Share This Page
Drug Sales Trends for AZATHIOPRINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for AZATHIOPRINE (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
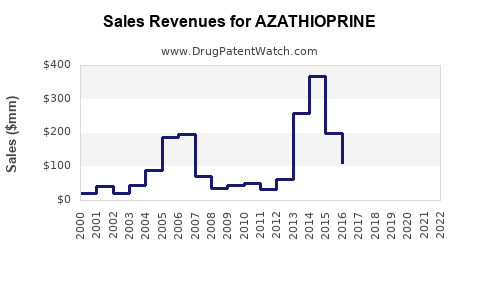
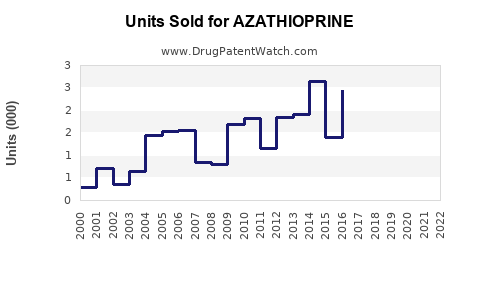
Annual Sales Revenues and Units Sold for AZATHIOPRINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AZATHIOPRINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AZATHIOPRINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AZATHIOPRINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
AZATHIOPRINE MARKET ANALYSIS AND SALES PROJECTIONS
Azathioprine, an immunosuppressive drug, has maintained a steady market presence for decades, primarily treating autoimmune diseases and preventing organ transplant rejection. Its established efficacy, combined with a generic status, leads to predictable sales patterns influenced by disease prevalence, prescribing trends, and healthcare policy. The market is mature, with limited novel patent activity, making demand largely driven by its therapeutic utility and cost-effectiveness.
WHAT IS THE CURRENT MARKET SIZE AND GROWTH TRAJECTORY FOR AZATHIOPRINE?
The global azathioprine market is estimated to be between $150 million and $200 million annually. Growth is projected to be modest, typically in the low single digits (1-3%) per year. This trajectory is primarily influenced by the stable demand for its established indications and the lack of significant new therapeutic applications or competitive patent-protected entrants.
Key market drivers include:
- Prevalence of Autoimmune Diseases: Conditions such as rheumatoid arthritis, Crohn's disease, ulcerative colitis, and multiple sclerosis, for which azathioprine is a treatment option, demonstrate consistent or slightly increasing incidence rates.
- Organ Transplantation Rates: While not the sole immunosuppressant used, azathioprine's role in post-transplant management contributes to demand. Global organ transplant volumes, though subject to donor availability, show a gradual upward trend.
- Generic Availability and Cost-Effectiveness: As a fully genericized drug, azathioprine offers a significant cost advantage compared to newer biologics and targeted therapies. This makes it an attractive option for healthcare systems and patients seeking affordable treatment.
Conversely, market restraints include:
- Emergence of Newer Therapies: The development of more specific and often more effective biologic and small molecule drugs for autoimmune diseases and transplant rejection poses a competitive challenge. These newer agents, while more expensive, may offer improved safety profiles or efficacy in certain patient populations.
- Safety Profile: Azathioprine has known side effects, including bone marrow suppression, gastrointestinal issues, and increased infection risk, which can limit its use in some patients or necessitate close monitoring.
- Biosimilar Competition: While azathioprine itself is a small molecule, the rise of biosimil versions of biologic drugs previously competing with azathioprine can put further price pressure on the entire immunosuppressive market, indirectly affecting azathioprine's market share dynamics.
The market is characterized by a fragmented manufacturing landscape, with multiple generic pharmaceutical companies producing azathioprine. This competition generally keeps prices low and stable.
WHICH THERAPEUTIC AREAS DOMINATE AZATHIOPRINE'S MARKET SHARE?
The primary therapeutic areas driving azathioprine sales are autoimmune diseases and organ transplantation.
-
Autoimmune Diseases: This segment accounts for the largest portion of azathioprine's market share.
- Inflammatory Bowel Disease (IBD): Azathioprine is a cornerstone therapy for maintaining remission in Crohn's disease and ulcerative colitis. Its efficacy in reducing the need for corticosteroids makes it a valuable maintenance drug. Studies indicate that it is used in a significant percentage of IBD patients requiring long-term immunosuppression [1].
