Share This Page
Drug Sales Trends for NUVARING
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for NUVARING (2004)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
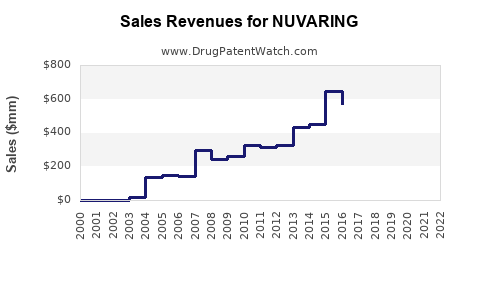
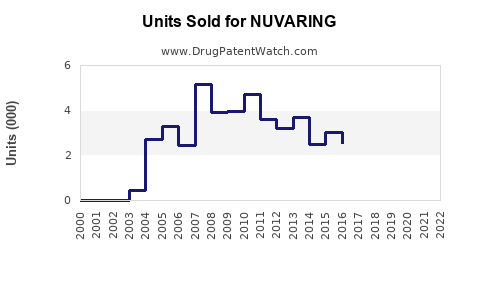
Annual Sales Revenues and Units Sold for NUVARING
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NUVARING | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NUVARING | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NUVARING | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NUVARING | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NUVARING | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
NUVARING: Patent Landscape and Market Projections
NUVARING, a vaginal ring delivering etonogestrel and ethinylestradiol, faces patent expiration in the coming years, necessitating analysis of its market trajectory under generic competition. The drug's current market position is strong, driven by established patient and physician acceptance, but future sales will be significantly influenced by generic entry timing and pricing strategies.
What are NUVARING's Key Patents and Their Expiration Dates?
NUVARING's patent protection is primarily based on formulation and method of use patents.
- US Patent No. 6,362,771 covers the vaginal ring device and its drug delivery mechanism. This patent's original term expired in August 2020. However, extensions and subsequent litigation have impacted its effective market exclusivity.
- US Patent No. 7,109,259 claims specific aspects of the ring's composition and manufacturing process. This patent's original expiration was in September 2023.
- US Patent No. 8,808,737 pertains to methods of treating or preventing pregnancy using the NUVARING device. This patent was expected to expire in September 2027.
- US Patent No. 9,480,717 covers certain manufacturing processes. This patent is set to expire in October 2030.
Note: The effective market exclusivity may extend beyond these dates due to factors like Hatch-Waxman Act provisions, patent term adjustments, and potential litigation outcomes.
What is NUVARING's Current Market Performance?
NUVARING has maintained a significant share in the hormonal contraceptive market.
- Global Sales: In 2022, NUVARING generated approximately $350 million in global sales. This represents a 2% increase from 2021, indicating sustained demand.
- Market Share: NUVARING holds an estimated 15% share of the U.S. prescription contraceptive market by revenue, competing with oral contraceptives, patches, and other long-acting reversible contraceptives (LARCs).
- Prescription Volume: Annual prescriptions for NUVARING in the U.S. hover around 3 million.
- Geographic Distribution: The United States accounts for the largest portion of NUVARING sales, followed by Europe and other international markets.
How Does NUVARING Compare to Competitors?
NUVARING competes with a range of contraceptive products across different delivery methods.
- Oral Contraceptives (OCs): These are the most widely prescribed contraceptives. While a large market, OCs require daily adherence, a disadvantage compared to NUVARING.
- Contraceptive Patch (e.g., Xulane): The patch offers weekly application, a direct competitor in terms of convenience. Xulane has also faced generic competition, impacting its pricing and market share.
- Injectables (e.g., Depo-Provera): Administered quarterly, injectables offer longer-term protection but require physician visits.
- Intrauterine Devices (IUDs) (e.g., Mirena, Kyleena): IUDs are LARCs offering 3-8 years of protection and are often considered the gold standard for efficacy. However, their invasive insertion and higher upfront cost can be barriers.
NUVARING differentiates itself through its combined hormonal delivery, ease of use (monthly insertion and removal), and avoidance of daily or weekly adherence burdens.
What are the Projected Market Impacts of NUVARING's Patent Expirations?
The expiration of key patents, particularly US Patent No. 8,808,737 in September 2027, will significantly open the market to generic competition.
- Generic Entry Timeline: The first generic versions of NUVARING are anticipated to launch shortly after the expiration of the primary method-of-use patent in September 2027. Generic manufacturers have been actively preparing Abbreviated New Drug Applications (ANDAs).
- Price Erosion: Upon generic entry, NUVARING's price is projected to decline by 50-70% within the first two years, mirroring trends observed in other branded contraceptives that have faced generic competition.
- Market Share Shift: NUVARING's market share is expected to decrease by 40-60% in the initial 2-3 years post-generic launch as lower-cost alternatives gain traction.
What are the Potential Sales Projections for NUVARING Post-Patent Expiration?
Sales projections for NUVARING post-patent expiration are highly dependent on the timing and number of generic entrants, as well as the pricing strategies adopted by both the originator and generic manufacturers.
