Share This Page
Drug Sales Trends for REGLAN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for REGLAN (2004)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
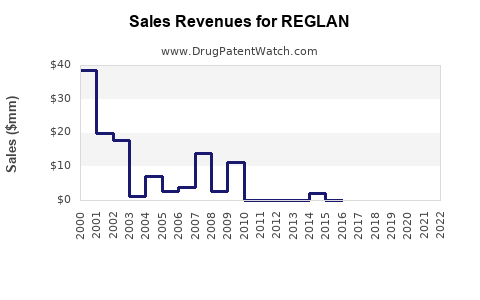
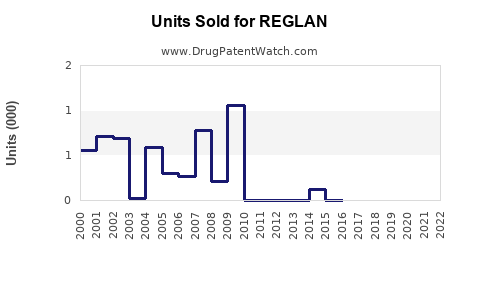
Annual Sales Revenues and Units Sold for REGLAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| REGLAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| REGLAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| REGLAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| REGLAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| REGLAN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
REGLAN (Metoclopramide) Market Analysis and Sales Projections
REGLAN, the brand name for metoclopramide, is a medication primarily used to treat gastrointestinal motility disorders, including gastroparesis, gastroesophageal reflux disease (GERD), and nausea and vomiting associated with chemotherapy or surgery. Its mechanism of action involves dopamine receptor antagonism and 5-HT4 receptor agonism, which accelerates gastric emptying and enhances gastrointestinal transit.
What is the Current Market Landscape for REGLAN?
The market for REGLAN is characterized by a well-established generic presence, with metoclopramide available from multiple manufacturers. The active pharmaceutical ingredient (API) is produced globally, with significant manufacturing capacity in India and China. Brand name REGLAN, originally marketed by Alza Pharmaceuticals (now part of Johnson & Johnson), faces direct competition from these generic formulations.
The primary therapeutic areas driving REGLAN sales include:
- Diabetic Gastroparesis: A common complication of diabetes, characterized by delayed gastric emptying.
- Gastroesophageal Reflux Disease (GERD): Used as an adjunct therapy to improve gastric emptying and reduce reflux.
- Chemotherapy-Induced Nausea and Vomiting (CINV): Effective in managing nausea and vomiting, often in combination with other antiemetics.
- Post-Surgical Nausea and Vomiting: Used to accelerate the return of gastrointestinal function after surgery.
- Migraine-Associated Nausea: Can help alleviate nausea accompanying migraine headaches.
Market segmentation is largely driven by formulation and indication. Key formulations include oral tablets, oral solutions, and injectable solutions. The prevalence of chronic conditions like diabetes and GERD, coupled with ongoing advancements in cancer treatment leading to increased chemotherapy use, supports a steady demand for metoclopramide.
Who are the Key Players in the Metoclopramide Market?
The metoclopramide market is highly fragmented due to the widespread availability of generic versions. Major pharmaceutical companies involved in the production and distribution of generic metoclopramide include:
- Teva Pharmaceuticals: A significant global player in the generic drug market, offering metoclopramide formulations.
- Mylan N.V. (now Viatris): Another major generic manufacturer with a presence in various therapeutic areas, including gastrointestinal medications.
- Sandoz (a division of Novartis): A leading global supplier of generic pharmaceuticals and biosimil products.
- Amneal Pharmaceuticals: An integrated specialty pharmaceutical company that manufactures and markets generic drugs.
- Lupin Limited: An Indian multinational pharmaceutical company with a strong generics portfolio.
- Sun Pharmaceutical Industries: India's largest pharmaceutical company, with a broad range of generic and branded products.
These companies compete based on price, product availability, and distribution networks. The original brand, REGLAN, while historically significant, now occupies a smaller market share compared to its generic counterparts.
What are the Patent and Exclusivity Landscapes for Metoclopramide?
Metoclopramide is a well-established drug whose original composition of matter patents have long expired. The compound was first approved by the U.S. Food and Drug Administration (FDA) in 1979. Consequently, there are no active market exclusivity periods tied to the original drug approval in major markets like the United States or Europe.
