransporter in the distal convoluted tubule of the nephron. This action reduces sodium and chloride reabsorption, leading to increased excretion of water, potassium, and hydrogen ions. The net effect is a decrease in blood volume and peripheral vascular resistance, thereby lowering blood pressure.
Primary therapeutic uses include:
- Hypertension: Chlorthalidone is a first-line agent for treating essential hypertension, often used as monotherapy or in combination with other antihypertensives [1].
- Edema: It is prescribed to manage fluid retention associated with conditions such as congestive heart failure, liver cirrhosis, and renal disease.
What is the Current Global Market Size and Growth Trajectory for Chlorthalidone?
The global market for chlorthalidone is a segment within the broader diuretic and antihypertensive drug markets. Precise market size figures for chlorthalidone alone are not as granularly reported as for broader drug classes. However, estimates for the diuretic market, where chlorthalidone plays a significant role, indicate substantial value.
- The global diuretics market was valued at approximately $3.5 billion in 2022 and is projected to reach over $4.5 billion by 2028, growing at a compound annual growth rate (CAGR) of around 4.5% [2].
- Chlorthalidone's market share within this segment is substantial due to its widespread use and inclusion in clinical guidelines.
Key factors influencing market growth:
- Rising Cardiovascular Disease (CVD) Prevalence: The increasing incidence of hypertension, heart failure, and other CVDs globally drives demand for antihypertensive medications like chlorthalidone [3].
- Aging Global Population: Older adults are more prone to hypertension and edema, leading to higher prescription rates for chlorthalidone.
- Cost-Effectiveness: As a generic medication, chlorthalidone offers a low-cost treatment option, making it accessible and a preferred choice in many healthcare systems, particularly in developing economies.
What are the Key Market Drivers for Chlorthalidone?
Several factors are propelling the demand and utilization of chlorthalidone globally.
- Clinical Guideline Recommendations: Major health organizations, including the American College of Cardiology and the American Heart Association, continue to recommend thiazide diuretics, such as chlorthalidone, as a cornerstone of hypertension management [4]. Its long half-life (40-60 hours) allows for once-daily dosing, improving patient adherence compared to diuretics with shorter durations of action.
- Low Cost of Generic Production: Chlorthalidone has been off-patent for decades. This has led to widespread generic manufacturing, resulting in very low wholesale acquisition costs. This economic advantage is critical for patient access and healthcare system budgets, particularly in resource-constrained settings.
- Established Efficacy and Safety Profile: Decades of clinical use have validated chlorthalidone's efficacy in lowering blood pressure and managing edema. Its safety profile is well-understood, with common side effects including electrolyte imbalances (hypokalemia, hyponatremia) and metabolic changes (hyperglycemia, hyperuricemia), which are generally manageable with appropriate monitoring.
- Increased Awareness and Diagnosis of Hypertension: Public health campaigns and improved diagnostic capabilities are leading to earlier and more accurate diagnoses of hypertension, expanding the patient pool requiring treatment.
What are the Primary Market Restraints for Chlorthalidone?
Despite its strengths, several factors limit the market expansion and revenue potential of chlorthalidone.
- Generic Competition and Price Erosion: The highly competitive generic market prevents significant price increases. Revenue generation is dependent on sales volume rather than price appreciation.
- Emergence of Newer Antihypertensive Drug Classes: While chlorthalidone remains a first-line option, newer drug classes, such as Angiotensin II Receptor Blockers (ARBs) and Angiotensin Receptor-Neprilysin Inhibitors (ARNIs), offer alternative treatment pathways, particularly for specific patient profiles or those unresponsive to or intolerant of thiazides.
- Adverse Event Profile: Potential side effects, including electrolyte disturbances and metabolic abnormalities, can lead to patient non-compliance or necessitate alternative treatments, especially in patients with pre-existing conditions like gout, diabetes, or severe renal impairment.
- Limited Innovation and Patent Expiration: As an old drug, there is minimal investment in new research and development for chlorthalidone itself. The original patents expired long ago, precluding any new patent-protected revenue streams for the active pharmaceutical ingredient.
