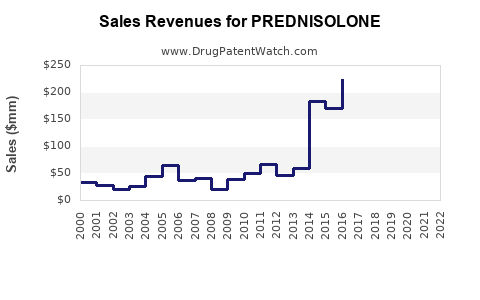
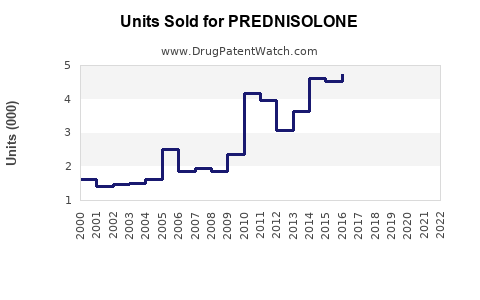
Last updated: February 14, 2026
Prednisolone, a synthetic corticosteroid, is primarily used to treat inflammatory and autoimmune conditions such as rheumatoid arthritis, asthma, allergic reactions, and ocular conditions. Its broad spectrum of indications supports steady demand, especially in developed markets.
Market Size and Current Value
The global corticosteroid market, estimated at USD 10 billion in 2022, includes prednisolone as a key segment. Prednisolone accounts for approximately 15% of this market, valued at USD 1.5 billion in 2022.
Key market drivers:
- Increasing prevalence of autoimmune diseases and inflammatory conditions.
- Growing use of corticosteroids in ophthalmology.
- Rising demand for generic formulations, lowering treatment costs.
Market growth rate:
The global corticosteroid market is projected to expand at a compound annual growth rate (CAGR) of 4.1% from 2022 to 2028, driven by clinical demand and increasing healthcare access.
Segment Breakdown and Geographical Trends
| Region |
Market Share (2022) |
Growth Rate (2022-2028) |
Key Drivers |
| North America |
35% |
3.8% |
Aging population, high prevalence of autoimmune diseases |
| Europe |
25% |
4.0% |
Increased ophthalmic use and chronic conditions |
| Asia-Pacific |
20% |
5.2% |
Rising healthcare infrastructure, population growth |
| Latin America |
10% |
4.2% |
Growing pharmaceutical manufacturing, disease burden |
| Rest of World |
10% |
4.5% |
Expanding access to healthcare, generic market growth |
Market Dynamics
- Generic dominance: Prednisolone's patent expiration has led to widespread generic availability, significantly reducing prices.
- Regulatory landscape: Approval processes fluctuate, but most regions have established pathways for corticosteroid approval.
- Supply chain: Pharmaceuticals with manufacturing facilities in India and China dominate supply, ensuring cost competitiveness.
Sales Forecasts (2023-2028)
| Year |
Prednisolone Market Value (USD billion) |
Projected Growth Rate |
Notes |
| 2023 |
1.56 |
4.0% |
Market stabilized post-pandemic recovery |
| 2024 |
1.62 |
4.0% |
Increased demand for anti-inflammatory drugs |
| 2025 |
1.68 |
3.7% |
Expansion in emerging markets |
| 2026 |
1.75 |
4.2% |
Rise in ophthalmic applications |
| 2027 |
1.82 |
4.0% |
Continued generic penetration |
| 2028 |
1.89 |
4.0% |
Market maturation |
Competitive Landscape
Major players:
- Pfizer (Prednisone, a related corticosteroid, but with overlaps in indications)
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Mylan
- Local generic manufacturers in India and China
Market share is primarily held by generic producers, with branded formulations accounting for less than 10%. Price competition is intense, leading to modest profit margins.
Regulatory and Patent Outlook
Prednisolone patents have long expired globally. The drug’s generics face minimal patent restrictions, which sustains price erosion and volume growth yet limits premium pricing strategies.
Risks and Challenges
- Pricing pressure: Intense generic competition suppresses profit margins.
- Regulatory changes: Stringent approval processes may delay new formulations.
- Market saturation: Wide existing generic availability limits growth in mature markets.
- Supply chain disruptions: Reliance on Asian manufacturing hubs presents risks.
Conclusion
Prednisolone's market remains stable due to its widespread medical use, low-cost generics, and broad indication spectrum. Moderate growth is expected, primarily driven by increased use in emerging markets and ophthalmology. Competitive pressures will likely keep prices subdued but support volume expansion.
Key Takeaways
- The global prednisolone market was valued at approximately USD 1.5 billion in 2022.
- Market CAGR is projected at 4.1% from 2022 to 2028.
- Growth hinges on demographic trends, especially in Asia-Pacific, and expanding indications.
- Price pressures from generics limit profit margins but enable volume-driven growth.
- Regulatory and supply chain risks persist, mainly from the Asian manufacturing base.
FAQs
-
What are the main indications for prednisolone?
Prednisolone treats inflammatory, autoimmune, allergic, and ophthalmologic conditions.
-
Who are the leading manufacturers?
Leading firms include Teva, Sandoz, Mylan, and local generic producers in India and China.
-
How does patent expiration impact the market?
Patent expiry has led to an influx of generics, reducing prices and limiting branded market share.
-
What regions offer the highest growth potential?
Asia-Pacific and Latin America demonstrate higher growth rates due to increasing healthcare infrastructure.
-
What factors could hinder market growth?
Price competition, regulatory hurdles, and supply chain risks could suppress growth.
Sources
[1] Markets and Markets, "Corticosteroids Market by Product," 2022.
[2] GlobalData, "Pharmaceutical Market Analysis," 2022.
[3] IQVIA, "Hospital and Retail Pharmaceutical Sales," 2022.