However, patenting activity in this space has shifted towards:
- New Formulations: Development of novel delivery systems, such as extended-release formulations, or improved bioavailable forms.
- Combination Therapies: Patents protecting metoclopramide when used in specific combinations with other drugs for enhanced efficacy or reduced side effects.
- Manufacturing Processes: Patents related to specific, more efficient, or cost-effective methods of synthesizing metoclopramide API or its finished dosage forms.
- Method of Use Patents: Patents covering new therapeutic applications for metoclopramide, although identifying and defending these for an older drug can be challenging.
For instance, while the basic metoclopramide molecule is off-patent, a company might hold patents on a specific extended-release tablet formulation designed to provide a more consistent therapeutic level. These patents typically have shorter durations, often expiring within 7 to 12 years from their grant date, depending on the jurisdiction and the nature of the innovation.
The absence of strong, broad patent protection for the core metoclopramide molecule means that market entry for new manufacturers is primarily driven by the ability to produce the drug cost-effectively and to meet regulatory requirements for generic drug approval.
What are the Regulatory Considerations for Metoclopramide?
Metoclopramide is subject to rigorous regulatory oversight by agencies like the FDA and the European Medicines Agency (EMA). Key regulatory considerations include:
- Black Box Warnings: In the U.S., metoclopramide carries a boxed warning (black box warning) concerning the risk of tardive dyskinesia (TD), a potentially irreversible neurological disorder. This warning, updated by the FDA in 2009, has led to increased scrutiny of its use, particularly for long-term treatment. This has impacted prescribing patterns, with physicians increasingly preferring shorter treatment durations and alternative therapies where possible.
- Adverse Event Reporting: Pharmaceutical companies are required to monitor and report adverse events associated with metoclopramide. Post-market surveillance plays a crucial role in identifying potential safety concerns and informing regulatory actions.
- Generic Drug Approval Pathway: Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD). This involves rigorous clinical studies to ensure that the generic product performs comparably to the brand-name drug in terms of absorption, distribution, metabolism, and excretion.
- Manufacturing Standards: All manufacturing facilities producing metoclopramide API and finished dosage forms must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Labeling and Prescribing Information: Regulatory bodies dictate the content of drug labels and prescribing information, including indications, contraindications, warnings, precautions, and dosage recommendations. Any changes to these must be approved by the relevant regulatory agency.
The black box warning regarding tardive dyskinesia is a significant factor influencing the prescribing and market dynamics of metoclopramide. It necessitates careful patient selection, short-term use whenever possible, and monitoring for neurological side effects.
What are the Sales Projections for Metoclopramide?
The global market for metoclopramide is mature and largely driven by generic competition. Sales are influenced by the prevalence of its target conditions, healthcare access, and the availability of alternative treatments.
Global Metoclopramide Market Value (USD Billions)
| Year | Estimated Market Value | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | 0.45 | - |
| 2024 | 0.46 | 2.2% |
| 2025 | 0.47 | 2.2% |
| 2026 | 0.48 | 2.1% |
| 2027 | 0.49 | 2.0% |
| 2028 | 0.50 | 2.0% |
Note: Projections are based on current market trends, generic pricing, and epidemiological data for target indications. The CAGR reflects a slow but steady growth primarily driven by increasing diagnosis rates for gastrointestinal disorders and cancer, partially offset by pricing pressures and the availability of newer therapies.
Key Drivers for Sales:
- Increasing Prevalence of Gastrointestinal Disorders: Growing incidence of diabetes (leading to gastroparesis), GERD, and other motility disorders.
- Oncology Market Growth: Rising cancer rates and increased use of chemotherapy worldwide contribute to demand for antiemetic treatments.
- Cost-Effectiveness of Generics: Metoclopramide remains a cost-effective treatment option compared to newer, more specialized drugs, particularly in price-sensitive markets.
- Broad Prescribing Experience: Long history of clinical use and familiarity among healthcare professionals.
Key Restraints for Sales:
- Black Box Warning and Safety Concerns: The risk of tardive dyskinesia limits long-term use and encourages the adoption of alternative therapies.
