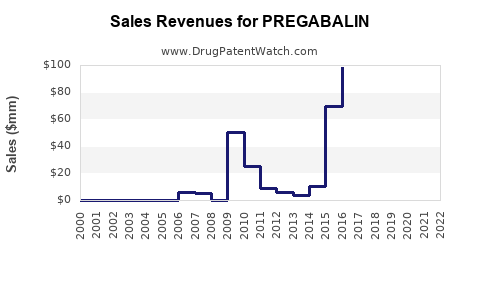
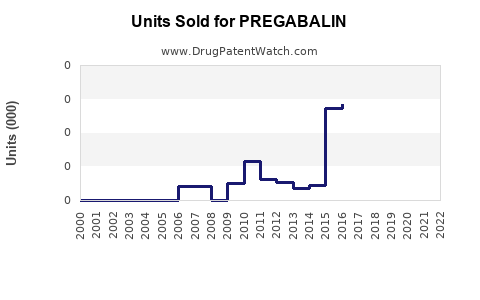
Last updated: February 20, 2026
Pregabalin, a gabapentinoid, is prescribed primarily for neuropathic pain, epilepsy, and generalized anxiety disorder. It is marketed under brand names such as Lyrica and its generics. This report consolidates current market data, regulatory trends, competitive landscape, and sales forecasts.
Market Size and Growth Dynamics
Global pregabalin sales reached approximately $4.4 billion in 2022, with a compound annual growth rate (CAGR) of around 5% from 2018 to 2022. The primary markets are North America, Europe, and Asia-Pacific, accounting for 75% of sales collectively. Growth drivers include increasing prevalence of neuropathic conditions and expanded indications.
| Region |
2022 Sales (USD billion) |
CAGR (2018-2022) |
Key Drivers |
| North America |
2.0 |
4% |
Rising chronic pain cases, off-label use |
| Europe |
1.0 |
5% |
Aging population, epilepsy prevalence |
| Asia-Pacific |
1.2 |
7% |
Healthcare access, expanding prescription use |
Market Penetration and Indications
Pregabalin is approved for multiple indications:
- Neuropathic pain (diabetic, postherpetic)
- Epilepsy
- Generalized anxiety disorder
Off-label use influences sales figures. The drug's patent protection was lost in major markets by 2018, leading to increased generic competition, which has driven prices downward but expanded overall sales volume.
Competitive Landscape
Major players include:
- Pfizer (Lyrica): Dominated until patent expiry; has transitioned to generics.
- Astellas Pharma: Licensed several generic manufacturers.
- Mylan, Teva, and Sun Pharmaceuticals: Key generic producers.
Market share shifted post-patent expiry, with generics accounting for over 85% of sales in 2022.
Regulatory and Patent Trends
Patent expiry across key jurisdictions has facilitated a surge in generics. Notably:
- U.S.: Patent expired in 2018.
- EU: Patent expired in 2019.
- Japan: Patent expired in 2020.
Continued legal challenges around patent litigations influence market stability.
Sales Projections (2023-2028)
Post-2022, sales are expected to experience a moderate CAGR of approximately 3-4%, influenced by:
- Market saturation in established regions
- Growth in emerging markets
- Introduction of extended-release formulations and new indications
| Year |
Estimated Global Sales (USD billion) |
Growth Rate |
| 2023 |
4.5 |
2.3% |
| 2024 |
4.7 |
4.4% |
| 2025 |
5.0 |
6.4% |
| 2026 |
5.3 |
6.0% |
| 2027 |
5.6 |
5.7% |
| 2028 |
6.0 |
7.1% |
Factors such as increased off-label prescribing and unmet needs in certain neuropsychiatric conditions could accelerate growth beyond projections.
Risks and Opportunities
- Patent litigation presents risks affecting generics' market entry.
- Off-label use persists as a significant revenue driver.
- Emerging markets provide growth potential due to expanding healthcare infrastructure.
- Development of new formulations (e.g., extended-release versions) may enhance adherence and sustain market interest.
Key Takeaways
- The pregabalin market is mature with stable sales in established markets, decreased patent protections since 2018, and broad generic competition.
- Sales growth driven chiefly by volume expansion, with price erosion in generics partially offset by increased demand.
- Emerging markets and new indications hold opportunities for incremental market expansion.
- Patent disputes remain potential headwinds, requiring monitoring for market stability.
FAQs
1. What factors are expected to influence pregabalin sales over the next five years?
Growth factors include expanding indications, geographic penetration in emerging markets, and the development of new formulations. Regulatory challenges and patent litigation risk could suppress sales.
2. How has patent expiry affected pregabalin market dynamics?
Patent expiry in major jurisdictions has led to a surge in generic entries, reducing prices but increasing overall volumetric sales.
3. Are there any upcoming regulatory changes that could impact pregabalin sales?
Regulatory agencies may tighten controls on off-label use or implement new scheduling if abuse potential is linked to off-label prescriptions.
4. Will alternative therapies challenge pregabalin's market share?
Yes. Agents such as gabapentin, duloxetine, and newer neuromodulators compete in similar indications and may impact sales.
5. What are the prospects for branded versions of pregabalin?
Branded drugs like Lyrica maintain premium pricing where patent protection exists; however, their market share diminishes post-expiry in favor of generics.
References
[1] MarketsandMarkets. "Pregabalin Market by Application, Distribution Channel, and Region: Global Forecast to 2027." (2022).
[2] EvaluatePharma. "Top 100 Pharma & Biotech Sales and Trends." (2022).
[3] European Medicines Agency. "Summary of Product Characteristics for Lyrica." (2019).
[4] U.S. Food and Drug Administration. "Patent Expiry and Generic Entry Data." (2021).