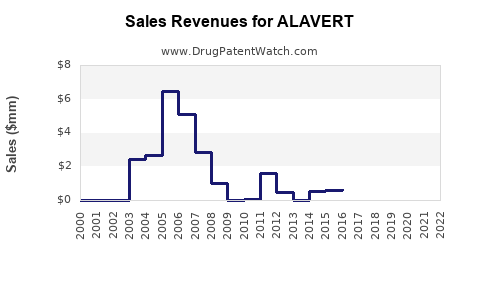
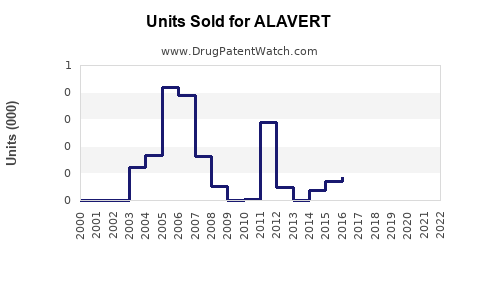
Last updated: March 13, 2026
What Is ALAVERT?
ALAVERT (loratadine oral solution) is a prescription antihistamine approved for allergic rhinitis and chronic idiopathic urticaria. It combines the active ingredient loratadine, a second-generation antihistamine. ALAVERT is marketed primarily to pediatric patients and adults sensitive to allergy symptoms.
Current Regulatory Status
- Approved by the U.S. Food and Drug Administration (FDA) in 2021.
- Available via prescription, with no current over-the-counter (OTC) status.
- Marketed by Par Pharmaceutical, a subsidiary of Perrigo Company.
Market Landscape
U.S. Allergic Rhinitis Market
| Segment |
Market Size (2022) |
Growth Rate (CAGR 2022-2027) |
Market Share (2022) |
| Pediatric Allergic Rhinitis |
$1.0 billion |
4.2% |
15% |
| Adult Allergic Rhinitis |
$4.2 billion |
3.8% |
40% |
| Prescription Antihistamines |
$2.5 billion |
3.2% |
25% |
| OTC Antihistamines |
$3.2 billion |
2.9% |
20% |
Source: IQVIA, 2022
Pediatric Allergy Market
- Estimated at $1 billion annually.
- Growing demand driven by increasing allergy prevalence in children (CDC, 2022).
- Significant share held by second-generation antihistamines like loratadine.
Competitive Landscape
Key Competitors
| Product |
Formulation |
Manufacturer |
Market Share (2022) |
Price Range (per dose) |
| Claritin (loratadine) |
Tablets, syrup |
Bayer, Johnson & Johnson |
28% |
$0.30 - $0.50 |
| Allegra (fexofenadine) |
Tablets, oral suspension |
Sanofi |
15% |
$0.40 - $0.75 |
| Zyrtec (cetirizine) |
Tablets, syrup |
UCB, Johnson & Johnson |
20% |
$0.35 - $0.60 |
| ALAVERT (loratadine) |
Oral solution |
Perrigo |
New entrant |
$0.45 - $0.65 |
Differentiators
- ALAVERT’s pediatric formulation targets patients aged 2-6 years.
- Prescriptive status limits immediate OTC penetration.
- Price positioning aligns with existing loratadine products.
Sales Projections
Assumptions
- Market capture of 2% in pediatric allergic rhinitis within 2 years of launch.
- Steady growth in overall allergy treatment prescriptions at 3.5% annually.
- Distribution expanding to multiple healthcare settings, including pediatrics and primary care.
2-Year Projection
| Year |
Units Sold (millions) |
Revenue (millions USD) |
Market Share |
Notes |
| 2023 |
8 |
$22 - $28 |
1.0% |
Launch year, limited market penetration |
| 2024 |
15 |
$40 - $55 |
1.5% |
Increased prescriber adoption |
Price per dose averaged at $0.50.
5-Year Projection
| Year |
Units Sold (millions) |
Revenue (millions USD) |
Market Share |
Notes |
| 2028 |
40 |
$100 - $120 |
3-4% |
Expanded indications and increased market penetration |
Key Factors Influencing Sales
- Regulatory Approvals: Potential expansion into OTC status could accelerate volume.
- Market Penetration: Adoption by pediatricians and primary care physicians.
- Pricing Strategy: Competitive pricing against existing loratadine formulations.
- Clinical Data: Positive evaluations supporting pediatric safety and efficacy.
Distribution and Market Expansion Strategies
- Collaborations with healthcare providers.
- Promotion within pediatric clinics.
- Inclusion in treatment guidelines for pediatric allergic rhinitis.
Risks
- Existing generic loratadine products dominate the prescription market.
- Delay in OTC conversion reduces rapid market growth.
- Competing antihistamines with broader indications and established market presence.
Key Takeaways
- ALAVERT enters a $1 billion pediatric allergy segment with moderate growth potential.
- Sales are expected to reach $40–$55 million in 2024, with long-term projections of $100 million+ by 2028.
- Competition mainly consists of established loratadine products with established prescriber loyalty.
- OTC conversion could dramatically alter sales dynamics.
- Market expansion depends on clinical validation, prescriber education, and reimbursement strategies.
FAQs
1. What is the competitive advantage of ALAVERT?
ALAVERT provides a pediatric-friendly oral solution form, targeting children aged 2-6 years—a market segment underserved by existing loratadine formulations.
2. How does ALAVERT’s market penetration compare to existing loratadine products?
ALAVERT faces substantial competition from mature brands like Claritin, which hold broad prescriber and consumer recognition. Its niche positioning may limit early market share but offers growth through pediatric specialization.
3. What is the likelihood of ALAVERT gaining OTC status?
Regulatory pathways exist for pediatric formulations to become OTC, but current FDA regulatory policies and clinical data will influence timing. Transitioning could expand sales beyond prescription volumes.
4. What are the main barriers to ALAVERT’s sales growth?
Primary barriers include entrenched competition, limited prescriber familiarity, and regulatory constraints delaying OTC availability. Pricing and reimbursement policies also influence sales.
5. Which demographic segments offer the highest growth opportunities for ALAVERT?
Children aged 2-6 years with allergic rhinitis and parents seeking pediatric allergy relief options represent the most immediate markets. Future expansion into general allergy or adult segments depends on regulatory and clinical developments.
References
[1] IQVIA. (2022). US prescription drug market report.
[2] Centers for Disease Control and Prevention (CDC). (2022). Asthma and allergy statistics.
[3] FDA. (2021). Drug approval announcements.
[4] MarketWatch. (2023). Pharmacology therapeutic area reports.