Share This Page
Drug Sales Trends for PULMICORT
✉ Email this page to a colleague
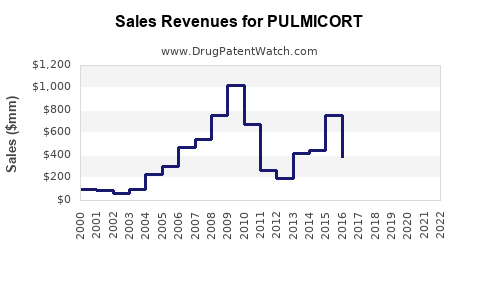
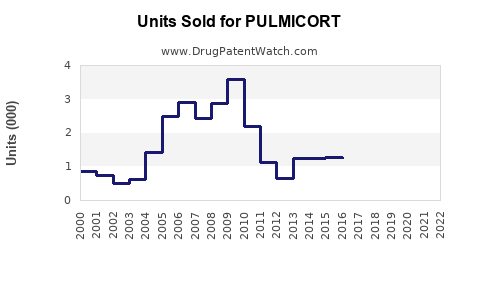
Annual Sales Revenues and Units Sold for PULMICORT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PULMICORT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PULMICORT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PULMICORT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PULMICORT: Market Landscape and Sales Projections
PULMICORT (budesonide) is a widely prescribed inhaled corticosteroid (ICS) used for the maintenance treatment of asthma. The global market for PULMICORT is characterized by a mature but stable demand driven by the prevalence of respiratory diseases, particularly asthma. Key market segments include pediatric and adult populations. The market faces competition from other ICS brands, generic budesonide formulations, and alternative asthma therapies. Sales are influenced by factors such as prescription rates, healthcare access, formulary placement, and patent expiries. Projections indicate continued steady growth, albeit at a moderate pace, with significant regional variations.
What is the current market position of PULMICORT?
PULMICORT, developed by AstraZeneca, holds a significant position in the global asthma treatment market. Its primary indication is for the maintenance treatment of asthma in patients aged 12 months and older. The drug is available in multiple formulations, including PULMICORT RESPULES (nebulized suspension) for infants and young children, and PULMICORT TURBOHALER (dry powder inhaler) for older children and adults.
Key Market Attributes:
- Brand Recognition: PULMICORT is a well-established and recognized brand within the ICS class.
- Efficacy and Safety Profile: The drug has a long history of use and a well-documented efficacy and safety profile for long-term asthma management.
- Formulation Diversity: The availability of both nebulized and dry powder inhaler formulations caters to different patient age groups and administration preferences.
- Prescription Volume: PULMICORT consistently ranks among the top ICS prescriptions globally.
- Market Share: While specific global market share figures fluctuate, PULMICORT remains a leading product within the ICS segment.
Competitive Landscape:
The ICS market is competitive. PULMICORT competes with other branded ICS products such as FLONASE (fluticasone propionate) and SYMBICORT (budesonide/formoterol), as well as numerous generic budesonide formulations. The introduction of combination inhalers, which pair an ICS with a long-acting beta-agonist (LABA), also presents a competitive dynamic. However, PULMICORT's monotherapy status and established patient base continue to support its market presence.
Market Dynamics:
- Asthma Prevalence: The ongoing prevalence of asthma globally, driven by environmental factors, genetics, and improved diagnostics, underpins sustained demand for maintenance therapies like PULMICORT. The World Health Organization estimates that over 300 million people worldwide have asthma.
- Generic Competition: The expiry of key patents for PULMICORT has led to the availability of generic budesonide inhalers. These generics typically offer lower price points, influencing market share dynamics and potentially eroding brand loyalty for some patient segments.
- Healthcare Policies and Reimbursement: Reimbursement policies, formulary decisions by insurance providers, and government healthcare initiatives impact the accessibility and affordability of PULMICORT, thereby affecting sales volumes.
- Physician Prescribing Habits: Physician preference, informed by clinical trial data, patient outcomes, and marketing efforts, is a critical driver of PULMICORT's market penetration.
What are the primary indications and patient populations for PULMICORT?
PULMICORT's primary indication is for the maintenance treatment of asthma. It is not used for the relief of acute bronchospasm.
Key Indications and Patient Groups:
- Asthma Maintenance: The core use of PULMICORT is to prevent and control asthma symptoms, including wheezing, shortness of breath, and chest tightness, on a daily basis. It reduces inflammation in the airways.
- Pediatric Patients: PULMICORT RESPULES are particularly important for infants and young children who may have difficulty using dry powder inhalers. Studies have demonstrated its efficacy and safety in this age group. For instance, a study published in the New England Journal of Medicine indicated that inhaled budesonide was effective in reducing exacerbations in children with mild to moderate asthma (1).
- Adult Patients: PULMICORT TURBOHALER is prescribed for adults and older children to manage their asthma.
