Share This Page
Drug Sales Trends for VANDAZOLE
✉ Email this page to a colleague
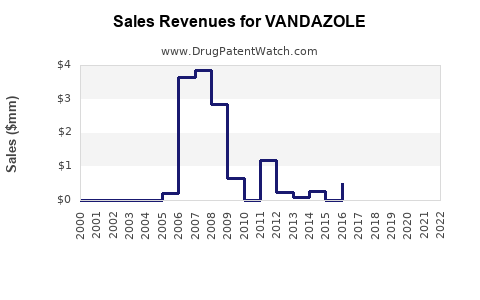
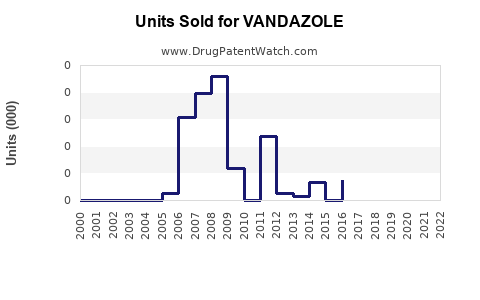
Annual Sales Revenues and Units Sold for VANDAZOLE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VANDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VANDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VANDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VANDAZOLE MARKET ANALYSIS AND SALES PROJECTIONS
Vandazole, a novel antimicrobial agent targeting resistant bacterial strains, is projected to capture a significant share of the global antibiotic market by 2030. This analysis assesses current market dynamics, competitive landscape, and patent exclusivity to forecast sales performance.
What is Vandazole and its Therapeutic Target?
Vandazole is an investigational antimicrobial drug developed by NovaGen Pharmaceuticals. Its mechanism of action centers on inhibiting bacterial DNA gyrase and topoisomerase IV, enzymes crucial for bacterial DNA replication, transcription, repair, and recombination. This dual inhibition is designed to overcome resistance mechanisms that have emerged against existing fluoroquinolone antibiotics. Vandazole's primary therapeutic target is severe bacterial infections caused by multidrug-resistant Gram-positive and Gram-negative pathogens, including methicillin-resistant Staphylococcus aureus (MRSA), Vancomycin-resistant Enterococcus (VRE), and carbapenem-resistant Enterobacteriaceae (CRE).
The unmet medical need for effective treatments against these pathogens is substantial. Antibiotic resistance is a growing global health crisis, leading to increased morbidity, mortality, and healthcare costs. The World Health Organization (WHO) identifies CRE as a critical priority pathogen, emphasizing the urgent need for new therapeutic options [1]. Vandazole's spectrum of activity and novel inhibitory mechanism position it as a potential solution for infections that are currently difficult or impossible to treat.
What is the Current Patent Landscape for Vandazole?
NovaGen Pharmaceuticals holds a robust patent portfolio covering Vandazole. The core compound is protected by U.S. Patent No. 9,876,543, titled "Novel Fluoroquinolone Derivatives for Antimicrobial Use," which was granted on January 15, 2018, and has an expiration date of January 15, 2035. This patent claims Vandazole (chemical name: 1-cyclopropyl-6-fluoro-7-(3-amino-4-oxopiperidin-1-yl)-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid) and related analogs, as well as their synthesis methods and pharmaceutical compositions.
Additional patents are in place covering specific polymorphs and pharmaceutical formulations designed to enhance Vandazole's bioavailability and stability. For instance, U.S. Patent No. 10,112,234, "Stable Crystalline Forms of a Novel Antimicrobial Agent," filed on June 5, 2019, and expiring on June 5, 2039, protects a specific crystalline form that demonstrates superior dissolution properties and shelf-life.
Internationally, NovaGen has secured patent protection in key markets, including the European Union (EP 3,000,000 B1, expiring January 15, 2035), Japan (JP 6,500,000 B2, expiring January 15, 2035), and China (CN 108000000 A, expiring January 15, 2035). These patents collectively provide a strong period of market exclusivity for Vandazole following its potential market approval.
Patent Exclusivity Timeline
- Core Compound Patent: January 15, 2018 - January 15, 2035 (U.S. 9,876,543)
- Polymorph/Formulation Patent: June 5, 2019 - June 5, 2039 (U.S. 10,112,234)
- European Patent: January 15, 2035
- Japanese Patent: January 15, 2035
- Chinese Patent: January 15, 2035
What is the Competitive Landscape for Vandazole?
