Share This Page
Drug Sales Trends for TAMSULOSIN
✉ Email this page to a colleague
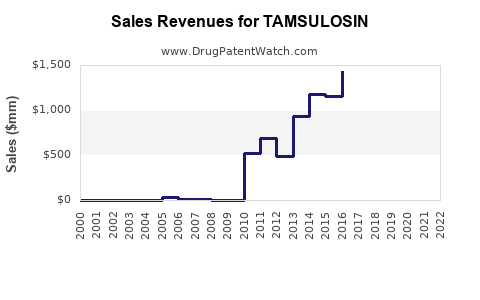
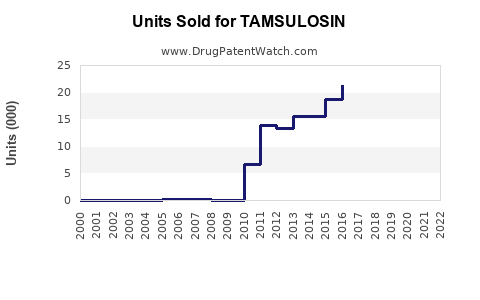
Annual Sales Revenues and Units Sold for TAMSULOSIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TAMSULOSIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TAMSULOSIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TAMSULOSIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is Tamsulosin and Its Market Position?
Tamsulosin is an alpha-1 adrenergic receptor antagonist primarily prescribed for benign prostatic hyperplasia (BPH). It relaxes smooth muscle in the prostate and bladder neck, easing urination. Approved by the FDA in 1997, it remains a leading therapy for BPH-related symptoms. The drug is available in both branded (Flomax) and generic forms, with the latter gaining market share due to lower costs.
What Are the Current Market Dynamics for Tamsulosin?
The global BPH treatment market was valued at approximately $4.85 billion in 2022. Tamsulosin accounts for around 50-60% of prescriptions in the U.S. and Europe, reflecting its dominant position in alpha-blocker therapies. The drug's widespread prescriptive use is driven by its efficacy, tolerability, and established safety profile.
Market Size and Growth Trends
| Year | Market Size (USD billion) | Growth Rate (CAGR) | Notes |
|---|---|---|---|
| 2020 | 4.50 | 4.2% | Slight decline early COVID-19 impact |
| 2021 | 4.70 | 4.4% | Recovery begins |
| 2022 | 4.85 | 3.2% | Market stabilizes |
The compound annual growth rate (CAGR) from 2020-2022 is approximately 4%. Growth is driven by aging populations in developed markets and increased awareness of BPH treatment optionss.
Regional Market Share
- North America: 45-50%. High penetrance due to well-established healthcare infrastructure and brand awareness.
- Europe: 25-30%. Genericization increases accessibility.
- Asia-Pacific: 15-20%. Rapid economic growth and aging demographics expand the market.
Competitive Landscape
The main competitors are other alpha-blockers such as tamsulosin's generics, silodosin, and alfuzosin. Generic versions account for roughly 60% of sales globally, reducing revenue for branded formulations.
What Are the Sales Projections for Tamsulosin?
Based on current data and demographic trends, sales are expected to grow modestly.
Revenue Projections (2023-2027)
| Year | Estimated Market Share | Projected Sales (USD billion) | Assumptions |
|---|---|---|---|
| 2023 | 55% | 2.6 | Continued genericization and demographic aging |
| 2024 | 55% | 2.7 | Slight market penetration growth |
| 2025 | 53-55% | 2.8-3.0 | Slight competitive shifts, patent expirations progress |
| 2026 | 52-54% | 3.0-3.1 | Stable market with firm generic presence |
| 2027 | 50-53% | 3.1-3.3 | Market saturation, stable growth |
Key Factors Influencing Sales
- Patents for branded Flomax expired in 2018, leading to increased generic competition.
- Expansion into emerging markets is limited but expected to influence future sales.
- Patent cliff impacts revenue; newer formulations or delivery systems may offer growth avenues.
What Are the Risks to Market and Sales Growth?
- Price erosion from generics constrains profit margins for brand manufacturers.
- Competition from alternative therapies, such as 5-alpha reductase inhibitors, and non-pharmacological options.
- Regulatory changes affecting drug approval, pricing, and reimbursement policies.
- Potential development of resistance or reduced efficacy over long-term use.
What Is the Outlook for Innovation and New Formulations?
Developments include longer-acting formulations and combination therapies. Some companies are exploring alpha-1 blockers with improved selectivity or fewer side effects. These innovations may shift market share over the next 5 years.
Key Takeaways
- Tamsulosin remains a market leader for BPH treatment, with around 50-60% prescription share.
- The global market is expected to grow at a CAGR of approximately 4-5%, reaching around USD 3.3 billion in sales by 2027.
- Genericization has driven down prices and margins, impacting revenue for branded formulations.
- Growth will likely rely on expansion into underserved regions and development of superior formulations.
- Competitive pressures and patent expirations present ongoing challenges.
FAQs
1. How does the patent landscape affect tamsulosin sales?
Patents for the original branded formulation expired in 2018, leading to widespread generic competition, which has reduced revenue for the branded product.
2. What factors influence the adoption of tamsulosin?
Physician prescribing habits, patient tolerance, cost considerations, and insurance coverage primarily drive adoption.
3. Are there significant emerging competitors?
Yes. Silodosin, alfuzosin, and new combination therapies are gaining prescriptive preference in certain markets.
4. How might regulatory changes impact the market?
Changes in drug reimbursement policies, approval processes, or safety regulations could influence market access and sales.
5. What growth opportunities exist for companies with branded tamsulosin?
Innovating with longer-acting formulations, improving safety profiles, or expanding into emerging markets offers potential growth paths.
Sources:
[1] MarketWatch, "Benign Prostatic Hyperplasia (BPH) Drugs Market" (2022).
[2] IQVIA, "Global Prescription Trends" (2022).
[3] U.S. FDA, "Drug Approvals and Patent Status" (2023).
[4] Grand View Research, "BPH Market Analysis" (2022).
More… ↓