- Patients with Moderate to Severe Asthma: While also used in milder cases, PULMICORT is a cornerstone therapy for patients with moderate to severe persistent asthma who require daily treatment to maintain symptom control.
Exclusions:
- PULMICORT is not indicated for the relief of acute bronchospasm or status asthmaticus.
- It is not a substitute for rapid-acting bronchodilators.
Dosage and Administration:
- PULMICORT RESPULES: Administered via a jet nebulizer. Dosing is typically once or twice daily.
- PULMICORT TURBOHALER: A dry powder inhaler that requires the patient to inhale deeply and quickly. Dosing is also typically once or twice daily.
The specific dosage is determined by the severity of the patient's asthma and their response to treatment.
What are the key patent expiries and their impact on the market?
The patent landscape for PULMICORT has significantly evolved, leading to increased generic competition. The active pharmaceutical ingredient, budesonide, has been off-patent in major markets for some time.
Key Patent Expiries and Status:
- Active Ingredient Patents: The primary patents protecting the budesonide molecule expired years ago in the United States and Europe. For example, U.S. Patent No. 4,250,166, related to budesonide, expired in the early 2000s.
- Formulation and Delivery System Patents: AstraZeneca has historically held patents related to specific PULMICORT formulations and its delivery devices, such as the TURBOHALER. These patents have also expired or are nearing expiry in various jurisdictions. The TURBOHALER device patent, for instance, has faced challenges and expired in key markets.
- Market Entry of Generics: Following patent expiries, numerous generic manufacturers have launched budesonide inhalation suspensions and dry powder inhalers. These generics offer significant cost savings compared to the branded PULMICORT.
Impact on the Market:
- Price Erosion: The entry of generics has led to substantial price erosion for budesonide inhalers. This directly impacts the revenue generated by branded PULMICORT.
- Market Share Shift: Generic budesonide products have captured a significant share of the market, particularly among price-sensitive payers and patients.
- Increased Accessibility: Lower prices for generic budesonide have improved accessibility for a broader patient population, potentially increasing overall budesonide utilization.
- AstraZeneca's Strategy: AstraZeneca has focused on maintaining its market share through brand loyalty, physician education, and the development of new combination products (e.g., Symbicort). However, the direct PULMICORT brand faces ongoing pressure.
- Regional Variations: The impact of patent expiry and generic penetration varies by country, depending on local patent laws, regulatory approval processes for generics, and national healthcare policies.
The primary impact of patent expiries on PULMICORT is the intensified competition from lower-cost generic alternatives, leading to a more fragmented and price-sensitive market segment.
What are the projected sales figures and growth drivers for PULMICORT and its generics?
Projecting precise future sales figures for a branded product facing significant generic competition is complex. Instead, the analysis focuses on the overall budesonide inhalation market, which includes branded PULMICORT and its generic counterparts.
Projected Market Growth for Budesonide Inhalations (Global):
The global market for budesonide inhalations is projected to experience moderate, steady growth over the next five to seven years.
- Estimated Current Market Size (Budesonide Inhalations): Approximately $2.5 billion to $3.0 billion USD (aggregate for branded and generic, global).
- Projected Compound Annual Growth Rate (CAGR): 3% to 5% annually.
Key Growth Drivers:
- Continued Asthma Prevalence: The persistent and rising global prevalence of asthma, particularly in developing economies, will sustain demand for maintenance therapies.
- Increasing Access to Healthcare: Improvements in healthcare infrastructure and increased insurance coverage in emerging markets will expand the patient base for inhaled therapies.
- Pediatric Use: The established safety and efficacy of budesonide in pediatric populations, especially via nebulization (RESPULES), continue to drive demand in this critical segment.
- Physician Familiarity and Trust: Healthcare providers have extensive experience with budesonide, fostering continued prescribing habits.
- Cost-Effectiveness of Generics: The availability of affordable generic budesonide makes it a preferred option for many healthcare systems and patients, supporting overall market volume.
- Slight Increase in Asthma Diagnoses: Better diagnostic tools and increased awareness may lead to more diagnoses and subsequent treatment initiation.
Factors Limiting Growth:
- Intensified Generic Competition: Ongoing price competition among generic manufacturers will cap revenue growth for the overall market, even if unit sales increase.
- Advancement of Combination Therapies: The market is increasingly shifting towards ICS/LABA combinations for more severe asthma, which may reduce the standalone use of budesonide monotherapy in certain patient segments.
- Emergence of Novel Therapies: Biologics and other novel treatments for severe asthma may divert some patients away from traditional ICS therapies, though these are typically for refractory cases.
- Stagnant or Declining Use in Developed Markets: In highly developed markets, the budesonide market may see flat or only marginal growth due to saturation and the prevalence of combination therapies.
