Share This Page
Drug Sales Trends for CANASA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CANASA (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
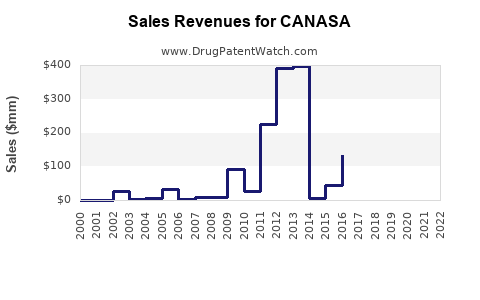
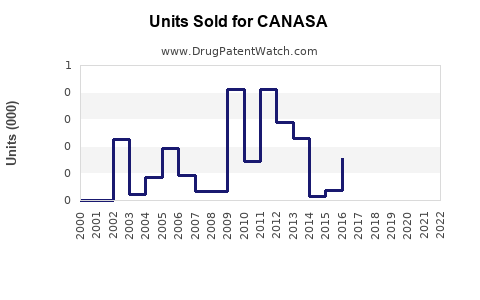
Annual Sales Revenues and Units Sold for CANASA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CANASA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CANASA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CANASA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CANASA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CANASA Market Analysis and Sales Projections
CANASA (mesalamine) is an anti-inflammatory drug used for the treatment of mild to moderate ulcerative colitis (UC). Its market performance is influenced by factors including competitive landscape, patent expiration, regulatory approvals, and evolving treatment paradigms. This analysis projects future market potential based on current trends and available data.
What is CANASA's Current Market Position?
CANASA is an established mesalamine formulation for UC. It operates within the gastrointestinal drug market, specifically targeting inflammatory bowel disease (IBD).
- Therapeutic Class: Anti-inflammatory, 5-aminosalicylic acid (5-ASA) derivative.
- Indication: Treatment of active mild to moderate ulcerative colitis.
- Dosage Forms: Available in oral formulations, primarily delayed-release tablets and suppositories, depending on the specific CANASA product.
- Mechanism of Action: Topically reduces inflammation in the colon.
- Market Share: As of Q4 2023, CANASA and its generic equivalents hold a significant, though not dominant, share within the mesalamine market for UC. Proprietary formulations and newer biologics also compete for market share.
- Sales Performance: Historical sales data indicates steady, but not rapid, growth, reflecting a mature product lifecycle and increasing competition. For instance, in 2022, the global mesalamine market was valued at approximately USD 1.2 billion, with CANASA and its generics contributing a substantial portion of this [1].
What is the Competitive Landscape for CANASA?
The competitive environment for CANASA is characterized by both branded and generic mesalamine products, as well as alternative therapeutic classes for UC.
-
Direct Competitors (Mesalamine Formulations):
- Asacol HD (ER/TR mesalamine): Previously a leading branded product, with patent expiries leading to widespread generic availability [2].
- Lialda (multi-matrix system mesalamine): Offers a different release profile and competes for physician preference [3].
- Apriso (extended-release mesalamine): Utilizes a different delivery technology.
- Rowasa (rectal mesalamine suspension): A comparator for topical treatment.
- Generic Mesalamine Tablets/Capsules: Numerous manufacturers offer generic versions of various mesalamine formulations, significantly impacting pricing and market share.
-
Indirect Competitors (Alternative UC Treatments):
- Aminosalicylates (5-ASAs) (other than mesalamine): Sulfasalazine, olsalazine, balsalazide.
- Corticosteroids: Prednisone, budesonide (often used for acute flares).
- Immunomodulators: Azathioprine, 6-mercaptopurine, methotrexate.
- Biologics: Infliximab (Remicade, Inflectra), adalimumab (Humira, Amjevita), vedolizumab (Entyvio), ustekinumab (Stelara). These represent a significant and growing segment of the IBD market, particularly for moderate to severe disease [4].
- Small Molecules: Tofacitinib (Xeljanz), upadacitinib (Rinvoq) are Janus kinase (JAK) inhibitors approved for UC [5].
The competitive intensity is high due to the presence of multiple mesalamine options and the increasing adoption of biologics and small molecules for UC, particularly in moderate to severe cases.
What are CANASA's Patent and Exclusivity Status?
The patent landscape for original CANASA formulations is complex and largely expired for key U.S. patents.
