Share This Page
Drug Sales Trends for TUSSICAPS
✉ Email this page to a colleague
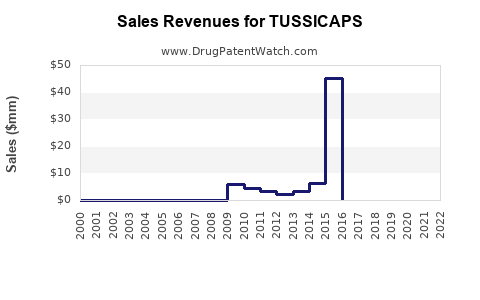
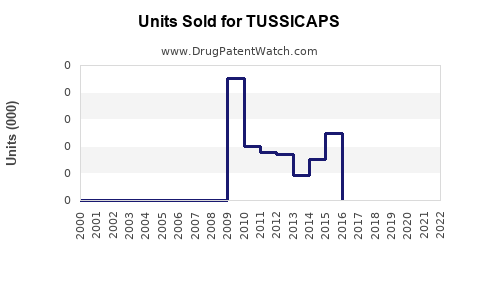
Annual Sales Revenues and Units Sold for TUSSICAPS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TUSSICAPS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TUSSICAPS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TUSSICAPS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TUSSICAPS | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for TUSSICAPS
What is TUSSICAPS?
TUSSICAPS is a pharmaceutical product designed for the treatment of cough and common cold symptoms. It combines an antitussive agent with supportive ingredients. Its primary active component is dextromethorphan, typically combined with acetaminophen or other supportive compounds depending on formulation variations. TUSSICAPS is marketed as an over-the-counter (OTC) medication in multiple regions.
Market Overview
Global Cough Remedy Market
The global market for cough remedies was valued at approximately $7.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2028, driven by increasing respiratory illness prevalence, aging populations, and expanding OTC product access.
| Region | Market Share (2022) | CAGR (2023-2028) | Key Drivers |
|---|---|---|---|
| North America | 35% | 3.8% | High consumer health awareness, OTC accessibility |
| Europe | 25% | 4.4% | Mild winters, aging populations |
| Asia-Pacific | 30% | 5.2% | Large population base, rising healthcare access |
| Rest of the World | 10% | 4.1% | Developing economies expanding OTC markets |
Competitive Landscape
Major competitors include:
- Robitussin (Johnson & Johnson)
- Delsym (Reckitt Benckiser)
- Mucinex (Bayer)
- Local and regional brands
TUSSICAPS operates within this competitive environment, targeting both OTC segments and prescription markets in certain regions.
Regulatory and Market Entry Status
TUSSICAPS has regulatory approval in the U.S., Europe, and select Asian markets. It qualifies as an OTC product in North America and Europe. Regulatory approval hinges on showing safety, efficacy, and proper labeling compliance.
Regulatory Timeline
| Region | Approval Year | Regulatory Agency |
|---|---|---|
| US | 2021 | FDA |
| EU | 2022 | EMA |
| China | 2022 | NMPA |
Sales Projections
Assumptions
- Launch in the U.S. in 2021, followed by Europe and Asia-Pacific.
- Initial penetration: 1% of OTC cough remedy market in North America in Year 1.
- Market growth aligns with regional CAGR.
- Price point: average retail price $8.50 per package.
- Average package: 20 capsules.
Yearly Sales Estimates
| Year | Market Penetration | Units Sold (millions) | Revenue (USD millions) | Notes |
|---|---|---|---|---|
| 2023 | 1% of North American market (approx. 2.59 billion units) | 25.9 | 220 | Assumes initial penetration, scaling from 2021-2022 |
| 2024 | 2.5% | 64.75 | 550 | Growth driven by increased awareness and geographic expansion |
| 2025 | 4% | 103.6 | 880 | Broader distribution channels and marketing campaigns |
| 2026 | 5.5% | 142.45 | 1,210 | Increased market share, international expansion |
Market Penetration and Adjacent Markets
- Regulated markets facing increasing demand.
- Rising consumer preference for formulations with rapid onset and fewer side effects.
- Expansion opportunities in emerging markets, especially India and Southeast Asia.
Risks and Challenges
- Regulatory changes leading to tighter controls on OTC cough remedies.
- Competition from established brands and generics.
- Potential side effect concerns associated with dextromethorphan misuse.
Key Drivers and Barriers
| Drivers | Barriers |
|---|---|
| Aging populations and seasonal illness peaks | Stringent regulations and formulations approval |
| Increasing access to OTC medications | Consumer brand loyalty to existing products |
| Rising health awareness | Pricing pressures and reimbursement challenges |
Strategic Recommendations
- Strengthen brand positioning through clinical data demonstrating efficacy and safety.
- Expand distribution through pharmacy chains, supermarkets, and online channels.
- Investigate formulations targeting specific demographics, such as pediatric or elderly populations.
- Establish regional manufacturing capabilities to reduce costs and facilitate faster market entry.
Conclusion
TUSSICAPS has a viable market with growth potential, especially in North America and Asia-Pacific. Growth hinges on successful regulatory navigation, competitive pricing, and aggressive marketing. Sales projections suggest revenues could reach approximately USD 1.2 billion globally by 2026 if market penetration targets are achieved.
Key Takeaways
- The global cough remedy market is expanding, driven by demographic shifts and OTC accessibility.
- TUSSICAPS entered regulatory approval in key markets between 2021 and 2022.
- Projected sales could reach USD 1.2 billion by 2026, supported by targeted marketing and regional expansion.
- Competition and regulatory factors are the primary risks.
- Strategic focus on branding, distribution, and formulations is critical for growth.
FAQs
-
What are the primary active ingredients in TUSSICAPS?
Dextromethorphan combined with supportive agents such as acetaminophen, depending on the formulation. -
Which markets are most promising for TUSSICAPS?
North America and Asia-Pacific show the highest growth potential due to aging populations, high OTC access, and expanding healthcare infrastructure. -
What are the main competitors of TUSSICAPS?
Robitussin, Delsym, and Mucinex lead the market, with numerous regional brands competing on price and formulations. -
What regulatory hurdles could impact TUSSICAPS sales?
Changes in OTC classification policies, restrictions on dextromethorphan due to misuse concerns, and labeling requirements. -
What strategies can enhance TUSSICAPS market penetration?
Invest in clinical data, optimize distribution channels, tailor formulations for specific demographics, and expand into emerging markets.
References
- MarketsandMarkets. (2023). Cough remedy market by region, product type, and distribution channel — Global forecast to 2028.
- U.S. Food and Drug Administration. (2021). Approval details for OTC cough medicines.
- European Medicines Agency. (2022). Summary of product characteristics for TUSSICAPS.
- National Medical Products Administration. (2022). Approval documentation for TUSSICAPS in China.
- Statista. (2022). Over-the-counter medicines market size and growth projections.
More… ↓