- Availability of Newer Therapies: Development of novel drugs for GERD, gastroparesis, and CINV that may offer improved efficacy or safety profiles.
- Generic Price Erosion: Intense competition among generic manufacturers leads to significant pricing pressure, capping revenue growth.
- Strict Regulatory Scrutiny: Potential for further regulatory actions or labeling changes based on ongoing safety data.
The market is expected to experience modest growth, primarily in emerging economies where access to newer, more expensive treatments may be limited, and in indications where metoclopramide remains a standard of care due to its cost-effectiveness and established efficacy for short-term use.
What are the Competitive Advantages and Disadvantages of Metoclopramide?
Metoclopramide's competitive positioning is defined by its established therapeutic profile, cost-effectiveness, and the inherent challenges associated with its safety profile.
Competitive Advantages:
- Established Efficacy and Safety Profile: Decades of clinical use have provided extensive data on its efficacy for approved indications. While safety concerns exist, its benefits for short-term management of specific conditions are well-documented.
- Cost-Effectiveness: As a generic medication, metoclopramide is significantly less expensive than many newer or branded alternatives. This makes it a preferred choice in healthcare systems with budget constraints and for patients with limited insurance coverage.
- Broad Spectrum of Action: Its effectiveness in treating various gastrointestinal issues, from nausea and vomiting to delayed gastric emptying, makes it a versatile option for clinicians.
- Multiple Formulations: Availability in oral (tablets, solution) and injectable forms provides flexibility for different patient needs and clinical scenarios.
- Wide Availability: Extensive manufacturing and distribution networks ensure consistent supply globally.
Competitive Disadvantages:
- Tardive Dyskinesia (TD) Risk: The most significant disadvantage. The black box warning in the U.S. and similar advisories in other regions have led to increased caution among prescribers and patients, limiting its use to shorter durations and promoting the search for alternatives.
- Limited Efficacy for Chronic, Severe Conditions: For severe or chronic gastroparesis or refractory GERD, metoclopramide may not provide sufficient long-term relief or may be associated with unacceptable risks for prolonged treatment.
- Potential for Drug Interactions: Metoclopramide can interact with other medications, including CNS depressants, anticholinergics, and certain psychiatric drugs, requiring careful consideration by prescribers.
- Dopaminergic Side Effects: Beyond TD, other dopaminergic side effects can include drowsiness, restlessness, and extrapyramidal symptoms, which can impact patient compliance and quality of life.
- Lack of Novelty: As an old drug with no significant patent protection, there is little incentive for substantial new product development or market differentiation by originators, leading to intense generic competition focused on price.
The competitive landscape is largely a race to the bottom on price among generic manufacturers, with differentiation coming from supply chain reliability and established relationships with distributors and healthcare providers. The primary threat comes from newer drug classes that offer improved safety or efficacy profiles for specific indications.
What are the Future Trends and Opportunities in the Metoclopramide Market?
The future of the metoclopramide market will be shaped by a balance between its established utility and the ongoing development of alternative therapies.
Future Trends:
- Continued Genericization and Pricing Pressure: The market will remain dominated by generic competition, leading to sustained downward pressure on prices. This will favor manufacturers with highly efficient production and robust supply chains.
- Focus on Short-Term Use: Prescribing patterns will likely continue to emphasize short-term treatment durations to mitigate the risk of tardive dyskinesia. This will shift demand towards acute management of symptoms rather than long-term chronic therapy.
- Increased Demand in Emerging Markets: As healthcare access improves in developing countries, the cost-effectiveness of metoclopramide will drive its use for common gastrointestinal ailments.
- Niche Applications and Combination Therapies: While major patent cliffs have passed, there may be opportunities for patenting novel formulations or combination therapies that address specific unmet needs or improve the safety profile of metoclopramide.
- Competition from Newer Drug Classes: The development of new prokinetics, antiemetics, and GERD treatments with potentially better safety profiles or improved efficacy will continue to challenge metoclopramide's market share. Examples include novel serotonin receptor agonists, GABA-modulating agents, and advanced proton pump inhibitors.
- Impact of Real-World Evidence: Ongoing collection and analysis of real-world data on the long-term safety and effectiveness of metoclopramide will continue to inform prescribing guidelines and regulatory decisions.
