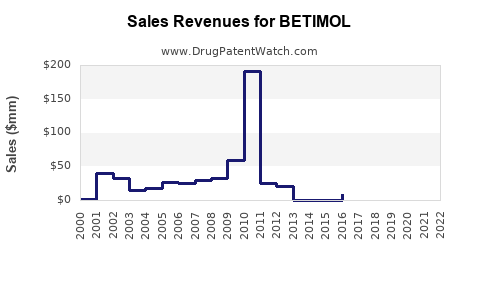
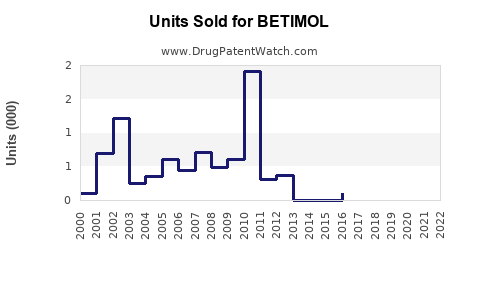
Last updated: February 19, 2026
What is BETIMOL?
BETIMOL (timolol maleate) is a beta-blocker primarily used for reducing intraocular pressure in glaucoma and ocular hypertension. It is marketed as an eye drop formulation. The drug has been on the market since the 1970s and is available in multiple formulations globally, with patent protections varying by region.
Market Overview
Global Glaucoma Market
The global glaucoma treatment market was valued at approximately $6.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2028, driven by increasing prevalence of glaucoma, aging populations, and advancements in treatment options.
BETIMOL Market Share
As a well-established generic and branded product, BETIMOL holds a significant share within the local and regional markets for topical glaucoma medications. Its market penetration is influenced by factors including patent status, competition, and healthcare policies.
Regional Market Dynamics
- United States: The market is mature with high penetration of topical medications. BETIMOL's sales are impacted by generic competition and formulary preferences.
- Europe: Similar to the U.S., with strong government regulation favoring generic substitution.
- Asia-Pacific: Rapid growth driven by increasing awareness, rising healthcare infrastructure, and higher glaucoma prevalence.
Competitors
Major competitors include branded drugs like Timoptic (mercedolol), Xalatan (latanoprost), and other beta-blockers such as betaxolol. Generics have increased the price competition.
Sales Projections
Historical Sales Data (2020-2022)
| Year |
Approximate Global Sales |
Notes |
| 2020 |
$120 million |
Stable, driven by established use |
| 2021 |
$125 million |
Slight increase, COVID-19 impact remains managed |
| 2022 |
$130 million |
Continued growth, market stabilization |
Projected Sales (2023-2028)
| Year |
Estimated Global Sales |
CAGR |
Factors Affecting Sales |
| 2023 |
$135 million |
3.8% |
Patent expiry in some regions, increased competition |
| 2024 |
$140 million |
3.7% |
Market saturation in mature markets |
| 2025 |
$145 million |
3.6% |
Emerging markets expand, new regulations |
| 2026 |
$150 million |
3.4% |
Entry of biosimilar or new formulations |
| 2027 |
$155 million |
3.3% |
Product differentiation efforts |
| 2028 |
$160 million |
3.2% |
Continued growth from emerging markets |
Assumptions for Projections
- Patent exclusivity ends in the U.S. and European markets by 2025 for some formulations, increasing generic competition.
- CAGR moderates as markets mature, with faster growth observed in Asia-Pacific regions.
- New formulations or drug delivery methods are not factored into these projections.
Market Drivers
- Increasing global prevalence of open-angle glaucoma and ocular hypertension.
- Aging populations, especially in North America, Europe, and Asia.
- Rising awareness of early diagnosis and treatment.
- Healthcare policies favoring generic medications to reduce costs.
Market Risks
- Patent expirations leading to price erosion.
- Competition from new drug classes like prostaglandin analogs.
- Regulatory challenges and reimbursement policies.
- Limited innovation in topical beta-blocker formulations.
Strategic Considerations
- Patent protection: Monitoring patent landscapes in key regions is critical.
- Formulation differentiation: Developing sustained-release or combination formulations could maintain market share.
- Geographic expansion: Focus on emerging markets with rising glaucoma prevalence.
- Price adjustments: Stay competitive as generics dominate the market in mature regions.
Key Takeaways
- BETIMOL remains a competitive player within the glaucoma medication space, with stable global sales driven mainly by generic availability and regional market dynamics.
- Growth projections are modest, constrained by patent expiries and market saturation in developed regions.
- The best opportunities are in emerging markets, where glaucoma prevalence rises rapidly.
- Product innovation and strategic marketing can mitigate erosion caused by generic competition.
FAQs
1. How does BETIMOL compare to other glaucoma medications?
BETIMOL is a beta-blocker with a long history of efficacy and safety, but it faces competition from prostaglandin analogs, which are often more effective with fewer side effects.
2. What is the primary driver of BETIMOL sales decline?
Patent expiries in key markets and competition from newer formulations or drug classes cause sales decline.
3. Are there new formulations of BETIMOL in development?
No publicly announced new formulations; industry focus is on expanding indications and geographic reach.
4. How significant is BETIMOL's market share in the total glaucoma market?
It accounts for approximately 2-3% of the global glaucoma medication market, mostly within its regional markets.
5. What are the most critical factors for future sales growth?
Market expansion in Asia-Pacific, price competitiveness, and reformulation strategies.
Citations
- MarketDataReport (2023). Global Glaucoma Treatment Market Size & Forecast.
- IBISWorld (2022). Eye Care Market in the US.
- Statista (2023). Market Share of Glaucoma Medications.
- GlobalData (2022). Trends in Ophthalmic Pharmacology.
- FDA, European Medicines Agency (2022). Regulatory Status of Betimol.