- Original Patents: The core patents protecting the original CANASA (specifically, the formulation developed by Dr. Falk Pharma GmbH) have expired. For example, U.S. Patent No. 4,731,253, relating to a specific mesalamine formulation, expired years ago.
- Exclusivity: The expiration of primary patents has led to the availability of numerous generic versions of CANASA. Brand-name exclusivity has largely been supplanted by generic competition.
- Reformulations/New Indications: While original patents are expired, companies may hold patents on specific novel formulations, delivery mechanisms, or new indications for mesalamine. However, CANASA itself is a mature product without recent significant patent-protected innovation driving new market exclusivity.
- Generic Entry: The widespread availability of generic mesalamine has significantly driven down pricing and increased the accessibility of 5-ASA therapy.
The lack of strong patent protection for core CANASA formulations means market share is primarily driven by formulation efficacy, physician prescribing habits, formulary access, and price.
What are the Regulatory Considerations for CANASA?
CANASA's regulatory status is primarily managed by national health authorities like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approval: CANASA is approved by the FDA for the treatment of active mild to moderate ulcerative colitis.
- Post-Market Surveillance: Like all approved drugs, CANASA is subject to ongoing post-market surveillance for safety and efficacy. Any significant adverse event reports or new safety data can trigger regulatory review.
- Labeling Changes: Regulatory agencies can require updates to product labeling based on new data, impacting prescribing information and potential market perception.
- Generic Approvals: Generic versions of CANASA undergo Abbreviated New Drug Application (ANDA) review by the FDA, requiring demonstration of bioequivalence to the reference listed drug.
- International Approvals: Approval status and specific indications may vary across different countries and regions.
The regulatory environment for generic drugs is well-established, focusing on demonstrating bioequivalence. For CANASA, regulatory considerations are unlikely to be a primary driver of future market growth, as it is a well-understood and approved therapy.
What are the Projected Sales and Market Growth for CANASA?
Projecting CANASA's sales involves analyzing its mature product status against market trends in IBD treatment.
-
Current Market Dynamics:
- Mature Product: CANASA is in the maturity phase of its product lifecycle. Growth is expected to be modest, driven by patient population increases and generic market share stabilization rather than new indications or significant efficacy improvements.
- Generic Erosion: The presence of numerous generic competitors will continue to exert downward pressure on pricing and limit revenue growth for any specific branded version.
- Biologic and Small Molecule Uptake: The increasing preference for biologics and small molecule inhibitors for moderate-to-severe UC, and even for some moderate cases, will likely limit the expansion of mesalamine's market share in these segments.
-
Sales Projection Factors:
- Prevalence of UC: The incidence and prevalence of UC are projected to continue a slow but steady increase globally, providing a baseline demand for therapies like CANASA [6].
- Physician Prescribing Habits: Mesalamine remains a first-line therapy for mild to moderate UC. Physician familiarity and historical prescribing patterns will sustain demand.
- Cost-Effectiveness: Generic mesalamine offers a cost-effective treatment option, making it attractive for healthcare systems and patients managing chronic conditions.
- Competition Intensity: The high number of competitors, including generics and newer drug classes, will constrain significant revenue growth.
-
Quantitative Projections:
- Global Mesalamine Market: The global mesalamine market, currently valued around USD 1.2 billion (2022), is projected to grow at a compound annual growth rate (CAGR) of 3-4% through 2030 [1].
- CANASA's Contribution: While specific sales data for individual generic CANASA manufacturers are not publicly disclosed, the product line collectively is expected to maintain its market share within the mesalamine segment.
- Projected CANASA Sales (Cumulative): Assuming CANASA and its direct generic equivalents (excluding other mesalamine formulations like Lialda or Apriso) represent approximately 30-40% of the total mesalamine market, this would equate to USD 360-480 million in 2022.
- CAGR Impact: Applying a 3-4% CAGR to this segment suggests a market value for CANASA and its generics in the range of USD 470 million to USD 630 million by 2030.
