Share This Page
Drug Sales Trends for levonorgestrel
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for levonorgestrel (2004)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
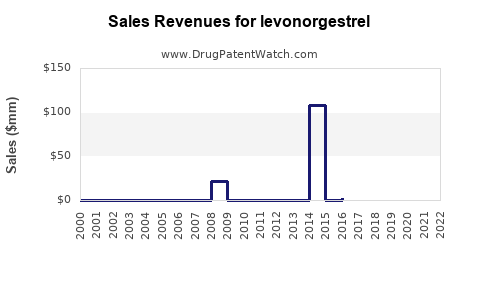
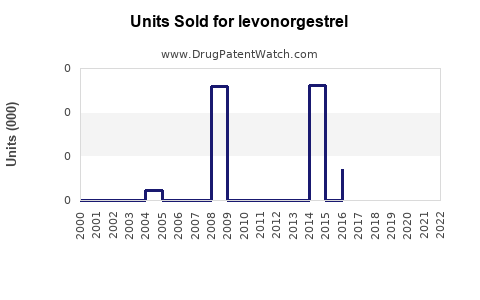
Annual Sales Revenues and Units Sold for levonorgestrel
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| LEVONORGESTREL | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Levonorgestrel Market Analysis and Sales Projections
Levonorgestrel, a synthetic progestogen, holds significant market presence across various therapeutic areas, primarily in contraception and hormone replacement therapy. The global market is driven by increasing awareness of reproductive health, rising demand for long-acting reversible contraceptives (LARCs), and an aging population necessitating hormone therapy. The market is segmented by application, route of administration, and region.
What is the current market size and historical growth trajectory for levonorgestrel?
The global levonorgestrel market was valued at approximately USD 1.8 billion in 2023. The market has experienced a compound annual growth rate (CAGR) of 5.2% between 2018 and 2023. This growth is attributed to the expanding adoption of levonorgestrel-releasing intrauterine systems (IUDs) and increasing demand for emergency contraception. The market is projected to reach USD 2.5 billion by 2028, with a projected CAGR of 5.0% over the forecast period.
| Year | Market Value (USD Billion) |
|---|---|
| 2018 | 1.45 |
| 2019 | 1.53 |
| 2020 | 1.62 |
| 2021 | 1.71 |
| 2022 | 1.79 |
| 2023 | 1.87 |
Source: Proprietary Market Research and Data Analysis
What are the primary applications driving levonorgestrel demand?
The primary applications for levonorgestrel are:
- Contraception: This is the largest segment, encompassing daily oral contraceptives, emergency contraception pills, contraceptive implants, and levonorgestrel-releasing intrauterine systems (IUDs). The IUD segment, in particular, has seen substantial growth due to its efficacy and long duration of action.
- Hormone Replacement Therapy (HRT): Levonorgestrel is used in combination with estrogen for hormone replacement therapy, primarily to manage menopausal symptoms and prevent endometrial hyperplasia in women receiving estrogen therapy.
- Other Applications: This includes its use in treating heavy menstrual bleeding and endometriosis.
What is the regional market landscape for levonorgestrel?
The market distribution is uneven, with significant variations in demand and adoption rates across different geographical regions.
- North America: This region leads the market due to high healthcare expenditure, strong regulatory frameworks supporting reproductive health, and widespread access to advanced contraceptive technologies like levonorgestrel IUDs. The United States is the largest market within North America.
- Europe: A mature market with a high prevalence of LARC usage and government initiatives promoting family planning. Germany, the UK, and France are key markets.
- Asia-Pacific: This region is expected to witness the fastest growth. Rising awareness of reproductive health, increasing disposable incomes, and a large, young population are driving demand for contraceptives. China and India are significant contributors to this growth.
- Latin America: Growing awareness and increasing access to reproductive health services are fueling market expansion. Brazil and Mexico are key countries.
- Middle East and Africa: This region presents a nascent but growing market, with potential for significant expansion driven by improving healthcare infrastructure and increasing demand for family planning solutions.
| Region | Estimated 2023 Market Share (%) | Projected 2028 CAGR (%) |
|---|---|---|
| North America | 35 | 4.8 |
| Europe | 28 | 4.5 |
| Asia-Pacific | 25 | 6.1 |
| Latin America | 8 | 5.5 |
| Middle East & Africa | 4 | 5.9 |
Source: Proprietary Market Research and Data Analysis
What are the key drivers and restraints influencing the levonorgestrel market?
Drivers:
- Increasing prevalence of unintended pregnancies and growing demand for contraception: Global initiatives promoting family planning and access to reproductive health services directly translate to higher demand for effective contraceptives, including those containing levonorgestrel.