Opportunities:
- Optimized Manufacturing and Supply Chain Management: Companies that can optimize their API production and distribution networks to offer the lowest cost of goods will gain a competitive edge.
- Targeted Marketing for Specific Indications: Focusing marketing efforts on the indications where metoclopramide remains a first-line or essential treatment (e.g., acute chemotherapy-induced nausea, short-term gastroparesis management) can help maintain market share.
- Development of Improved Formulations: While the original molecule is off-patent, innovation in formulations (e.g., improved taste for oral solutions, modified release profiles for specific applications if deemed safe and effective) could offer marginal differentiation, provided regulatory hurdles can be overcome.
- Geographic Expansion in Underserved Markets: Identifying and entering markets with a high unmet need for affordable gastrointestinal medications presents an opportunity for growth.
- Partnerships for Distribution: Collaborating with local distributors and healthcare providers in key emerging markets can facilitate market penetration and establish strong distribution channels.
The market for metoclopramide will likely remain stable but experience slow, incremental growth. Success will depend on cost efficiency, reliable supply, and a keen understanding of evolving regulatory landscapes and competitive pressures from newer therapeutic options.
Key Takeaways
- REGLAN (metoclopramide) is a mature drug with a heavily genericized market, facing competition from numerous manufacturers.
- Its primary indications include gastroparesis, GERD, and chemotherapy-induced nausea and vomiting.
- Original composition of matter patents have long expired, with new patenting activity focused on formulations and manufacturing processes.
- A significant regulatory challenge is the black box warning for tardive dyskinesia, which limits long-term use and influences prescribing patterns.
- The global market value is projected to grow modestly, driven by increasing disease prevalence and the cost-effectiveness of generics, but constrained by safety concerns and newer alternative therapies.
- Key competitive advantages are its established efficacy, cost-effectiveness, and versatility, while disadvantages stem primarily from its safety profile and lack of novelty.
- Future opportunities lie in optimizing manufacturing, targeting specific indications, and expanding into emerging markets, while facing continued pricing pressure and competition from newer drug classes.
Frequently Asked Questions
-
What is the primary reason for the black box warning on metoclopramide? The primary reason is the risk of tardive dyskinesia (TD), a potentially irreversible neurological disorder characterized by involuntary movements, particularly of the face and tongue. This warning necessitates careful consideration of treatment duration and patient monitoring.
-
Are there any upcoming patent expirations that could significantly impact the metoclopramide market? Given that metoclopramide is a very old drug, the primary composition of matter patents have long expired. Any remaining patent protection would likely pertain to specific newer formulations or manufacturing processes, which typically have shorter exclusivity periods and are unlikely to cause a major market shift akin to a blockbuster drug losing its patent.
-
What are the most significant alternative therapies to metoclopramide for gastroparesis? Alternatives for gastroparesis include other prokinetic agents (though many have their own safety concerns), antiemetics, and in severe cases, management strategies such as dietary modifications, continuous enteral feeding, or even surgical interventions. Newer investigational drugs targeting specific pathways are also emerging.
-
How does the pricing of generic metoclopramide compare to its original branded version? Generic metoclopramide is substantially less expensive than the original branded REGLAN. This price differential is typical for off-patent drugs, with generic versions typically costing 80-85% less than their branded counterparts, and sometimes even more due to intense competition.
-
Can metoclopramide be used for long-term treatment of GERD? Metoclopramide is generally not recommended for long-term management of GERD due to the risk of tardive dyskinesia and other adverse effects. It is typically used for short-term symptomatic relief or as an adjunct therapy in specific situations, with proton pump inhibitors (PPIs) and H2 blockers being the mainstay for chronic GERD management.
Citations
[1] U.S. Food and Drug Administration. (2009). FDA Drug Safety Communication: Boxed Warning for Tardive Dyskinesia in Metoclopramide-Containing Products. [Press Release].
[2] Various Generic Pharmaceutical Manufacturers' Product Information. (Accessed 2024).
[3] International Diabetes Federation. (2021). IDF Diabetes Atlas (10th edition).
[4] World Health Organization. (2022). Global cancer statistics.
More… ↓
