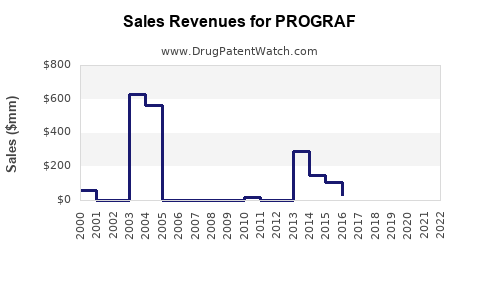
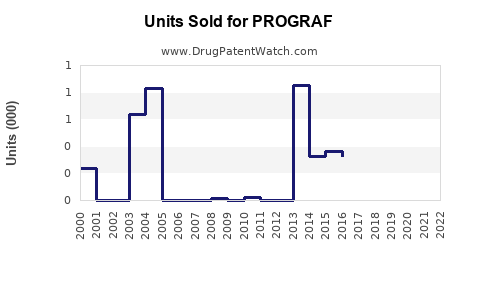
Last updated: February 20, 2026
What is the Current Market Size for PROGRAF?
PROGRAF (Tacrolimus) is an immunosuppressant primarily used to prevent organ rejection in kidney, liver, and heart transplant patients. As of 2022, the global market for transplant immunosuppressants was valued at approximately $4.8 billion, with PROGRAF accounting for about 35% of this segment, equating to roughly $1.68 billion.
Market Segments and Geographic Breakdown
| Region |
Market Share (%) |
Revenue (USD billions) |
Notes |
| North America |
45% |
0.756 |
Dominates due to high transplant activity and reimbursement coverage. |
| Europe |
30% |
0.504 |
Significant market with mature healthcare systems. |
| Asia-Pacific |
20% |
0.336 |
Fast-growth region driven by increasing transplant procedures. |
| Others |
5% |
0.084 |
Including Latin America, Middle East, Africa. |
What Factors Drive Growth in the PROGRAF Market?
- Increasing transplant procedures: Annually, over 40,000 kidney, 10,000 liver, and 2,000 heart transplants occur globally, with a rising trend driven by advancements in surgical techniques and expanded indications.
- Chronic rejection management needs: Tacrolimus remains first-line for preventing organ rejection, maintaining steady demand.
- Market penetration of generics: Generic tacrolimus formulations introduced in late 2010s exert price pressure but have not significantly eroded branded sales due to physician preference for branded PROGRAF.
- Regulatory approvals: New formulations or combination therapies approved for expanded indications could extend market longevity.
What Are the Competitive Dynamics?
| Competitors |
Market Share (%) |
Key Products |
Differentiators |
| Astellas Pharma (PROGRAF) |
80% |
PROGRAF |
Brand recognition, clinical data superiority |
| Generics |
10% |
Tacrolimus formulations from Mylan, Hikma |
Price, availability |
| Other branded drugs |
10% |
Cyclosporine, sirolimus |
Different mechanisms, niche use |
What Are Sales Forecasts for PROGRAF (2023-2030)?
Projection models based on current trends, transplant growth rates, and competitive factors project:
| Year |
Estimated Market Size (USD billions) |
Growth Rate (%) |
Notes |
| 2023 |
2.0 |
- |
Slight market growth amid generic competition |
| 2025 |
2.4 |
12.5% |
Increased transplant volume, new detection methods |
| 2027 |
2.9 |
20.8% |
Potential approval of new formulations or indications |
| 2030 |
3.7 |
22.4% |
Market stabilization, increasing transplant volume |
Key Assumptions
- Transplant procedures increase at 3-4% annually globally.
- No major patent expirations reduce branded sales before 2028.
- Regulatory approvals of combination therapies or new formulations may accelerate sales growth.
- Price erosion from generics is contained due to brand loyalty and physician preference.
Risks and Opportunities
Risks
- Patent expiration could reduce revenue by up to 70% in affected markets.
- Price competition from generics could lower margins.
- Regulatory delays or adverse outcomes may hinder approval of new formulations.
Opportunities
- Expansion into underdeveloped markets with increasing transplant procedures.
- Development of new formulations, including extended-release options.
- Combination regimens improving patient outcomes could increase market share.
Key Takeaways
- The global market for transplant immunosuppressants is approximately $4.8 billion; PROGRAF accounts for roughly $1.68 billion.
- Sales are driven by transplant procedure volume, regional healthcare infrastructure, and branded preference.
- Projected to grow from $2.0 billion in 2023 to around $3.7 billion in 2030.
- Growth is tempered by generic competition but supported by ongoing transplant needs and potential pipeline developments.
FAQs
1. What is the primary indication for PROGRAF?
It prevents organ rejection in kidney, liver, and heart transplant recipients.
2. How does PROGRAF compare price-wise to generics?
Brand PROGRAF typically costs 2-3 times more than generic tacrolimus formulations.
3. What is the patent status of PROGRAF?
Patent protections for PROGRAF expired in major markets between 2017-2018, leading to increased generic competition.
4. Are there new formulations or combinations in development?
Yes, some formulations, including extended-release versions, are under review or marketed in certain regions.
5. Which regions are expected to see the highest growth in PROGRAF sales?
Asia-Pacific and Latin America are expected to experience the fastest growth due to rising transplant procedures and expanding healthcare infrastructure.
References
[1] MarketWatch. (2022). Global immunosuppressant market size.
[2] IMS Health. (2022). Transplant procedures and drug utilization report.
[3] Astellas Pharma. (2022). PROGRAF product information.
[4] World Health Organization. (2022). Organ transplant statistics.
[5] Evaluate Pharma. (2023). Pharmaceutical sales forecasts.