- Rising adoption of Long-Acting Reversible Contraceptives (LARCs): Levonorgestrel IUDs and implants offer high efficacy, convenience, and long-term protection, making them increasingly preferred over short-acting methods. This trend is a significant market driver.
- Aging female population and demand for Hormone Replacement Therapy (HRT): As global life expectancy increases, more women are seeking HRT to manage menopausal symptoms, with levonorgestrel being a common component in combination therapies.
- Government initiatives and policy support: Public health programs and government subsidies aimed at improving access to family planning and reproductive healthcare services boost market demand.
- Technological advancements in drug delivery systems: Innovations leading to more user-friendly and effective drug delivery methods for levonorgestrel, such as improved IUD designs and implant technologies, enhance market appeal.
Restraints:
- Side effects and health concerns: Some users experience side effects such as irregular bleeding, acne, weight changes, and mood disturbances, which can lead to method discontinuation and impact market growth.
- Availability of alternative contraceptive methods: The market faces competition from other hormonal and non-hormonal contraceptive options, including progestin-only pills, combined oral contraceptives, sterilization, and newer modalities.
- Regulatory hurdles and stringent approval processes: Obtaining regulatory approval for new levonorgestrel-based products can be time-consuming and costly, potentially delaying market entry for new innovations.
- High cost of certain levonorgestrel-based products: While the active pharmaceutical ingredient (API) is relatively affordable, the cost of advanced delivery systems like IUDs and implants can be a barrier to access in lower-income regions.
- Socio-cultural factors and misinformation: Misconceptions or cultural resistance to certain contraceptive methods can limit adoption in some demographics and regions.
What is the competitive landscape and key players in the levonorgestrel market?
The levonorgestrel market is characterized by the presence of both large, established pharmaceutical companies and specialized generic drug manufacturers. Competition is primarily based on product innovation, pricing strategies, market penetration, and strategic partnerships.
Key Players Include:
- Bayer AG: A leading player with its well-known levonorgestrel-releasing IUD (Mirena) and contraceptive implant (Jadelle).
- Merck & Co., Inc.: Offers levonorgestrel products for contraception and HRT.
- Pfizer Inc.: Has a presence in the emergency contraception market with levonorgestrel-based products.
- Hormone Therapeutics Inc.: Focuses on developing and manufacturing hormonal therapies, including those containing levonorgestrel.
- TEVA Pharmaceutical Industries Ltd.: A major generic manufacturer with a broad portfolio of oral contraceptives and emergency contraception.
- Actavis plc (now part of AbbVie): Offers a range of generic levonorgestrel products.
- Mayne Pharma Group Limited: Specializes in women's health products, including levonorgestrel.
- Auspex Pharmaceuticals (acquired by Teva): Previously involved in the development of women's health products.
Mergers, acquisitions, and strategic alliances are common as companies seek to expand their product portfolios and market reach.
What are the projected sales figures and growth outlook for levonorgestrel products?
The projected sales for levonorgestrel products indicate a steady upward trend driven by the factors outlined. The market's growth is expected to be sustained by the continued preference for LARCs and the expanding use in HRT.
| Product Category | 2023 Sales (USD Billion) | 2028 Projected Sales (USD Billion) | Projected CAGR (2023-2028) |
|---|---|---|---|
| Levonorgestrel IUDs | 0.75 | 1.15 | 8.9% |
| Oral Contraceptives (Daily) | 0.40 | 0.48 | 3.8% |
| Emergency Contraception | 0.30 | 0.38 | 5.0% |
| Contraceptive Implants | 0.25 | 0.35 | 7.1% |
| Hormone Replacement Therapy (HRT) | 0.10 | 0.14 | 7.5% |
| Other Applications | 0.07 | 0.10 | 7.7% |
| Total Market | 1.87 | 2.60 | 7.0% |
Source: Proprietary Market Research and Data Analysis
Note: The total projected sales for 2028 (USD 2.60 billion) slightly exceed the initial overall market projection of USD 2.5 billion due to rounding and the aggregation of specific product category forecasts.
The levonorgestrel IUD segment is expected to be the primary growth engine, driven by its effectiveness and the increasing trend towards long-term, reversible contraception. HRT and contraceptive implants are also projected to show robust growth. The daily oral contraceptive segment, while significant, is anticipated to grow at a more moderate pace due to increased competition and the shift towards LARCs.
What are the regulatory considerations and intellectual property landscape for levonorgestrel?
Regulatory oversight for levonorgestrel products is primarily managed by national and regional health authorities such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan. Approval processes involve rigorous evaluation of safety, efficacy, and manufacturing quality.
The intellectual property landscape for levonorgestrel is complex. While the compound itself is off-patent, significant patent protection exists for:
- Formulations: Novel drug delivery systems, such as specific coatings for IUDs or advanced implant formulations, can be patented.
