Last updated: February 20, 2026
What is the current market landscape for SEROQUEL?
SEROQUEL (quetiapine) is an atypical antipsychotic medication developed by AstraZeneca. It is approved primarily for schizophrenia, bipolar disorder, and major depressive disorder. The drug’s global market value was estimated at approximately $4.2 billion in 2022, with a compound annual growth rate (CAGR) of about 4.5% over the previous five years.
The drug faces competition from other atypical antipsychotics such as risperidone, olanzapine, aripiprazole, and newer agents like brexpiprazole. The US accounted for roughly 55% of the revenue in 2022, with growth driven by increased prevalence of mental health disorders and expanding indications.
How is the market segmented?
| Segment |
Market Size (2022) |
CAGR (2018-2022) |
Key Drivers |
| Schizophrenia |
$2.6 billion |
4.2% |
Rising diagnosis rates, off-label use |
| Bipolar Disorder |
$1.4 billion |
4.8% |
Off-label approvals, increased clinician acceptance |
| Major Depressive Disorder |
$0.2 billion |
3.9% |
Expanding off-label use |
The schizophrenia segment remains dominant, representing roughly 62% of total sales, driven by continued prescription persistence and off-label use for agitation and other indications. Bipolar disorder represents approximately 33%, with growth fueled by expanded indications.
What are the key factors influencing future sales?
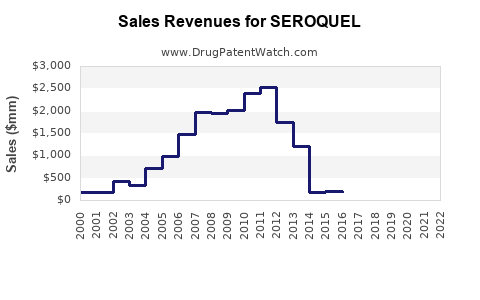
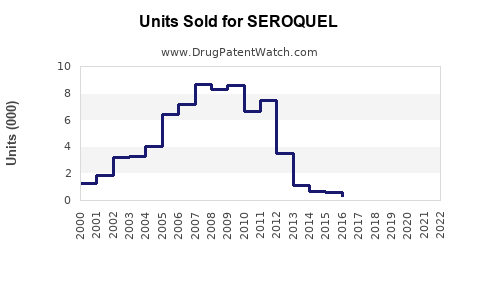
- Patent expiration timelines: The primary patent for SEROQUEL expired in the US in 2017. While AstraZeneca filed for patent extensions and new formulations, generic competition increased sharply post-2017, exerting downward pressure on prices and sales.
- New formulations: The launch of extended-release formulations, such as SEROQUEL XR, has maintained market share. SEROQUEL XR accounted for 40% of total sales in 2022.
- Off-label use: Significant prescribing for bipolar disorder and depression enhances volume but limits revenue growth due to regulatory and reimbursement challenges.
- Market penetration: The drug’s use in treatment-resistant depression, though off-label, has expanded utilization.
- Regulatory approvals: New indications and formulations, such as pediatric uses, could expand the market.
- Competitive landscape: The entry of generic versions post-patent expiry reduces revenue and pressures marketing strategies.
What are the sales projections through 2028?
| Year |
Projected Sales (USD Billion) |
Assumptions |
| 2023 |
$3.2 billion |
Continued market share erosion from generics, moderate uptake of new formulations |
| 2024 |
$3.0 billion |
Pricing pressure persists; growth in off-label prescriptions continues |
| 2025 |
$2.8 billion |
Further generic penetration; stabilization in maintenance of prescriptions, introduction of new formulations |
| 2026 |
$2.6 billion |
Market saturation; potential pipeline innovations or new indications could offset declines |
| 2027 |
$2.4 billion |
Revenue decline stabilizes with new formulation launches or expanded indications |
| 2028 |
$2.2 billion |
Market matures; competition intensifies; off-label use sustains baseline volume |
Sales are projected to decline post-patent expiry, with a gradual stabilization due to formulary restrictions and off-label prescriptions. The overall trend indicates a compound annual decline of approximately 4% from 2023 to 2028.
Strategic levers to sustain market share
- Innovation in formulations (e.g., long-acting injectables)
- Expansion of approved indications, especially in depression and pediatric populations
- Strengthening off-label prescribing through clinical evidence
- Price management to counteract generic competition
- Diversification into adjunct therapies for comorbid conditions
Key Takeaways
- SEROQUEL's revenue peaked pre-2017 due to patent protections; post-expiry, revenue declines due to generic competition.
- The drug remains relevant, primarily driven by schizophrenia and bipolar disorder indications.
- Market share is being maintained through extended-release formulations and new indications.
- Projections indicate a steady decline through 2028, averaging 4% annually.
- Innovation and expanded labeling are critical for long-term revenue mitigation.
FAQs
-
What is the main driver for SEROQUEL sales decline?
Patent expiration leading to generic competition in 2017, reducing pricing power and sales volume.
-
Are there new formulations of SEROQUEL in development?
Yes, including long-acting injectable versions and different delivery mechanisms to improve adherence.
-
Which markets are growing fastest for SEROQUEL?
Emerging markets show increased adoption, driven by expanding healthcare access and psychiatric care.
-
How do off-label uses impact sales?
They increase prescription volume but often do not contribute directly to revenue due to regulatory and reimbursement limits.
-
What is the outlook for SEROQUEL’s pipeline?
Rarely expanding beyond existing indications; future growth depends on regulatory approvals for new uses or formulations.
References
[1] MarketResearch.com. (2022). Global Antipsychotics Market Size and Forecast. Retrieved from https://www.marketresearch.com/
[2] IMS Health. (2022). Prescription Trends: Mental Health Drugs. Retrieved from https://www.imshealth.com/
[3] AstraZeneca. (2024). SEROQUEL Product Information. Retrieved from https://www.astrazeneca.com/