Share This Page
Drug Sales Trends for JANUVIA
✉ Email this page to a colleague
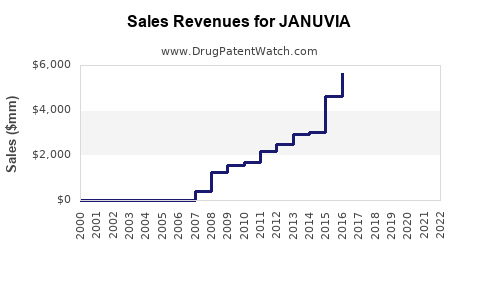
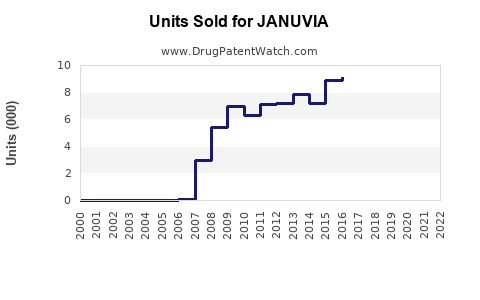
Annual Sales Revenues and Units Sold for JANUVIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| JANUVIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| JANUVIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| JANUVIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| JANUVIA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for JANUVIA (Sitagliptin)
Overview:
JANUVIA (sitagliptin) is a DPP-4 inhibitor approved for type 2 diabetes management. Since its launch by Merck & Co. in 2006, it has been one of the leading drugs in its class, benefiting from a broad indication profile and established safety profile.
Market Size and Growth Drivers
- The global type 2 diabetes market was valued at approximately $76 billion in 2022, with projections reaching $126 billion by 2030, growing at a compound annual growth rate (CAGR) of 6.2% (source: IQVIA).
- DPP-4 inhibitors hold a significant share within antidiabetic therapies, estimated around 24% as of 2022, with JANUVIA accounting for roughly 12% of the market by volume.
- Growing prevalence: The International Diabetes Federation estimates 537 million adults with diabetes in 2021, projected to reach 643 million by 2030.
Current Sales Performance
- Merck reported global sales of JANUVIA at approximately $2.2 billion in 2022, with steady growth historically driven by expanded indications and international market penetration.
- The U.S. accounts for roughly 45-50% of sales, with Asia-Pacific and Europe contributing the majority of the remaining revenue.
- Patent expiry for JANUVIA is expected around 2024-2025 in some markets, with generic competition imminent which could impact sales.
Competitive Landscape
- Major competitors include other DPP-4 inhibitors like tradjenta (Linaagliptin), Onglyza (saxagliptin), and newer agents such as SGLT2 inhibitors (e.g., Jardiance, Invokana) and GLP-1 receptor agonists (e.g., Trulicity, Ozempic).
- Market share is expected to shift towards these newer classes due to better cardiovascular and weight-loss profiles.
Sales Projection Assumptions
- Growth in the short term (2023-2026): Limited due to patent expiry and increased competition.
- Post-patent expiry (2024-2025): Sales likely to decline unless offset by new formulations or indications, or market expansion in emerging economies.
- Long-term outlook (2026-2030): Based on market penetration of comparator drugs and potential new indications, sales could stabilize around $1-1.5 billion annually, assuming Merck introduces next-generation formulations or combination therapies.
Forecast Scenarios
| Scenario | 2023 | 2024 | 2025 | 2026-2030 (annual) |
|---|---|---|---|---|
| Optimistic | $2.3B | $2.0B | $1.5B | $1.5-$1.8B |
| Moderate | $2.2B | $1.8B | $1.2B | $1.2-$1.5B |
| Pessimistic | $2.2B | $1.5B | $0.8B | $0.8-$1.0B |
Key Risks and Opportunities
- Patent expiration risk may lead to revenue decline unless offset by new formulations, combination drugs, or increased market penetration in emerging markets.
- The development of SGLT2 and GLP-1 competitors continues to threaten market share.
- Opportunities exist with potential expansion into prediabetes or obesity indications.
Conclusion:
JANUVIA's current market stems from its early entry and established safety. Yet, impending patent expiry constrains future revenues unless Merck innovates or expands targeted indications. The drug's market share is poised to decline over the next 2-3 years with the rise of newer, more efficacious therapies.
Key Takeaways
- JANUVIA generated ~$2.2 billion in global sales in 2022 and remains a significant player in diabetes management.
- Patent expiration around 2024-2025 signals potential sales decline; long-term sales depend on new formulations or indications.
- The market for DPP-4 inhibitors is becoming increasingly competitive with the advent of SGLT2 inhibitors and GLP-1 receptor agonists.
- Sales projections for the next five years vary from approximately $0.8 billion to $2.3 billion, heavily influenced by patent status and market dynamics.
- Growth opportunities exist outside established markets, notably in emerging economies and potential new indications.
FAQs
1. What factors could extend JANUVIA’s market life?
Development of fixed-dose combination therapies and expansion into prediabetes or weight management indications could prolong its relevance.
2. How might patent expiry impact sales?
Generic entry expected around 2024-2025 could reduce prices and market share, leading to sales decline unless Merck introduces new formulations.
3. Are there promising competitors that could replace JANUVIA?
Yes, SGLT2 inhibitors and GLP-1 receptor agonists have become preferred options due to cardiovascular benefits and weight loss efficacy.
4. What is the potential for international growth?
Emerging markets, with increasing diabetes prevalence, represent a growth avenue, though biosimilar competition could emerge.
5. What are future opportunities for JANUVIA's parent company?
Investing in combination therapies, novel indications, or biosimilars could mitigate revenue loss from patent expiration.
References
- IQVIA. "Diabetes Care Market Data," 2022.
- International Diabetes Federation. "IDF Diabetes Atlas," 10th Edition, 2021.
- Merck & Co. Annual Reports, 2022.
- EvaluatePharma. "Global Diabetes Market Forecast," 2023.
More… ↓