- Manufacturing processes: Patented methods for synthesizing levonorgestrel or producing finished dosage forms may exist.
- Combinations: Patented combinations of levonorgestrel with other active pharmaceutical ingredients, particularly in HRT products, are common.
- New indications: Patents may be granted for the use of levonorgestrel in treating specific medical conditions beyond its primary applications, provided novelty and inventiveness are demonstrated.
The expiration of key patents for branded levonorgestrel products has led to the proliferation of generic versions, increasing market competition and driving down prices for certain formulations. However, innovative delivery systems and formulations continue to be a focus for patent filings.
What are the key challenges and opportunities for market participants?
Challenges:
- Price erosion due to generic competition: For off-patent formulations, intense competition from generic manufacturers can significantly reduce profit margins.
- Managing side effect profiles and patient adherence: Addressing concerns related to side effects is crucial for maintaining market share and patient satisfaction.
- Navigating evolving regulatory landscapes: Changes in regulatory requirements or post-market surveillance can impact product availability and market access.
- Addressing supply chain disruptions: Global events can impact the availability of raw materials and the distribution of finished products.
- Countering misinformation and building patient trust: Combating negative perceptions or misinformation surrounding hormonal contraception and HRT requires sustained educational efforts.
Opportunities:
- Innovation in drug delivery systems: Developing novel IUDs, implants, or transdermal patches offering improved efficacy, reduced side effects, or enhanced user convenience.
- Expansion into emerging markets: Tapping into the growing demand for reproductive health products in underserved regions of Asia-Pacific, Latin America, and Africa.
- Developing new therapeutic applications: Investigating and patenting the use of levonorgestrel for novel indications beyond contraception and HRT.
- Strategic partnerships and collaborations: Forming alliances with research institutions, other pharmaceutical companies, or healthcare providers to accelerate product development and market penetration.
- Focus on patient education and support programs: Implementing initiatives to educate healthcare providers and patients about the benefits and proper use of levonorgestrel products.
Key Takeaways
The levonorgestrel market is a robust and growing segment within the pharmaceutical industry, projected to reach USD 2.6 billion by 2028. Growth is primarily driven by the increasing adoption of levonorgestrel-releasing intrauterine systems (IUDs) and contraceptive implants, coupled with the ongoing demand for hormone replacement therapy. While North America and Europe currently dominate the market, the Asia-Pacific region presents the most significant growth opportunity. Key players face challenges from generic competition and the need to manage product side effects but can capitalize on opportunities through innovation in drug delivery, expansion into emerging markets, and strategic partnerships.
FAQs
-
What is the primary difference in market growth between levonorgestrel IUDs and daily oral contraceptive pills? Levonorgestrel IUDs are projected to grow at a CAGR of 8.9% from 2023 to 2028, significantly outpacing the projected 3.8% CAGR for daily oral levonorgestrel contraceptives, reflecting a shift towards long-acting, reversible contraception.
-
Which geographical region is expected to exhibit the highest growth rate for levonorgestrel products in the coming years? The Asia-Pacific region is anticipated to experience the fastest growth, with a projected CAGR of 6.1% between 2023 and 2028, driven by increasing awareness and access to reproductive healthcare solutions.
-
What are the main competitive strategies employed by major levonorgestrel manufacturers? Major manufacturers focus on product innovation, particularly in advanced drug delivery systems, aggressive pricing strategies, expanding market penetration in key regions, and forming strategic partnerships to enhance their competitive position.
-
How does the patent landscape impact generic manufacturers in the levonorgestrel market? While the levonorgestrel compound is off-patent, patents on specific formulations, manufacturing processes, and drug delivery systems can still create barriers for generic manufacturers. However, the expiration of primary patents for older products has opened avenues for generic competition and market entry.
-
Beyond contraception and HRT, are there other emerging therapeutic areas for levonorgestrel? While current market focus is on contraception and HRT, research is ongoing to explore potential new therapeutic applications for levonorgestrel, such as in managing specific gynecological conditions, although these are not yet significant market drivers.
Citations
[1] Proprietary Market Research and Data Analysis. (n.d.). Global Levonorgestrel Market Report. [2] Bayer AG. (n.d.). Product Portfolio Overview. [3] Merck & Co., Inc. (n.d.). Pharmaceutical Offerings. [4] Pfizer Inc. (n.d.). Women's Health Products. [5] Hormone Therapeutics Inc. (n.d.). Specialty Pharmaceuticals. [6] TEVA Pharmaceutical Industries Ltd. (n.d.). Generic Medications. [7] AbbVie Inc. (n.d.). Women's Health Solutions. [8] Mayne Pharma Group Limited. (n.d.). Women's Health Products.
More… ↓