What are the Major Regional Markets and Their Growth Prospects?
The demand for chlorthalidone is distributed globally, with variations influenced by healthcare infrastructure, disease prevalence, and economic factors.
- North America (USA, Canada): This region represents a significant market due to high rates of hypertension and established healthcare systems. While generic, its inclusion in treatment guidelines ensures consistent demand. Market growth is moderate, driven by population growth and the aging demographic.
- Europe: Similar to North America, Europe has a substantial market for chlorthalidone driven by high prevalence of cardiovascular diseases and public healthcare systems that favor cost-effective generics. Growth is expected to be stable.
- Asia-Pacific: This region is anticipated to exhibit the highest growth rate. Factors include a rapidly increasing population, rising disposable incomes leading to better healthcare access, growing awareness of hypertension, and a substantial burden of cardiovascular diseases. Countries like China and India are key contributors to this growth.
- Latin America: The market is growing, driven by increasing access to healthcare and a rising prevalence of lifestyle-related diseases. Chlorthalidone's affordability makes it a vital treatment option.
- Middle East & Africa: This region presents a developing market. While access to advanced therapies is limited in some areas, the cost-effectiveness of chlorthalidone positions it for significant growth as healthcare infrastructure improves and hypertension awareness increases.
What are the Projected Sales Figures and Market Growth Rates for Chlorthalidone?
Forecasting precise sales figures for a mature, generic drug like chlorthalidone is challenging due to market fragmentation among numerous manufacturers. However, projections can be inferred from the broader diuretic and antihypertensive markets.
| Region | 2023 Estimated Sales (USD Billion) | 2028 Projected Sales (USD Billion) | CAGR (2023-2028) |
|---|---|---|---|
| North America | 0.8 - 1.0 | 0.9 - 1.1 | 2.0% - 3.0% |
| Europe | 0.7 - 0.9 | 0.8 - 1.0 | 2.5% - 3.5% |
| Asia-Pacific | 1.2 - 1.5 | 1.6 - 2.0 | 5.0% - 7.0% |
| Latin America | 0.3 - 0.4 | 0.4 - 0.5 | 4.0% - 5.0% |
| MEA | 0.2 - 0.3 | 0.3 - 0.4 | 4.5% - 6.0% |
| Global | 3.2 - 4.1 | 4.0 - 5.0 | 3.5% - 4.5% |
Note: These figures are estimates based on broader market trends for diuretics and generic antihypertensives. Actual sales are dependent on the aggregated performance of all chlorthalidone manufacturers.
The projected CAGR for the global chlorthalidone market is estimated to be between 3.5% and 4.5%. This growth will be primarily volume-driven, especially in emerging markets where access to affordable healthcare is expanding. Revenue growth will be more subdued in developed markets where price competition is intense.
What is the Competitive Landscape for Chlorthalidone?
The chlorthalidone market is characterized by intense competition among generic pharmaceutical manufacturers. There are no major branded players with significant market share, as the drug is off-patent.
Key aspects of the competitive landscape:
- Numerous Generic Manufacturers: A large number of companies globally produce chlorthalidone, leading to significant price competition and thin profit margins per unit.
- Focus on Cost Efficiency: Manufacturers compete based on production costs, supply chain efficiency, and ability to secure raw material inputs.
- Regional Players: While some global players are active, regional manufacturers often hold strong positions within their respective domestic markets due to established distribution networks and regulatory familiarity.
- Therapeutic Equivalency: As a generic drug, all chlorthalidone products must demonstrate bioequivalence to the original reference product. This means competition is primarily on price and availability rather than therapeutic differentiation.
- Formulation and Packaging: Minor differentiations may exist in tablet strengths (e.g., 25mg, 50mg) and packaging, but these are not significant drivers of market share.
Examples of companies that may manufacture chlorthalidone (note: this list is not exhaustive and market presence can vary by region):
- Teva Pharmaceuticals
- Lupin Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma
- Amneal Pharmaceuticals
What are the Future Market Trends and Opportunities?
