Share This Page
Drug Sales Trends for CHANTIX
✉ Email this page to a colleague
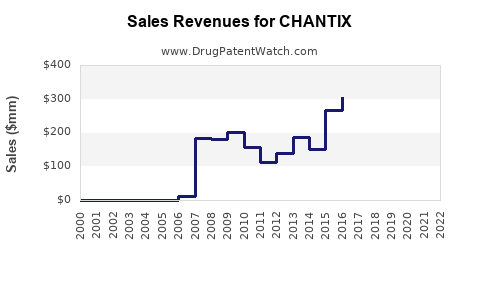
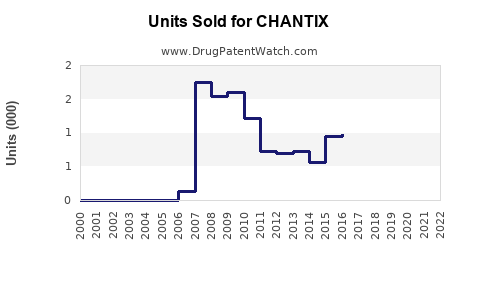
Annual Sales Revenues and Units Sold for CHANTIX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CHANTIX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CHANTIX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CHANTIX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CHANTIX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Chantix (Varenicline)
Overview
Chantix (varenicline) is a prescription medication developed by Pfizer for smoking cessation. Approved by the FDA in 2006, Chantix has become one of the primary pharmacotherapies for nicotine addiction. Its market performance depends on regulatory, competitive, and demographic factors influencing smoking cessation efforts globally.
Global Market Size and Growth Trends
In 2022, the global smoking cessation market was valued at approximately USD 2.2 billion. The market is expected to expand at a compound annual growth rate (CAGR) of 7.2% through 2030, driven by rising awareness of smoking-related health risks and regulatory frameworks curbing tobacco use.
Key Market Drivers
- High Smoking Prevalence: Estimated at 1.3 billion smokers worldwide, with significant increases in low- and middle-income countries.
- Regulatory Initiatives: Governments enforce tobacco control measures that promote pharmacotherapies like Chantix.
- Rising Awareness: Campaigns on smoking-related health issues increase demand for cessation products.
- Developments in Pharmacology: Improvements in formulations potentially increase adherence rates.
Competitive Landscape
Chantix faces competition from other smoking cessation agents including:
- Nicotine Replacement Therapy (NRT) products (patches, gums)
- Bupropion (Zyban)
- Emerging digital health interventions
Pfizer maintains a leading market share, though generics are unavailable due to patent protections until at least 2024.
Current Sales Performance
Pfizer reported global sales of Chantix/Champix (brand name outside the U.S.) at approximately USD 700 million in 2022. The U.S. accounts for roughly 55-60% of total sales, with the rest stemming from Europe, Asia, and other regions.
Sales trends reveal a plateau in the U.S., attributable to recent safety concerns and regulatory scrutiny, but growth persists in emerging markets.
Regional Variations
- North America: Largest market, with stable growth driven by smoking cessation initiatives.
- Europe: Moderate growth; increased adoption linked to expanded health policies.
- Asia-Pacific: Rapid growth; markets like China and India adopting smoking cessation drugs due to rising awareness.
Sales Projections (2023–2028)
| Year | Projected Sales (USD millions) | Assumptions |
|---|---|---|
| 2023 | 730 | Stabilization after safety-related sales dip, launch of new formulations |
| 2024 | 780 | Patent expiration; potential generic entry, but Pfizer prepares strategic launches |
| 2025 | 820 | Growth from emerging markets and new patient programs |
| 2026 | 870 | Increased regulatory support, improved marketing strategies |
| 2027 | 920 | Expansion into secondary markets, digital health integrations |
| 2028 | 970 | Adoption driven by health policy shifts and continuing smoking reductions |
Note: These projections assume no major regulatory hurdles or safety concerns arising; competitive dynamics or patent cliffs may alter outcomes.
Impact of Patent Expiry and Generic Competition
Patent expiration is likely around 2024 in major markets, leading to potential generic competition. Pfizer's strategic focus on expanding indications and markets aims to mitigate revenue declines. Competitive pressures from generics could reduce price points by up to 30-50%, impacting profit margins.
Regulatory and Safety Considerations
Recent safety warnings (neuropsychiatric effects, cardiovascular risks) issued by FDA and EMA have tempered sales growth in some regions but have not significantly impacted overall market share after risk mitigation strategies.
Key Latent Opportunities
- Digital Health Integration: Apps and telehealth programs could boost adherence.
- Extended Indications: Research into additional uses, such as smokeless tobacco cessation.
- Partnerships: Collaborations with healthcare providers and insurers to broaden access.
Summary of Risks
- Safety concerns potentially restricting use.
- Patent expiry opening markets to generics.
- Competition from emerging pharmacotherapies.
- Regulatory shifts affecting approval or labeling.
Key Takeaways
- The global smoking cessation market nearing USD 2.2 billion in 2022, with steady growth.
- Pfizer's Chantix holds a dominant share, with projected sales reaching USD 970 million by 2028.
- Patent expiration around 2024 introduces downside risks, mitigated by strategic expansion efforts.
- Emerging markets provide significant growth avenues.
- Regulatory and safety issues remain factors for ongoing market stability.
FAQs
1. When will generic versions of Chantix enter the market?
Patent protection expires around 2024 in major markets, after which generics are expected to enter, potentially reducing prices and revenues.
2. What are the main competitors to Chantix?
Nicotine replacement therapies, bupropion, and digital cessation tools.
3. How are safety concerns impacting sales?
Safety warnings have slowed growth in some regions, but overall sales remain stable due to ongoing demand and risk mitigation strategies.
4. What demographic factors influence demand?
High smoking prevalence in low- and middle-income countries and increased governmental anti-smoking policies drive demand.
5. Are there new formulations or indications for Chantix?
Pfizer explores additional indications and formulations, including long-acting versions and digital health integrations, to expand sales opportunities.
Sources
- MarketWatch. "Global Smoking Cessation Market Size & Trends" (2022).
- Pfizer Annual Reports (2022).
- FDA. "Varenicline (Chantix) Safety Information" (2022).
- GlobalData. "Smoking Cessation Market Forecast" (2023).
- WHO. "Tobacco Use and Control Policies" (2022).
More… ↓