- Rheumatoid Arthritis (RA): While newer disease-modifying antirheumatic drugs (DMARDs) and biologics are now preferred for many RA patients, azathioprine remains an option, particularly for those who are treatment-naive or have failed other therapies, or in regions where cost is a major consideration.
- Other Autoimmune Conditions: Azathioprine is also prescribed off-label or for specific indications in conditions such as lupus (systemic lupus erythematosus), autoimmune hepatitis, and certain dermatological conditions like psoriasis and eczema, though these represent smaller market segments.
-
Organ Transplantation: Azathioprine is part of the immunosuppressive regimen for patients receiving solid organ transplants (e.g., kidney, liver, heart). It is often used in combination with other agents like corticosteroids and calcineurin inhibitors. While newer potent immunosuppressants have gained prominence, azathioprine's cost-effectiveness can make it a component of maintenance therapy, especially in resource-limited settings or for long-term management to reduce the burden of more toxic agents [2].
The relative contribution of these segments is subject to evolving treatment guidelines and the introduction of new therapies. However, IBD remains the most substantial driver of azathioprine demand.
WHAT ARE THE KEY GEOGRAPHICAL MARKETS FOR AZATHIOPRINE SALES?
The geographical distribution of azathioprine sales mirrors the prevalence of its target diseases and the healthcare infrastructure of various regions.
- North America (United States & Canada): This region represents a significant market due to the high prevalence of autoimmune diseases and well-established organ transplantation programs. The presence of a mature generic pharmaceutical market and a healthcare system that balances innovation with cost-effectiveness supports steady azathioprine sales.
- Europe: Similar to North America, Europe has a large patient population with autoimmune conditions and active transplant networks. Countries with strong national health services often rely on cost-effective generics like azathioprine for chronic disease management. The United Kingdom, Germany, France, and Italy are notable markets.
- Asia-Pacific: This region is a growing market for azathioprine. Increasing awareness of autoimmune diseases, improving healthcare access, and a large population base contribute to demand. Countries like India, China, Japan, and South Korea are key contributors, with India being a major manufacturing hub for generic drugs.
- Latin America: Brazil, Mexico, and Argentina are the primary markets in this region. The increasing prevalence of chronic diseases and a growing demand for affordable healthcare solutions drive azathioprine sales.
- Middle East & Africa: While currently a smaller market, it is expected to grow as healthcare infrastructure develops and access to essential medicines improves.
The mature nature of the azathioprine market means that sales growth in developed regions is primarily driven by volume increases related to disease incidence rather than price appreciation. Emerging markets offer greater growth potential due to expanding healthcare access and a higher proportion of patients benefiting from cost-effective generic treatments.
WHAT IS THE PATENT AND REGULATORY LANDSCAPE SURROUNDING AZATHIOPRINE?
Azathioprine is a well-established drug, and its original patents expired decades ago. There are no active composition of matter patents that would grant market exclusivity for the molecule itself.
- Patent Expiration: The primary patents protecting azathioprine expired in the late 1980s and early 1990s. This has led to its status as a fully genericized drug.
- New Patent Activity: While new patents for azathioprine are rare, some may exist related to:
- New Formulations: Development of novel delivery systems, extended-release formulations, or combination products incorporating azathioprine.
- Manufacturing Processes: Proprietary methods for synthesizing azathioprine that offer improved efficiency, purity, or cost reduction.
- New Indications: While highly unlikely for a drug with its safety profile and established uses, a patent could theoretically be sought for a novel, specific therapeutic application if groundbreaking evidence emerged. However, such patents are exceptionally rare for old drugs unless a significant repurposing is discovered.
- Regulatory Status: Azathioprine is approved by major regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others. Manufacturers must adhere to stringent Good Manufacturing Practices (GMP) and bioequivalence requirements to gain and maintain market approval for generic versions.
- Orphan Drug Status: Azathioprine is not designated as an orphan drug for any of its primary indications.
The absence of patent protection means that competition in the azathioprine market is driven by manufacturing efficiency, distribution networks, and market access rather than intellectual property rights. Regulatory hurdles focus on ensuring the quality and bioequivalence of generic formulations.
WHAT ARE THE PROJECTED SALES FIGURES AND GROWTH RATES FOR AZATHIOPRINE OVER THE NEXT FIVE YEARS?
Projecting sales for a mature, genericized drug like azathioprine involves considering stable demand patterns, disease incidence, and competitive pressures.