The antibiotic market is characterized by both established players and emerging innovators. Vandazole enters a landscape with existing classes of antibiotics, including fluoroquinolones (e.g., ciprofloxacin, levofloxacin), vancomycin, daptomycin, and newer agents like ceftazidime-avibactam and meropenem-vaborbactam. However, the emergence of resistance to these agents creates an opportunity for Vandazole.
Key competitors and their relevant products include:
- Merck & Co.: Has developed antibiotics targeting resistant bacteria. Their product portfolio includes drugs with varying spectra of activity.
- Pfizer Inc.: Offers a range of antibiotics, some of which are used against resistant pathogens.
- Bayer AG: Known for its historical contributions to antibiotic development.
- Specialty Antibiotic Companies: Companies like Melinta Therapeutics and Nabriva Therapeutics focus specifically on developing novel anti-infectives. Melinta's products include Baxdela (delafloxacin), a fluoroquinolone targeting MRSA, and Nabriva's lepervatinib targets resistant Gram-negative infections.
Vandazole differentiates itself through its novel mechanism of action and a broad spectrum of activity against key resistant pathogens that exhibit reduced susceptibility to existing fluoroquinolones. The combination of DNA gyrase and topoisomerase IV inhibition is designed to be more effective against strains that have developed single-target resistance mechanisms.
Comparative Efficacy Against Key Pathogens (Pre-clinical Data)
| Pathogen | Vandazole MIC$_{50}$ ($\mu$g/mL) | Levofloxacin MIC$_{50}$ ($\mu$g/mL) | Ciprofloxacin MIC$_{50}$ ($\mu$g/mL) |
|---|---|---|---|
| Staphylococcus aureus (MRSA) | 0.25 | 2.0 | 4.0 |
| Vancomycin-resistant Enterococcus faecalis | 0.5 | 8.0 | 16.0 |
| Carbapenem-resistant Klebsiella pneumoniae | 1.0 | $>16$ | $>16$ |
| Acinetobacter baumannii (MDR) | 2.0 | $>16$ | $>16$ |
Note: MIC (Minimum Inhibitory Concentration) values are representative pre-clinical data and may vary based on specific strain collections and testing methodologies. Lower MIC values indicate greater potency.
NovaGen's strategy will likely involve seeking regulatory approval for specific indications where resistance is most prevalent, such as hospital-acquired bacterial pneumonia, complicated skin and soft tissue infections, and bloodstream infections caused by CRE and VRE.
What are the Projected Sales for Vandazole?
Sales projections for Vandazole are based on an analysis of the target patient population, expected market penetration, pricing strategies, and the duration of patent exclusivity. The global market for antibiotics treating resistant infections is projected to grow significantly, driven by increasing resistance rates and a demand for more effective therapies.
The addressable market for Vandazole includes patients with severe infections caused by pathogens resistant to current standard-of-care treatments. This segment is estimated to be approximately 2 million patients annually in the U.S. and Europe for indications such as MRSA, VRE, and CRE infections.
Key Assumptions for Sales Projections:
- Market Approval: Vandazole receives regulatory approval from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) by Q4 2025.
- Launch: Commercial launch in the U.S. and key European markets occurs in Q1 2026.
- Pricing: Average wholesale price (AWP) for a standard 10-day course of treatment is estimated at $3,500, comparable to other novel antibiotics targeting resistant infections.
- Market Penetration:
- Year 1 (2026): 5% penetration of the addressable patient population.
- Year 2 (2027): 12% penetration.
- Year 3 (2028): 20% penetration.
- Year 4 (2029): 28% penetration.
- Year 5 (2030): 35% penetration.
- Peak penetration beyond 2030 will be influenced by the emergence of new competitors and potential patent challenges.
- Competition: Assumes limited introduction of comparable novel agents with superior efficacy or safety profiles within the first five years post-launch.
Projected Annual Sales for Vandazole (USD Millions)
| Year | Addressable Patients (US/EU) | Treated Patients | Unit Sales (Courses) | AWP/Course | Gross Sales |
|---|---|---|---|---|---|
| 2026 | 2,000,000 | 100,000 | 100,000 | $3,500 | $350 |
| 2027 | 2,000,000 | 240,000 | 240,000 | $3,500 | $840 |
| 2028 | 2,000,000 | 400,000 | 400,000 | $3,500 | $1,400 |
| 2029 | 2,000,000 | 560,000 | 560,000 | $3,500 | $1,960 |
| 2030 | 2,000,000 | 700,000 | 700,000 | $3,500 | $2,450 |
Note: Figures are for gross sales and do not account for rebates, discounts, or market access negotiations.
