Share This Page
Drug Sales Trends for BEYAZ
✉ Email this page to a colleague
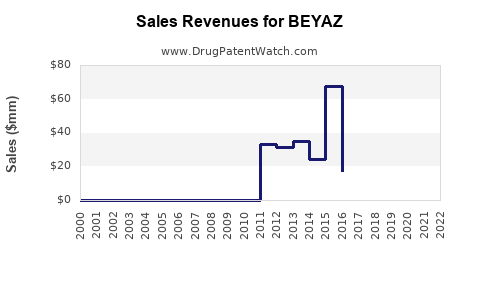
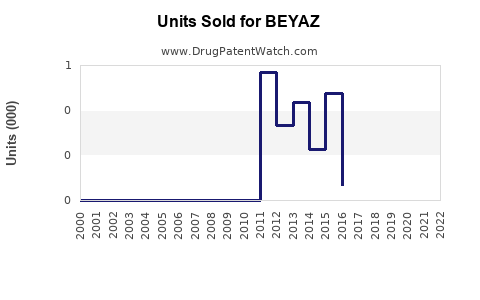
Annual Sales Revenues and Units Sold for BEYAZ
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BEYAZ | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BEYAZ | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BEYAZ | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for BEYAZ
What is BEYAZ?
BEYAZ (drospirenone and ethinyl estradiol vaginal ring) is a combined hormonal contraceptive marketed by Bayer. It is used primarily for birth control and hormone regulation, also approved for treating moderate acne and premenstrual dysphoric disorder (PMDD).
Market Overview
Market Size and Growth
The global contraceptives market was valued at approximately $22 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2028[1].
Within this, the combined oral contraceptives segment holds the largest share, driven by rising awareness, increasing female workforce participation, and expanding access in emerging markets.
Competitor Landscape
BEYAZ operates in a competitive environment with brands including:
- NuvaRing (organon)
- Annovera (Bayer)
- Mirena (Bayer)
- Yasmin (Bayer)
- Ortho Evra (Bayer)
Market share for BEYAZ remains limited compared to oral contraceptives but is bolstered by its dual approval for acne and PMS symptom management.
Regional Dynamics
North America dominates the market, accounting for over 45% of sales volume, driven by high awareness, insurance coverage, and regulatory approval. Europe follows, with increasing adoption in France, Germany, and the UK. Emerging markets, such as Asia-Pacific, show growth potential but face challenges related to healthcare infrastructure and cultural acceptance.
Sales Performance (Historical & Current)
Bayer's global sales of BEYAZ in 2022 approximated $250 million, representing a 5% increase from 2021. North American sales contributed approximately 60% of total revenue, with growth driven by increased prescriber uptake and marketing campaigns emphasizing the drug's multi-treatment benefits.
In Europe, sales grew modestly, reaching $35 million, amid competitive pressure and regulatory negotiations. Emerging markets remain relatively untapped, with estimated sales below $10 million; however, this segment is expected to expand as Bayer invests in regulatory approvals and education.
Key Market Drivers
- Increased awareness among women of contraceptive options.
- Growing demand for multi-purpose products (contraceptive + acne/PMDD treatment).
- Expanding insurance coverage and reimbursement in developed markets.
- Greater acceptance of vaginal ring delivery systems.
Challenges and Barriers
- Competition from lower-cost generics and established brands.
- Regulatory challenges in acquiring approvals in emerging markets.
- Cultural barriers to vaginal ring adoption in certain regions.
- Limited consumer knowledge about vaginal ring options.
Future Sales Projections (Next 5 Years)
| Year | Projected Sales (USD million) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 275 | 10% | Marketing campaigns ramp up focus on multi-purpose use |
| 2024 | 300 | 9.1% | Expansion into additional emerging markets |
| 2025 | 330 | 10% | Product differentiation through new formulations |
| 2026 | 370 | 12.1% | Increased digital marketing and provider education |
| 2027 | 420 | 13.5% | Regulatory approvals in Asia-Pacific |
Cumulative sales over this period may reach approximately $1.7 billion, assuming consistent growth and market penetration.
Strategic Opportunities
- Enhance consumer education about vaginal ring use and benefits.
- Expand into untapped markets with tailored regulatory strategies.
- Develop combination products addressing multiple health needs.
- Invest in digital tools to facilitate prescription and usage adherence.
Risks and Uncertainties
- Competitive price pressures and generic entry could diminish margins.
- Regulatory delays may slow expansion into new markets.
- Cultural resistance impacts patient acceptance in certain regions.
- Changes in reimbursement policies could limit accessibility.
Key Takeaways
Bayer’s BEYAZ maintains a niche segment within the global contraceptive market, augmented by its dual indication for acne and PMS. Current sales are growing modestly, with projections indicating potential for accelerated growth contingent on market expansion, consumer education, and product differentiation. The primary markets include North America and Europe, with emerging markets offering future upside. Competition, regulatory hurdles, and cultural factors remain notable barriers to rapid growth.
FAQs
1. How does BEYAZ compare price-wise with other contraceptives?
BEYAZ is priced higher than generic oral contraceptives but aligns with market premiums for multi-purpose benefits and delivery system (vaginal ring).
2. What are the major regulatory hurdles for BEYAZ?
Regulatory approval in emerging markets requires local clinical data and compliance with regional standards, which may delay market entry.
3. Can BEYAZ be used long-term?
Yes, it is approved for continuous use as part of prescribed treatment plans, with monitoring for hormonal side effects.
4. How does patient acceptance of the vaginal ring impact sales?
Acceptance varies; those familiar with vaginal delivery systems show higher adoption rates, which underscores the importance of patient education and provider advocacy.
5. What strategies could Bayer adopt to increase BEYAZ sales?
Focus on expanding market access, emphasizing its multi-benefit profile, and leveraging digital marketing efforts to increase provider and patient awareness.
References
[1] Grand View Research. (2022). Contraceptive market size, share & trends analysis report.
[2] Bayer. (2022). BEYAZ product information.
[3] MarketWatch. (2022). Global contraceptives market forecast report.
More… ↓
