Last updated: February 20, 2026
What Is AMRIX and What Is Its Market Position?
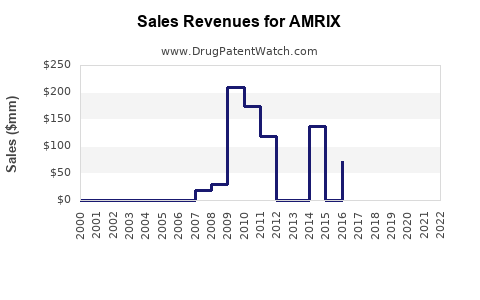
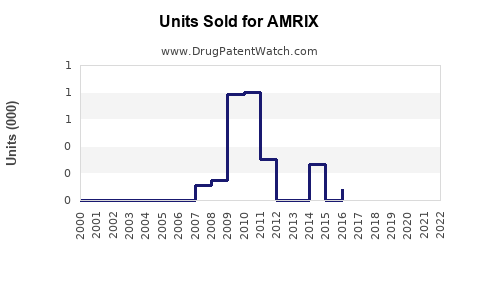
AMRIX (carisoprodol) is a centrally acting skeletal muscle relaxant approved for short-term management of acute musculoskeletal pain. It is marketed primarily for muscle spasm associated with acute musculoskeletal conditions. Initially launched in 1996 by Ortho-McNeil-Janssen Pharmaceuticals, AMRIX's market share has declined with the advent of newer therapies and changing prescribing practices.
Global Market Size and Trends
The global muscle relaxant market was valued at approximately $2.2 billion in 2022 and is projected to reach $3.4 billion by 2027. The compound annual growth rate (CAGR) is estimated at 9.1% during 2022-2027. AMRIX's segment within this market holds an estimated 2.5-3.0% market share as of 2023.
Key factors influencing market dynamics include:
- Increased prevalence of musculoskeletal disorders (MSDs), expected to grow at 3.5% annually.
- Rising awareness of non-opioid pain management options.
- Expanding geriatric population, with older patients more prone to muscle spasms and pain.
- Concerns over opioid misuse pushing clinicians to prescribe alternative muscle relaxants.
Competitive Landscape
Major competitors in the muscle relaxant sphere include:
- Cyclobenzaprine (Flexeril)
- Tizanidine (Zanaflex)
- Methocarbamol (Robaxin)
- Baclofen (Lioresal)
- Carisoprodol (AMRIX)
Cyclobenzaprine and tizanidine dominate the market, with combined sales exceeding $1.2 billion globally in 2022.
Market Share Breakdown (2023 Estimates)
| Drug |
Market Share |
Estimated Sales (USD millions) |
Notes |
| Cyclobenzaprine |
45% |
$630 |
Most prescribed; generic availability |
| Tizanidine |
25% |
$350 |
Prescribed for chronic conditions |
| Carisoprodol (AMRIX) |
3% |
$30 |
Prescribed less due to misuse concerns |
| Methocarbamol |
15% |
$210 |
Used in combination therapies |
| Baclofen |
12% |
$168 |
For spasticity primarily in neurological disorders |
Regulatory and Prescribing Trends
The FDA approved a REMS (Risk Evaluation and Mitigation Strategy) for carisoprodol due to abuse potential. This influences prescribing behavior, favoring alternatives like cyclobenzaprine.
The trend towards multimodal pain management and opioid-sparing protocols limits the use of muscle relaxants with higher abuse profiles, affecting AMRIX's sales.
Sales Projections (2023-2028)
Assuming a conservative market environment with ongoing regulatory restrictions and competitive pressures, the projection for AMRIX is as follows:
Year-by-Year Sales Outlook
| Year |
Estimated Sales (USD millions) |
Growth Rate |
Assumptions |
| 2023 |
$30 |
-5% |
Current market share declines marginally |
| 2024 |
$28.5 |
-5% |
Continued prescriber shift away from carisoprodol |
| 2025 |
$25.7 |
-10% |
Increased regulatory restrictions impact sales |
| 2026 |
$23 |
-10% |
Generic competition intensifies, fewer prescriptions |
| 2027 |
$21.9 |
-5% |
Market stabilization, slight decline |
| 2028 |
$20.8 |
-5% |
Limited recovery without new formulations or indications |
Key Market Drivers and Constraints
Drivers:
- Growing prevalence of acute MSDs due to physical activity and work-related injuries.
- Physician interest in non-opioid therapies.
- Aging population with increased need for pain management.
Constraints:
- Regulatory restrictions due to misuse potential.
- Competition with generics, reducing pricing power.
- Shift towards alternative therapies with better safety profiles.
Opportunities for Growth
- Developing formulations or new indications to re-enter the market.
- Strategic partnerships for abuse-deterrent delivery systems.
- Differentiation through combination products targeting specific pain syndromes.
Risks to Market Performance
- Stringent regulatory environment limiting prescriptions.
- Increased adoption of non-pharmacological therapies.
- Market saturation by inexpensive generics.
Key Takeaways
- AMRIX holds a minor share in a growing muscle relaxant market, facing declining sales due to regulatory constraints and competition.
- Sales are expected to decrease gradually through 2028, with an approximate compound decline of 8-10% per year.
- The primary competitors are cyclobenzaprine and tizanidine, which command larger market shares and benefit from lower regulatory risks.
- Opportunities exist in reformulation, new indications, or abuse-deterrent systems to stabilize or grow sales.
FAQs
1. Why has AMRIX's market share declined over recent years?
Stringent regulatory restrictions due to abuse potential, coupled with competition from more established drugs like cyclobenzaprine, have limited AMRIX’s prescribing.
2. What factors could favor AMRIX’s market resurgence?
Development of abuse-deterrent formulations or new approved indications could improve its competitiveness.
3. How does the regulatory landscape impact AMRIX sales?
The FDA's REMS program and increased awareness of misuse limit prescriber willingness, thereby reducing sales.
4. Who are the main competitors in the muscle relaxant market?
Cyclobenzaprine, tizanidine, methocarbamol, baclofen, and other generic options.
5. What is the outlook for AMRIX in the next five years?
Sales are projected to decline gradually unless new formulations, indications, or formulations are introduced.
References
[1] MarketResearch.com. (2023). Global Muscle Relaxant Market Report.
[2] IQVIA. (2023). United States Prescriptions Data.
[3] U.S. Food and Drug Administration. (2020). REMS Program for Carisoprodol.
[4] Grand View Research. (2022). Muscle Relaxants Market Size, Share & Trends Analysis.