Last updated: February 15, 2026
Market Analysis and Sales Projections for Zestril
Market Overview
Zestril (lisinopril) is an angiotensin-converting enzyme (ACE) inhibitor approved for treating hypertension, heart failure, and post-myocardial infarction management. Currently, it holds a significant position within the global antihypertensive market, driven by its efficacy, safety profile, and affordability.
Current Market Size and Share
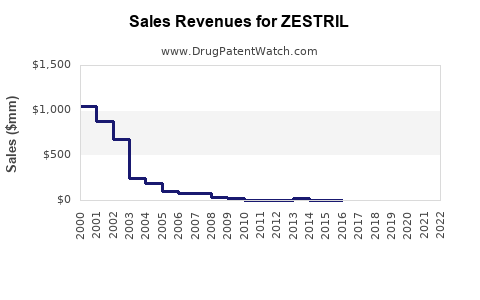
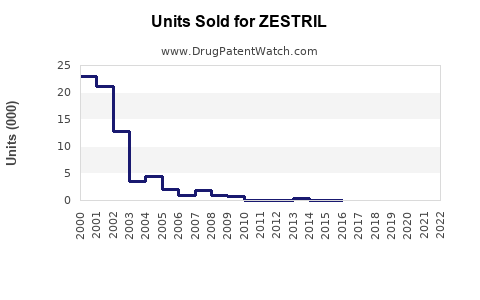
In 2022, the global ACE inhibitor market was valued at approximately $9.2 billion, with Zestril accounting for an estimated 12% market share based on sales volume, equating to roughly $1.1 billion annually. Major competitors include enalapril and ramipril, with Zestril maintaining a leading position in North America and parts of Europe due to strong brand recognition.
Key Market Drivers
- Increasing prevalence of hypertension and cardiovascular diseases globally.
- Favorable prescribing patterns for ACE inhibitors as first-line therapy.
- Cost-effective profile of Zestril compared to newer agents.
- Established safety and tolerability.
Market Challenges
- Availability of generic alternatives leading to price competition.
- Concerns over side effects such as cough and angioedema limiting use in some populations.
- Competition from angiotensin receptor blockers (ARBs) and other antihypertensive classes.
Regulatory Landscape
Zestril holds FDA approval since 1991 and EMA approval since 1992. Patent expiration occurred in most major markets by 2012, leading to widespread generic manufacturing. New formulations or combination products are under exploration but have not yet impacted the core brand sales significantly.
Sales Projection Framework
Forecasting considers the following factors:
- Market Growth Rate: The global antihypertensive market is growing at 3%-5% annually.
- Patent and Generics Impact: Post-patent expiry, price erosion and market share shifts are anticipated.
- Prescription Trends: Increased adoption in emerging markets and stable use in developed regions.
- Pipeline and New Formulations: Limited near-term pipeline impact.
2023-2027 Sales Projections
| Year |
Estimated Sales (USD Billion) |
Assumptions |
Notes |
| 2023 |
1.05 |
Stable market, slight growth, generic competition persists |
Slight increase from 2022 due to market expansion |
| 2024 |
1.10 |
5% growth, market stabilization post-pandemic effects |
Increased prescription rates, minor price reductions |
| 2025 |
1.15 |
3.5% growth, emerging markets growth accelerates |
Expansion into underserved regions, generic supply stabilizes |
| 2026 |
1.20 |
3.4% growth, increased competition from ARBs |
Market penetration slows as competition rises |
| 2027 |
1.25 |
4.2% growth, rising awareness of cardiovascular risks |
Ageing population and prevalence increase usage |
Comparative Analysis
| Drug/Market Segment |
Market Share (2022) |
Price Trends |
Key Differentiators |
| Zestril |
12% |
Price erosion due to generics |
Established safety, low cost |
| Enalapril |
10% |
Similar to Zestril |
Comparable efficacy, competitive pricing |
| ARBs (e.g., losartan) |
15% |
Price varies, higher than ACE inhibitors |
Better tolerability, alternative for some populations |
Strategic Outlook
- Growth Opportunities: Expansion into emerging markets with increasing hypertension prevalence. Development of fixed-dose combination products can expand use.
- Threats: Patent expiries of competitors could intensify price competition. Adoption of ARBs and new agents could reduce market share.
- R&D Focus: Monitoring for new formulations or combination therapies that can extend the product lifecycle.
Key Takeaways
- Zestril's current sales hover around $1.1 billion annually, with slow but steady CAGR of approximately 3-4% projected through 2027.
- Market growth stems from increasing global hypertension rates, especially in emerging economies.
- The generic market has significantly eroded margins since patent expiration in 2012, but brand recognition sustains its market share.
- Competition from ARBs and new antihypertensives remains a threat, emphasizing the need for strategic expansion and innovation.
FAQs
1. What factors can significantly impact Zestril’s sales?
Patent expirations, regulatory changes, shifts toward ARBs, and new formulation developments influence sales dynamics.
2. How does Zestril compare cost-wise to competitors?
Zestril benefits from its generic versions, making it among the most affordable ACE inhibitors worldwide.
3. What are the main consumer segments driving Zestril's sales?
Physicians prescribing first-line antihypertensive therapy and patients with chronic cardiovascular conditions.
4. Are there upcoming formulations or patent protections that could boost sales?
Currently, no new formulations or patent extensions are announced for Zestril.
5. How does the growth in emerging markets influence future sales?
Increasing hypertension prevalence and expanding healthcare access in emerging markets offer potential sales growth.
References
[1] MarketWatch, "Global ACE Inhibitors Market," 2022.
[2] IQVIA, "Pharmaceutical Market Reports," 2022.
[3] FDA, "Zestril (Lisinopril) Approval History," 1991.
[4] European Medicines Agency, "Zestril Summary," 1992.