Sales Projections for PULMICORT (Branded):
AstraZeneca's net sales for PULMICORT have been impacted by generic erosion. Sales figures are expected to continue a downward trend or stabilize at significantly lower levels compared to its peak.
- Estimated Current Annual Sales (PULMICORT Branded): Approximately $800 million to $1 billion USD (global).
- Projected Trend: Expect a continued decline in nominal sales, with sales stabilizing or growing negligibly at best, primarily driven by specific regions with slower generic uptake or strong brand loyalty.
Sales Projections for Generic Budesonide:
The generic budesonide segment is expected to show robust volume growth, translating into moderate revenue growth for the aggregate generic market.
- Projected CAGR (Generic Budesonide): 5% to 7%.
- Drivers: Increased prescription volume due to cost-effectiveness, wider market penetration in emerging economies, and aggressive pricing strategies by generic manufacturers.
The overall budesonide inhalation market is projected to see revenue growth driven by volume increases in the generic segment, offsetting the decline in branded PULMICORT sales.
What are the key regulatory and policy considerations impacting PULMICORT?
Regulatory and policy environments significantly shape the market access, pricing, and prescribing patterns for PULMICORT and its generic equivalents.
Key Regulatory and Policy Considerations:
-
FDA (U.S. Food and Drug Administration) and EMA (European Medicines Agency) Approvals:
- Approval Pathways: Both branded PULMICORT and its generic versions must undergo rigorous review and approval processes by regulatory bodies like the FDA and EMA. This includes demonstrating safety, efficacy, and bioequivalence for generics.
- Labeling Requirements: Regulatory agencies dictate the approved indications, contraindications, warnings, and precautions for PULMICORT, influencing physician prescribing.
- Post-Market Surveillance: Ongoing pharmacovigilance and risk management programs are mandated, which can lead to label updates or further regulatory scrutiny.
-
Generic Drug Policies:
- ANDA (Abbreviated New Drug Application): The FDA's ANDA pathway allows for the approval of generic drugs that are therapeutically equivalent to branded drugs. The Hatch-Waxman Act facilitates generic entry.
- Bioequivalence Standards: Generics must meet stringent bioequivalence standards, meaning they must deliver the same amount of active ingredient into the bloodstream as the branded drug.
- Patent Litigation: Patent challenges and litigation by generic companies against branded drug patents can accelerate generic market entry.
-
Healthcare Reimbursement and Payer Policies:
- Formulary Placement: Inclusion on insurance formularies is critical for market access. Payers often tier medications, with generics typically placed in lower, more cost-effective tiers.
- Prior Authorization: Some payers may require prior authorization for branded PULMICORT, especially if a therapeutically equivalent generic is available, encouraging generic substitution.
- Step Therapy: Payers may implement step-therapy protocols, requiring patients to first try a lower-cost generic option before approving a more expensive branded drug.
- Rebate Negotiations: Pharmaceutical manufacturers negotiate rebates with payers to secure favorable formulary status. The dynamics of these negotiations impact the net price and profitability of branded PULMICORT.
-
Government Healthcare Programs:
- Medicare and Medicaid (U.S.): These programs represent significant purchasing power and have specific rules regarding generic substitution and cost-containment.
- National Health Services (U.K., Canada, etc.): In countries with national health systems, central purchasing bodies and drug reimbursement committees play a key role in deciding which medications are available and at what cost.
-
Inhaler Device Regulations:
- Device Safety and Performance: Regulatory bodies also oversee the safety and performance of inhaler devices. Changes to the TURBOHALER or the approval of new generic device components can have market implications.
-
Environmental Regulations (Manufacturing):
- While not directly impacting patient use, environmental regulations related to pharmaceutical manufacturing can influence production costs for both branded and generic manufacturers.
The overarching regulatory trend is the push for generic substitution and cost containment, which continues to pressure the market position of branded PULMICORT, while facilitating broader access to budesonide therapy through generics.
What are the key competitive threats and opportunities for PULMICORT?
PULMICORT operates in a dynamic therapeutic area, facing evolving competitive pressures and limited opportunities for significant expansion as a monotherapy.
Key Competitive Threats:
-
Generic Budesonide Inhalers: This is the most significant threat. The availability of multiple generic versions of budesonide (both suspension and dry powder) at substantially lower price points erodes PULMICORT's market share and profitability.
- Example: Numerous manufacturers, including Teva Pharmaceuticals, Sun Pharmaceutical Industries, and Cipla, offer generic budesonide products.
-
ICS/LABA Combination Inhalers: Products that combine budesonide with a long-acting beta-agonist (LABA), such as Symbicort (budesonide/formoterol), are a major competitive force, especially for patients with moderate to severe asthma who require both bronchodilation and anti-inflammatory action.
