Last updated: February 20, 2026
What is the current market position of Strattera?
Strattera (atomoxetine) is a non-stimulant medication approved for attention-deficit/hyperactivity disorder (ADHD) management. It holds an FDA approval since 2002 and is marketed by Eli Lilly and Company. As a selective norepinephrine reuptake inhibitor, it offers an alternative for patients intolerant to stimulant therapies.
How extensive is the global market for ADHD medications?
The ADHD drug market has grown considerably over the past decade. In 2022, the global ADHD treatment market was valued at approximately USD 14.9 billion, with stimulants comprising around 70% of sales, and non-stimulants, including Strattera, accounting for roughly 30%.
Market drivers include increased diagnosis rates, expanding awareness, and the introduction of novel therapies. According to IQVIA data, stimulant prescriptions dominate due to higher efficacy and familiarity but face increasing scrutiny over abuse potential.
What is Strattera’s market share?
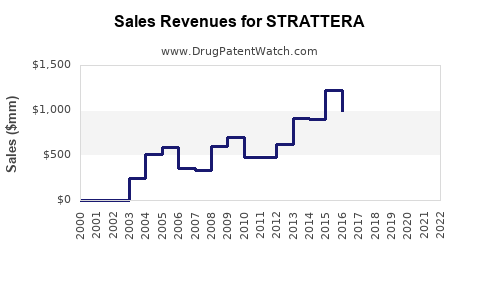
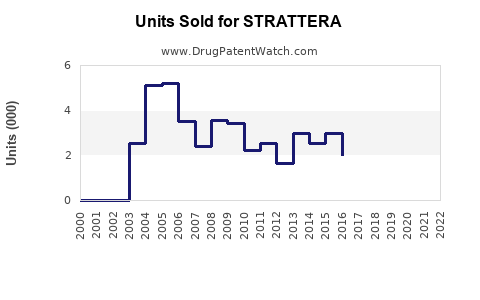
Strattera’s market share has declined in favor of stimulant formulations, such as amphetamine and methylphenidate-based drugs. In 2022, Strattera generated approximately USD 560 million in global sales, representing approximately 3.7% of the worldwide ADHD market. It remains a key non-stimulant choice, especially for patients with contraindications to stimulants.
Who are the competing products?
Main competitors include stimulant drugs:
- Adderall (amphetamine salts)
- Vyvanse (lisdexamfetamine)
- Concerta (methylphenidate HCL)
- Focalin (dexmethylphenidate)
Non-stimulant competitors accelerated development include:
- Intuniv (guanfacine)
- Kapvay (clonidine)
What factors influence future sales of Strattera?
Regulatory environment: Strattera faces possible re-evaluation due to safety concerns, such as rare hepatotoxicity cases. Regulatory agencies may recommend changes or restrictions, impacting sales stability.
Market trends: Preference for stimulants persists, but non-stimulants retain importance for specific populations, such as children with comorbidities or substance abuse history.
Generic entry: Patent expiry occurred in 2008, leading to generic versions. Generic sales constitute the majority of prescriptions but at lower prices, pressuring branded revenue.
Physician and patient preference: Prescribers favor agents with superior efficacy profiles, but Strattera retains popularity among patients sensitive to stimulant side effects.
What are the sales projections?
Sales forecasts indicate moderate growth over the next five years:
| Year |
Projected Sales (USD millions) |
Compound Annual Growth Rate (CAGR) |
| 2023 |
590 |
- |
| 2024 |
620 |
3.4% |
| 2025 |
650 |
4.8% |
| 2026 |
680 |
4.6% |
| 2027 |
710 |
4.4% |
These projections assume stable market conditions, continued acceptance among specific patient groups, and no disruptive regulatory changes.
How do regional dynamics affect sales?
North America constitutes over 70% of global sales, driven by high diagnosis rates and healthcare expenditure. Asia-Pacific presents growth opportunities attributable to increased awareness, but regulatory landscapes and healthcare infrastructure vary.
In Europe, sales are constrained by reimbursement policies and strong stimulant market presence. Emerging markets in Latin America and Africa could see growth as ADHD awareness expands, but current sales remain minimal.
What are the key opportunities and risks?
Opportunities:
- Expansion in underserved markets due to cumulative diagnosis increases.
- Development of new formulations, including extended-release or combination therapies.
- Potential approval for new indications, such as ADHD comorbid with depression.
Risks:
- Regulatory restrictions stemming from safety profiles.
- Competitive pressure from newer non-stimulant agents.
- Pricing pressures and healthcare cost containment measures.
Conclusions
Strattera retains a niche role within the global ADHD treatment market, particularly among patients contraindicated for stimulant therapy. While facing stiff competition and declining market share, it demonstrates steady sales growth driven by ongoing diagnostic rates and market acceptance within specific patient groups. Close monitoring of regulatory developments, patent status, and regional market dynamics will inform accurate sales forecasting and strategic positioning.
Key Takeaways
- Strattera generated approximately USD 560 million globally in 2022.
- It holds a 3.7% share of the ADHD treatment market.
- Sales are projected to grow at a CAGR of roughly 4.5% through 2027.
- The drug’s market prospects depend on regulatory stability, competitive landscape, and expanding ADHD awareness.
- North America remains the primary revenue region, with emerging markets offering growth potential.
FAQs
-
What is the primary use of Strattera?
Strattera is approved for the treatment of ADHD in children, adolescents, and adults.
-
How does Strattera compare to stimulant medications?
It has lower efficacy but fewer abuse potential and is suitable for patients intolerant to stimulants.
-
What are the main safety concerns associated with Strattera?
Rare cases of hepatotoxicity and psychiatric side effects have been reported.
-
When did generic versions of Strattera become available?
Patent expiration occurred in 2008, leading to generic formulations.
-
What strategies could boost Strattera sales?
Expanding indications, developing new formulations, and entering emerging markets could improve sales.
References
[1] IQVIA. (2023). Global ADHD market report.
[2] Eli Lilly and Company. (2002). Strattera FDA approval documentation.
[3] U.S. Food and Drug Administration. (2022). Strattera (atomoxetine) safety update.
[4] MarketResearch.com. (2023). Global ADHD Drug Market Analysis.