Projected Global Sales (USD Millions):
- 2024: $175
- 2025: $178
- 2026: $181
- 2027: $184
- 2028: $187
Projected Annual Growth Rate: 1.5% - 2.5%
Factors Influencing Projections:
- Stable Demand: The established efficacy and cost-effectiveness of azathioprine for chronic autoimmune conditions and transplant maintenance ensure a baseline demand that is unlikely to fluctuate dramatically.
- Disease Prevalence: Continued or slightly increasing incidence of IBD, RA, and other autoimmune conditions will sustain the need for azathioprine. Organ transplant volumes are also expected to see marginal increases.
- Generic Competition: The market will remain highly competitive among generic manufacturers, preventing significant price increases and capping overall revenue growth.
- Evolving Treatment Paradigms: The gradual adoption of newer, more expensive biologics and targeted therapies for certain autoimmune conditions may lead to a slow erosion of azathioprine's market share in specific patient subgroups, particularly in developed markets where cost is less of a barrier. However, its affordability will maintain its position as a critical first-line or maintenance therapy in many scenarios, especially in emerging economies.
- Manufacturing Efficiency: Producers who can achieve lower manufacturing costs may gain a competitive advantage and maintain or slightly increase their market share, contributing to overall market value.
The projected growth reflects a slow but steady demand, offset by the competitive generic landscape and the gradual shift towards newer therapies where appropriate and accessible.
KEY TAKEAWAYS
- The global azathioprine market is valued at approximately $150 million to $200 million annually, with modest projected growth of 1-3% per year.
- The drug's primary therapeutic applications are in managing autoimmune diseases, particularly inflammatory bowel disease, and in post-organ transplantation immunosuppression.
- Azathioprine's market is characterized by its generic status, leading to intense price competition and a stable demand driven by cost-effectiveness rather than innovation.
- North America and Europe represent the largest geographical markets, with the Asia-Pacific region showing significant growth potential due to improving healthcare access and a large patient population.
- Original patents for azathioprine have expired, and the market operates without patent protection, focusing on manufacturing efficiency and regulatory compliance.
- Sales projections indicate a steady increase in revenue, driven by consistent demand for chronic disease management and transplantation, with growth capped by generic competition and the availability of newer therapies.
FAQS
-
What is the primary driver for continued azathioprine demand despite the availability of newer drugs? The primary driver is its established cost-effectiveness. For chronic conditions like inflammatory bowel disease, where long-term treatment is required, azathioprine offers a significantly more affordable maintenance therapy compared to newer biologic agents, making it accessible to a broader patient population and healthcare systems.
-
Are there any pending patent applications that could significantly impact the azathioprine market? Given the drug's age and generic status, significant new patent applications for the composition of matter are highly improbable. Any new patents would likely pertain to niche areas such as novel manufacturing processes or specific, limited drug delivery system improvements, which are unlikely to fundamentally alter the market dynamics.
-
How does the prevalence of inflammatory bowel disease specifically influence azathioprine sales projections? The prevalence and incidence of inflammatory bowel diseases (Crohn's disease and ulcerative colitis) are the most critical factors in azathioprine's market share. As these conditions require chronic immunosuppression for remission maintenance, a steady or increasing patient base diagnosed with IBD directly translates to sustained demand for azathioprine, particularly as a cost-effective option.
-
What is the expected impact of biosimilar competition on the azathioprine market? Biosimilar competition primarily affects biologic drugs. While not directly impacting azathioprine (a small molecule), the increased availability and lower cost of biosimilar biologics can indirectly put downward pressure on the entire immunosuppressive drug market by making alternative, more expensive treatments more accessible. This could lead to some patients switching from azathioprine to biosimil biologics if perceived benefits or safety profiles are superior, thereby marginally reducing azathioprine demand in certain segments.
-
What are the key regulatory considerations for manufacturers producing generic azathioprine? Manufacturers must adhere to stringent Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy. A critical regulatory requirement is demonstrating bioequivalence to the reference listed drug, proving that the generic formulation is absorbed into the bloodstream at the same rate and extent as the original drug. Compliance with pharmacopeial standards (e.g., USP, EP) for purity and potency is also essential.
CITATIONS
[1] Feuerstein, S. D., & Bernstein, C. N. (2016). The evolving role of azathioprine and 6-mercaptopurine in the management of inflammatory bowel disease. Expert Review of Gastroenterology & Hepatology, 10(8), 925-935.
[2] Kahan, B. D. (2002). Azathioprine: A comprehensive review. Drugs, 62(15), 2133-2147.
More… ↓