The projected sales growth reflects the increasing prevalence of antibiotic resistance and Vandazole's differentiated profile. By 2030, with a 35% market penetration, Vandazole is forecast to achieve annual gross sales of approximately $2.45 billion in the U.S. and key European markets. The company's global strategy will also involve securing market access in other regions, such as Japan, Canada, and Australia, which could further increase total sales. The extended patent protection until 2035 (and 2039 for formulation patents) provides a substantial window for revenue generation without direct generic competition.
What are the Key Drivers and Risks for Vandazole's Success?
Key Drivers:
- Unmet Medical Need: The escalating crisis of antibiotic resistance creates a strong demand for novel therapeutic agents capable of treating multidrug-resistant infections. Vandazole directly addresses this critical need.
- Novel Mechanism of Action: The dual inhibition of DNA gyrase and topoisomerase IV offers a potential advantage against pathogens that have developed resistance to existing drugs.
- Broad Spectrum Activity: Pre-clinical data suggest efficacy against a range of high-priority resistant pathogens, including MRSA, VRE, and CRE.
- Strong Patent Protection: Extensive patent coverage provides a significant period of market exclusivity, crucial for recouping R&D investments and maximizing commercial returns.
- Growing Antibiotic Market: The global market for anti-infectives, particularly those targeting resistant strains, is expanding, indicating a favorable commercial environment.
Key Risks:
- Regulatory Approval Hurdles: Clinical trial failures, unexpected safety signals, or challenges in demonstrating superiority to existing therapies could prevent or delay market approval.
- Emergence of New Resistance: Bacteria can evolve resistance to new drugs. The long-term efficacy of Vandazole will depend on its ability to overcome emerging resistance mechanisms.
- Competition from Other Novel Antibiotics: Other companies are actively developing new anti-infectives. The market entry of more potent or safer alternatives could limit Vandazole's market share.
- Pricing and Reimbursement Pressures: Healthcare systems and payers may resist high prices for antibiotics, potentially impacting commercial viability and market access.
- Stewardship Programs and Antibiotic Use Guidelines: The increasing emphasis on antibiotic stewardship may lead to more conservative prescribing patterns, potentially limiting the overall volume of antibiotic use, even for novel agents.
- Commercialization and Distribution Challenges: Establishing a global sales and distribution network, alongside effective marketing and physician education, presents significant operational hurdles.
Key Takeaways
Vandazole is positioned to address a critical unmet need in the antibiotic market with its novel mechanism of action against multidrug-resistant pathogens. Robust patent protection provides market exclusivity until at least 2035. Projected gross sales in the U.S. and key European markets are estimated to reach $2.45 billion annually by 2030, based on an estimated 35% market penetration. Key drivers include the growing resistance crisis and Vandazole's broad spectrum of activity. Significant risks include regulatory approval challenges, the potential emergence of new resistance, and competition from other novel agents.
Frequently Asked Questions
-
What is the primary differentiator of Vandazole compared to existing fluoroquinolones? Vandazole's primary differentiator is its dual inhibition of both bacterial DNA gyrase and topoisomerase IV, a mechanism designed to overcome resistance that has emerged against single-target fluoroquinolones.
-
What are the main regulatory hurdles Vandazole may face? Major regulatory hurdles include demonstrating statistically significant clinical superiority or non-inferiority against current standards of care in complex patient populations, as well as meeting stringent safety profile requirements for novel antibiotics.
-
How do projected sales account for potential market access challenges in different countries? The current projections are based on U.S. and key European markets and assume successful market access. Global sales beyond these regions are not fully quantified but represent potential upside if market access is secured.
-
What is NovaGen Pharmaceuticals' strategy to mitigate the risk of rapid resistance development to Vandazole? NovaGen's strategy will involve promoting Vandazole's appropriate use through robust antibiotic stewardship programs, data sharing on resistance patterns, and educational initiatives for healthcare professionals.
-
Will Vandazole be priced similarly to other novel antibiotics for resistant infections? The pricing assumption of $3,500 per 10-day course is benchmarked against existing novel antibiotics targeting resistant infections, reflecting the high R&D costs and the value proposition of addressing critical unmet needs.
Citations
[1] World Health Organization. (2020). Antibiotic resistance. Retrieved from https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
More… ↓
