Last updated: February 14, 2026
What Is RALOXIFENE and Its Market Position?
RALOXIFENE is a selective estrogen receptor modulator (SERM) primarily used for the prevention and treatment of postmenopausal osteoporosis and to reduce the risk of invasive breast cancer in high-risk women. It is marketed under the brand name Evista by Eli Lilly and Company. The drug operates by binding to estrogen receptors, exerting both estrogen-agonist and antagonist effects depending on the tissue.
What Are the Current Market Dynamics?
The global osteoporosis drug market was valued at approximately USD 11 billion in 2022. Raloxifene accounts for a significant segment within this space, primarily driven by its dual indications and favorable safety profile compared to bisphosphonates. Its adoption is higher in the United States, driven by lengthy approval history and established guidelines.
The breast cancer prevention market, where Raloxifene competes with drugs like tamoxifen, is also expanding. As of 2022, the U.S. FDA approved Raloxifene for reducing the risk of invasive breast cancer in women at high risk.
What Are Current Sales Figures and Market Share?
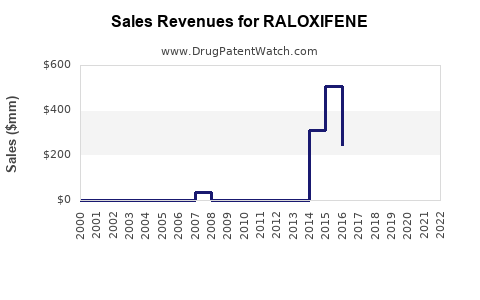
Eli Lilly reported worldwide sales of Evista at approximately USD 540 million in 2022, representing a decline from USD 600 million in 2021. The decrease reflects generic competition and patent expiry pressures.
In the U.S., Evista holds an estimated 18% market share in the osteoporosis segment, with the remainder split among bisphosphonates (e.g., alendronate and risedronate). Its breast cancer prevention indication is prescribed to a smaller patient population, with annual sales around USD 120 million globally.
What Are Sales Projections for the Next Five Years?
Projections consider patent expiry, competition, aging populations, and evolving treatment guidelines:
| Year |
Estimated Global Sales (USD millions) |
Assumptions |
| 2023 |
510 |
Continued generic erosion, moderate decline |
| 2024 |
490 |
Increased generic presence, stabilization |
| 2025 |
460 |
Market share reductions due to new therapies |
| 2026 |
420 |
Emerging alternatives limit growth |
| 2027 |
380 |
Further generic penetration, patent expiration |
Sales are expected to decline gradually, reaching about USD 380 million by 2027 absent new indications or formulations.
Key Growth Drivers
- Aging population increases osteoporosis and breast cancer risk.
- Growing awareness of osteoporosis management.
- Preventative use in high-risk women.
Key Challenges
- Patent expiry slated for 2024 in major markets.
- Competition from bisphosphonates and new agents such as denosumab.
- Generic entry erodes brand sales.
What Are Market Entry and Development Opportunities?
Inclusion of Raloxifene in combination therapies, new formulations (e.g., injectable or transdermal), or expanded indications (e.g., cardiovascular protection) could stabilize or grow its market share. Also, targeted marketing strategies toward high-risk populations may improve sales longevity.
What Is the Competitive Landscape?
Raloxifene faces competition primarily from:
- Bisphosphonates: Alendronate, risedronate, and zoledronic acid; higher sales volumes but different side effect profiles.
- Selective ER Modulators: Bazedoxifene, which is under development.
- Emerging Agents: Denosumab (Prolia), with larger adoption for osteoporosis and cancer-related indications.
How Do Pricing and Reimbursement Impact Sales?
Pricing strategies are influenced by generic competition, with brand sales declining as generics enter. Reimbursement policies, especially in the U.S., favor cost-effective treatments. Optimizing reimbursement pathways is crucial for maintaining sales margins.
Summary
RALOXIFENE currently generates annual sales close to USD 500 million globally, with a declining trend due to patent expiration and competitive pressures. Future growth hinges on new indications, formulations, and strategic market positioning.
Key Takeaways
- Raloxifene’s primary markets are osteoporosis and breast cancer prevention, with sales dominated by the U.S.
- Patent expiration in 2024 will accelerate generic erosion, reducing brand sales.
- The market is heavily competitive, with bisphosphonates and denosumab as primary rivals.
- Opportunities include new formulations and expanded indications.
- Market revenue is projected to decline to USD 380 million by 2027 without strategic innovations.
FAQs
1. When does the patent for Raloxifene expire?
Patents in major markets, including the U.S., are set to expire in 2024, enabling generic entries.
2. What are the main competitors to Raloxifene?
Bisphosphonates (e.g., alendronate) and denosumab (Prolia) are primary competitors, offering alternative treatment options with broader or different indications.
3. Can Raloxifene be used for cardiovascular protection?
Current approvals do not include cardiovascular indications. Research evaluating such indications is ongoing but not yet incorporated into standard practice.
4. What factors could reverse sales declines?
Expansion into new indications, improved formulations, or strategic partnerships developing into combination therapies could mitigate sales decline.
5. How significant is the brand Raloxifene in the osteoporosis market?
It accounts for roughly 18% market share in the U.S., primarily driven by its breast cancer prevention indication.
Sources:
- Eli Lilly Annual Reports 2022.
- MarketWatch: Global Osteoporosis Drugs Market 2022.
- U.S. FDA drug approvals.
- Bloomberg Intelligence: Biopharma Market Trends.