- Example: Symbicort (AstraZeneca) offers a dual mechanism of action, providing both maintenance and rescue therapy for some patients, which can simplify treatment regimens. Other ICS/LABA combinations like Advair/Seretide (fluticasone/salmeterol) also compete for the same patient population.
-
Other Branded ICS Products: While budesonide is a highly effective ICS, other branded ICS molecules (e.g., fluticasone propionate, mometasone furoate) continue to be prescribed, offering physicians alternative options.
-
Emerging Novel Asthma Therapies: Biologics and other targeted therapies (e.g., anti-IL5, anti-IgE) are increasingly used for severe, difficult-to-treat asthma. While these are not direct competitors for mild-to-moderate asthma treated by PULMICORT, they represent a diversion of R&D focus and investment within the broader asthma market.
-
Shifting Treatment Paradigms: A move towards personalized medicine and treat-to-target approaches may favor more individualized treatment strategies, potentially reducing reliance on fixed-dose monotherapies for some patient profiles.
Key Opportunities:
-
Continued Demand in Emerging Markets: As healthcare access and asthma diagnosis rates increase in emerging economies, there is an opportunity for sustained demand for cost-effective therapies like budesonide, including branded PULMICORT where payer dynamics allow.
- Example: Markets in Asia-Pacific and Latin America are experiencing significant growth in pharmaceutical spending and healthcare access.
-
Pediatric Niche: PULMICORT RESPULES remain a vital and preferred option for infants and young children due to ease of administration with nebulizers. This segment is relatively less susceptible to direct competition from dry powder inhaler generics.
-
Physician and Patient Loyalty: Decades of clinical experience and established trust in PULMICORT's efficacy and safety can foster continued physician prescribing and patient adherence, especially among long-term users who are comfortable with the product.
-
Potential for New Indications (Limited): While the primary indication is well-established, there is always a remote possibility of exploring or re-evaluating budesonide for other inflammatory respiratory conditions, although significant investment in this area for a mature molecule is unlikely.
-
Lifecycle Management (Limited): While major patent expiries have occurred, limited opportunities for lifecycle management might exist through novel drug delivery systems or patient support programs, though these are unlikely to drive substantial new revenue.
The primary competitive threat is the ongoing commoditization of budesonide through generics, limiting PULMICORT's growth as a branded monotherapy. Opportunities are largely confined to leveraging its established profile in specific niches and regions.
Key Takeaways
PULMICORT (budesonide) remains a significant product in the asthma maintenance treatment market, underpinned by its efficacy, safety profile, and established brand recognition. The market for budesonide inhalations is projected to grow at a moderate 3-5% CAGR globally, driven by persistent asthma prevalence and increasing healthcare access in emerging markets. However, branded PULMICORT faces substantial revenue pressure due to patent expiries and the widespread availability of lower-cost generic budesonide inhalers. Generic budesonide is expected to grow at 5-7% CAGR, capturing increasing market volume. Competitive threats are dominated by generic erosion and the growing adoption of ICS/LABA combination therapies. Opportunities for branded PULMICORT are primarily limited to maintaining its position in the pediatric nebulized segment and leveraging residual brand loyalty and market access in specific geographic regions. Regulatory policies and payer reimbursement strategies continue to favor generic substitution, further challenging the market position of the branded product.
FAQs
-
Will PULMICORT be discontinued by AstraZeneca? AstraZeneca has not announced plans to discontinue the PULMICORT brand. The company continues to market and support the product, particularly in regions where branded products maintain market share and in specific formulations like RESPULES for pediatric use.
-
How does PULMICORT compare in price to generic budesonide? Generic budesonide inhalers are significantly less expensive than branded PULMICORT. Prices for generic versions can be 30% to 70% lower, depending on the manufacturer, formulation, and geographic market.
-
Is PULMICORT still considered a first-line treatment for asthma? For mild asthma, inhaled corticosteroids like PULMICORT are generally considered a first-line maintenance therapy. However, for moderate to severe asthma, current treatment guidelines often recommend ICS/LABA combination therapies as a preferred first-line or early-step-up treatment.
-
What is the difference between PULMICORT RESPULES and PULMICORT TURBOHALER? PULMICORT RESPULES is a liquid suspension for nebulization, suitable for infants and young children who may have difficulty using dry powder inhalers. PULMICORT TURBOHALER is a dry powder inhaler device designed for older children and adults who can inhale forcefully.
-
Are there any new patents or intellectual property protecting PULMICORT? The primary patents protecting the budesonide molecule and key delivery devices have expired. AstraZeneca may hold patents on minor formulation improvements or manufacturing processes, but these are unlikely to prevent the widespread availability of generic budesonide.
Citations
[1] Pedersen, S., O'Callaghan, C., Kaad, P. S., et al. (1998). Budesonide in patients with mild to moderate asthma. The New England Journal of Medicine, 339(22), 1605-1611.
More… ↓