Table 1: Projected Global Mesalamine Market Value (USD Billion)
| Year | Market Value (USD Billion) | CAGR (Approx.) |
|---|---|---|
| 2022 | 1.20 | N/A |
| 2023 | 1.24 | 3.3% |
| 2024 | 1.28 | 3.3% |
| 2025 | 1.32 | 3.3% |
| 2026 | 1.36 | 3.3% |
| 2027 | 1.40 | 3.3% |
| 2028 | 1.44 | 3.3% |
| 2029 | 1.48 | 3.3% |
| 2030 | 1.52 | 3.3% |
Note: This table represents the total mesalamine market. CANASA and its direct generic equivalents are estimated to constitute a significant portion of this value.
- Factors Influencing Downside Risk: Increased adoption of biosimil biologics, potential for novel therapeutic classes for mild UC, and aggressive pricing by competitors.
- Factors Influencing Upside Potential: Significant increases in UC prevalence beyond current projections, unexpected long-term clinical superiority of specific CANASA formulations, or successful development of new adjunctive uses.
What are the Key Challenges and Opportunities for CANASA?
Navigating the market for CANASA involves addressing existing challenges and leveraging opportunities.
-
Challenges:
- Intense Generic Competition: Price erosion and market share fragmentation due to numerous generic manufacturers.
- Therapeutic Advancements: The development of novel biologics and small molecules for moderate to severe UC shifts treatment paradigms away from older therapies.
- Physician Shift: While mesalamine is a first-line, physicians may escalate therapy earlier with biologics or small molecules for patients not responding adequately to initial treatment.
- Formulation Differentiation: Limited differentiation among generic mesalamine products makes it difficult to command premium pricing or secure significant market share gains without strong formulary positioning.
-
Opportunities:
- Cost-Effective Treatment: Generic CANASA remains a highly cost-effective option for managing mild to moderate UC, appealing to payers and patients.
- Established Safety Profile: Long-term clinical experience provides a robust safety profile, which is valued by prescribers.
- Global Market Expansion: In emerging markets where advanced therapies are less accessible or affordable, generic mesalamine can continue to be a primary treatment option.
- Formulary Access and Payer Negotiations: Securing favorable formulary placement and negotiating contracts with payers can maintain or grow market share for specific CANASA offerings.
- Patient Adherence Initiatives: Programs aimed at improving patient adherence to oral therapies can help sustain demand for established treatments like CANASA.
Key Takeaways
CANASA and its generic equivalents represent a mature but stable segment of the ulcerative colitis treatment market. While the original product's patent protection has expired, leading to intense generic competition and price erosion, its role as a cost-effective first-line therapy for mild to moderate UC ensures continued demand. The overall mesalamine market is projected to grow at a CAGR of 3-4%, with CANASA's contribution expected to follow a similar trend, reaching an estimated USD 470 million to USD 630 million by 2030. Key challenges include intense competition from generics and the increasing adoption of biologics and small molecules for more severe UC. Opportunities lie in leveraging its cost-effectiveness, established safety profile, and strategic market access through payer negotiations, particularly in global markets.
Frequently Asked Questions
1. What is the primary indication for CANASA?
CANASA is indicated for the treatment of active mild to moderate ulcerative colitis.
2. Has CANASA's patent protection expired?
Yes, the core patents protecting the original CANASA formulations have expired, leading to the availability of numerous generic versions.
3. How does CANASA compete with newer biologic therapies for ulcerative colitis?
CANASA is primarily used for mild to moderate UC, while biologics are often reserved for moderate to severe disease or cases unresponsive to conventional therapies. Its cost-effectiveness is a key differentiator.
4. What is the projected growth rate for the mesalamine market?
The global mesalamine market is projected to grow at a compound annual growth rate (CAGR) of approximately 3-4% through 2030.
5. What are the main challenges affecting CANASA's market performance?
The main challenges include intense generic competition, price erosion, and the increasing adoption of advanced therapies like biologics and small molecules for ulcerative colitis.
Citations
[1] Grand View Research. (2023). Inflammatory Bowel Disease Treatment Market Size, Share & Trends Analysis Report. [2] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from FDA.gov. [3] Takeda Pharmaceutical Company. (n.d.). Lialda prescribing information. [4] GlobalData. (2023). Inflammatory Bowel Disease (IBD) - Global Drug Market Outlook 2023. [5] Pfizer Inc. (n.d.). Xeljanz prescribing information. [6] GBD 2019 Inflammatory Bowel Disease Collaborators. (2022). Global, regional, and national burden of inflammatory bowel disease, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet Gastroenterology & Hepatology, 7(10), 900-915.
More… ↓