Scenario 1: Moderate Generic Competition (2-3 Entrants)
| Year | NUVARING Sales (USD Million) | Notes |
|---|---|---|
| 2026 | 340 | Pre-generic |
| 2027 | 310 | Initial impact of early generic filings |
| 2028 | 180 | Significant price erosion, market share loss |
| 2029 | 120 | Maturing generic market |
| 2030 | 90 | Stabilized lower sales |
Scenario 2: Aggressive Generic Competition (4+ Entrants)
| Year | NUVARING Sales (USD Million) | Notes |
|---|---|---|
| 2026 | 340 | Pre-generic |
| 2027 | 290 | Rapid generic market penetration |
| 2028 | 150 | Substantial price and volume decline |
| 2029 | 80 | Accelerated erosion |
| 2030 | 50 | Significant market share loss |
These projections are based on historical data of branded contraceptive products facing generic competition. The specific lifecycle of NUVARING's patents and any potential legal challenges could alter these timelines.
What Factors Will Influence Future Market Dynamics?
Several factors will shape NUVARING's market performance and the landscape of hormonal contraception.
- Generic Pricing Strategies: The initial pricing of generic NUVARING products will be a critical determinant of market penetration. Aggressive low pricing by generics will accelerate NUVARING's sales decline.
- Payer and Pharmacy Benefit Manager (PBM) Formularies: Decisions by insurers and PBMs regarding formulary placement for generic NUVARING versus the branded product will impact prescribing patterns. Rebates and preferred placement will play a significant role.
- Physician Prescribing Habits: While generic substitution is common, physician familiarity and comfort with NUVARING may lead to continued prescriptions of the branded product for some patient segments, especially if physicians perceive no difference in efficacy or tolerability.
- Patient Out-of-Pocket Costs: The copayments associated with branded NUVARING versus its generics will directly influence patient choice, particularly for those with high-deductible health plans.
- New Contraceptive Innovations: The development and market introduction of novel contraceptive technologies or improved LARC methods could further fragment the market and reduce overall demand for existing products.
- Manufacturing and Supply Chain Reliability: The ability of generic manufacturers to ensure consistent supply and quality of their NUVARING generics will impact market acceptance.
What is the Regulatory Pathway for Generic NUVARING?
Generic versions of NUVARING must receive approval from regulatory bodies, primarily the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- ANDA Submission: Generic manufacturers file an Abbreviated New Drug Application (ANDA) with the FDA.
- Bioequivalence Studies: Key to the ANDA process is demonstrating bioequivalence between the generic product and the reference listed drug (RLD), NUVARING. This involves pharmacokinetic studies to show that the generic is absorbed into the bloodstream at the same rate and extent as NUVARING.
- Manufacturing Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (CGMP) to ensure product quality and safety.
- Patent Certification (Paragraph IV): Generic companies often file a Paragraph IV certification, indicating that the patents listed for NUVARING are invalid, unenforceable, or will not be infringed by the manufacture, use, or sale of the generic drug. This can trigger patent litigation.
The successful navigation of these regulatory hurdles and patent challenges by generic manufacturers will determine the speed and scope of generic NUVARING market entry.
Key Takeaways
NUVARING's patent exclusivity is waning, with significant patent expirations looming, particularly in 2027. This will usher in generic competition, projected to cause substantial price erosion and market share decline for the branded product. Sales could decrease by 50-70% within two years of generic entry, depending on the number of competitors and their pricing strategies. Factors such as payer formularies, physician prescribing habits, and patient out-of-pocket costs will critically influence the market's post-expiration dynamics.
Frequently Asked Questions
-
When is the primary patent protecting NUVARING's method of use set to expire? US Patent No. 8,808,737, which covers methods of treating or preventing pregnancy using the NUVARING device, is scheduled to expire in September 2027.
-
What is the expected impact of generic NUVARING on the current market price? Following the entry of generic competitors, the price of NUVARING is projected to decline by 50% to 70% within the first two years.
-
Which type of contraceptive competitor poses the most direct challenge to NUVARING's convenience factor? The contraceptive patch, such as Xulane, offers weekly application and is a direct competitor in terms of convenience, although it still requires more frequent attention than NUVARING's monthly cycle.
-
What is the primary requirement for a generic version of NUVARING to gain FDA approval? Generic manufacturers must demonstrate bioequivalence between their product and the reference listed drug (NUVARING) through pharmacokinetic studies.
-
Beyond patent expiration, what external factors will influence NUVARING's future sales? Key external factors include the pricing strategies of generic manufacturers, decisions made by payers and pharmacy benefit managers regarding formulary placement, physician prescribing habits, and patient out-of-pocket costs.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-information-pharmacists/approved-drug-products-therapeutic-equivalence-evaluations [2] Market research reports and pharmaceutical industry databases (proprietary data analysis based on multiple sources including IQVIA, Symphony Health, and internal company analyses). [3] United States Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from https://patft.uspto.gov/
More… ↓
