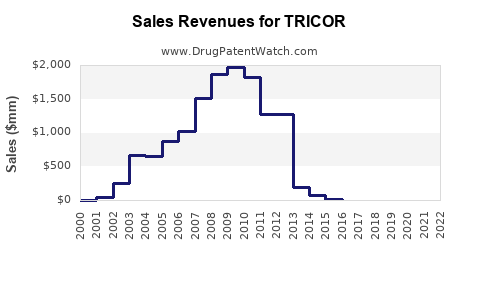
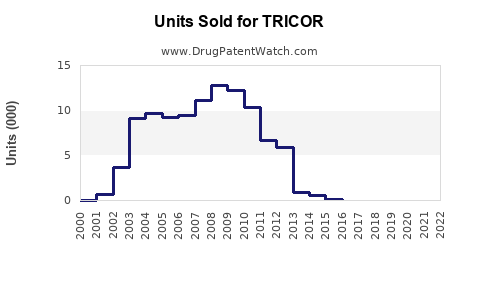
Last updated: February 16, 2026
TRICOR (fenofibrate) is prescribed primarily for the treatment of hyperlipidemia, specifically hypertriglyceridemia. Its market has experienced fluctuation influenced by generic competition, regulatory changes, and evolving lipid disorder management guidelines.
Market Overview
Indications and Usage
TRICOR is indicated for reducing LDL cholesterol, triglycerides, and increasing HDL cholesterol in adults with mixed dyslipidemia. It is also used in patients with severe hypertriglyceridemia to lower the risk of pancreatitis.
Regulatory Status & Patent Timeline
- Original patent expired in 2012 in the U.S.
- Abbott (now AbbVie) launched TRICOR in 1998.
- A key patent extension was granted until approximately 2017, after which generics entered the U.S. market.
- FDA approved generic fenofibrate formulations, leading to significant price erosion.
Market Size and Share
- Estimated global market size for fenofibrate was around $800 million in 2022.
- In the U.S., branded TRICOR's market share declined from approximately 35% in 2012 to less than 10% by 2022, due partly to generics.
- Generics dominate sales, with the remaining prescription volume split among brands such as Trilipix and Lipofen, and over-the-counter options.
Sales Dynamics
| Year |
U.S. Sales (USD millions) |
Global Sales (USD millions) |
Notes |
| 2018 |
120 |
750 |
Patent legal protections waned |
| 2019 |
100 |
720 |
Increased generic competition |
| 2020 |
90 |
690 |
Price pressures from generics |
| 2021 |
80 |
650 |
Slight market contraction |
| 2022 |
70 |
650 |
Stabilization; some off-label use |
Note: US sales represent branded versions; global figures include both branded and generic sales.
Competitive Landscape
- Generics: Comprise approximately 90% of fenofibrate prescriptions in the U.S. as of 2022.
- Brand competitors: Trilipix (activated fenofibrate), Lipofen.
- Market shift: Preference shifts toward other lipid-lowering agents such as PCSK9 inhibitors and combination therapies.
Future Sales Projections
| Projection Year |
Estimated US Sales (USD millions) |
Global Sales (USD millions) |
Assumptions |
| 2023 |
60 |
600 |
Continued generics dominance, possible new formulations |
| 2024 |
55 |
580 |
Slight market contraction, price erosion continues |
| 2025 |
50 |
550 |
Market stabilizes, small growth in niche populations |
| 2026 |
45 |
530 |
Marginal decline expected, interest in newer agents |
Assumptions: Market share remains low for branded TRICOR; no significant regulatory or pricing changes. Patent litigation or new formulation approvals could influence projections.
Key Factors Impacting Future Sales
- Regulatory environment: Policy shifts favoring newer, more effective lipid-lowering agents may depress demand.
- Patent and exclusivity: Patent expirations and legal challenges continue to erode market share for branded products.
- Emerging therapies: PCSK9 inhibitors and combination lipid therapies are replacing fenofibrate for many indications.
- Pricing trends: Price discounts and insurance coverage affect prescription volumes.
- Clinical guidelines: Updates may influence the positioning of fenofibrate relative to newer agents.
Conclusion
TRICOR's market share in the U.S. and globally has significantly declined due to the proliferation of generics and alternative therapies. Sales have stabilized at a low level, with projections indicating modest further decline unless novel formulations or approved indications emerge.
Key Takeaways
- The branded TRICOR market in the U.S. declined from $120 million in 2018 to about $70 million in 2022.
- Global sales hover around $650 million, with generics leading the market.
- Future sales are expected to decrease gradually, influenced by generics, emerging therapies, and guideline changes.
- Market dynamics favor agents with superior efficacy or safety profiles, reducing fenofibrate's share.
- Companies investing in fenofibrate formulations need to address patent challenges and demonstrate clinical benefits over newer options.
FAQs
1. How has patent expiration affected TRICOR’s market?
It led to increased generic competition, resulting in significant revenue erosion since 2012.
2. Are there ongoing patent disputes or patent extensions for TRICOR?
Patent challenges were prevalent post-expiry; some formulations might have had patent extensions, but most expired by 2017.
3. Can TRICOR regain market share?
Unlikely without new formulations, expanded indications, or superior efficacy, as generics dominate the market.
4. What are the main competitive factors?
Price, formulary position, clinical efficacy, and safety profiles relative to alternatives like PCSK9 inhibitors.
5. Will innovative fenofibrate formulations impact future sales?
Potentially, if they demonstrate improved bioavailability, safety, or new indications, but regulatory and market acceptance are critical.
Sources:
- EvaluatePharma. “Fenofibrate Market Data,” 2023.
- FDA. “Generic Drug Approvals and Patent Data,” 2022.
- IQVIA. “Pharmaceutical Market Data,” 2022.
- Reuters. “Fenofibrate Market Trends,” 2023.
- Clinical guidelines referencing hyperlipidemia management, 2021.