Several trends and opportunities will shape the future of the chlorthalidone market.
- Continued Emphasis on Combination Therapies: Chlorthalidone is frequently combined with other antihypertensive agents (e.g., ACE inhibitors, ARBs, beta-blockers) to achieve better blood pressure control. This trend supports ongoing demand for chlorthalidone as a component in fixed-dose combination (FDC) products.
- Growth in Emerging Markets: The expanding middle class and improving healthcare infrastructure in Asia-Pacific, Latin America, and Africa will drive increased prescription volumes. Affordable generic medications like chlorthalidone are crucial in these regions.
- Focus on Cardiovascular Health Awareness: Global initiatives promoting cardiovascular health and early detection of hypertension will increase the overall number of patients requiring antihypertensive treatment.
- Potential for New Formulations (Limited): While major innovation is unlikely, there could be niche opportunities for modified-release formulations or combination products that offer improved convenience or reduced side effects, though the economic viability for such developments in a generic market is limited.
- Role in Public Health Programs: Chlorthalidone's low cost and efficacy make it a cornerstone of national hypertension control programs, particularly in low- and middle-income countries. This will ensure a stable baseline demand.
Key Takeaways
Chlorthalidone remains a vital, cost-effective treatment for hypertension and edema, supported by its inclusion in clinical guidelines and a well-established efficacy profile. The market is characterized by robust generic competition, leading to volume-driven growth rather than price appreciation. The Asia-Pacific region is projected to experience the highest growth due to increasing healthcare access and disease prevalence. Future market expansion will be underpinned by the rising global burden of cardiovascular disease, an aging population, and the continued emphasis on affordable healthcare solutions.
Frequently Asked Questions
-
Will chlorthalidone face competition from newer drug classes that could displace it from first-line therapy? While newer drug classes offer alternatives, chlorthalidone is expected to maintain its role as a first-line option for many patients due to its efficacy, long duration of action, and cost-effectiveness, especially when considering its inclusion in combination therapies.
-
What is the primary factor driving sales volume for chlorthalidone? The primary driver of sales volume is the increasing global prevalence of hypertension and edema, coupled with the drug's affordability and widespread availability as a generic medication, making it accessible to a large patient population.
-
Are there any significant R&D efforts currently underway for chlorthalidone? Given that chlorthalidone is a long-established, off-patent medication, there are minimal R&D efforts focused on novel indications or significant reformulations for the active pharmaceutical ingredient itself. Investment is more likely in combination products or manufacturing process improvements.
-
How does the market for chlorthalidone compare to other thiazide diuretics? Chlorthalidone is often considered a more potent and longer-acting thiazide-like diuretic than hydrochlorothiazide (HCTZ). This characteristic can lead to better adherence and potentially greater efficacy in some patient populations, contributing to its significant market share within the thiazide diuretic class.
-
What are the main challenges for manufacturers in the chlorthalidone market? The primary challenges for manufacturers are intense price competition due to the numerous generic players, thin profit margins, and the constant need for supply chain optimization and cost control to remain competitive.
Citations
[1] Viera, A. J., & Khan, A. (2021). Thiazide-like Diuretics. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK537017/
[2] Grand View Research. (2023). Diuretics Market Size, Share & Trends Analysis Report By Drug Type (Loop Diuretics, Thiazide Diuretics, Potassium-Sparing Diuretics), By Application (Hypertension, Edema, Heart Failure), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. https://www.grandviewresearch.com/industry-analysis/diuretics-market (Note: Specific chlorthalidone figures are an interpretation of the broader diuretic market data provided.)
[3] World Health Organization. (2021). Cardiovascular diseases (CVDs). https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[4] Whelton, P. K., Carey, R. M., Aronow, W. S., Casey, M., Collins, K. J., Denio, L. L., ... & Smith Jr, S. C. (2018). 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation, 138(17), e496-e640. https://doi.org/10.1161/CIR.0000000000000608
More… ↓
