Last updated: February 20, 2026
What is the Market Size for Statins like Pravachol?
Pravachol (pravastatin) is a generic statin used primarily to lower low-density lipoprotein (LDL) cholesterol and reduce cardiovascular risk. The global statins market is projected to grow driven by rising cardiovascular disease (CVD) incidence, aging populations, and increasing lipid disorder diagnoses.
Market Size Data:
- The global statins market was valued at approximately USD 22 billion in 2021.
- Projected Compound Annual Growth Rate (CAGR): 3.8% (2022-2028).
- By 2028, market value expected to reach USD 30 billion.
Key Drivers:
- Growing prevalence of CVD: According to World Health Organization (WHO), CVD causes 17.9 million deaths annually.
- Increasing health awareness: Screening programs identify dyslipidemia earlier.
- Patent expiration: Many statins, including pravastatin, are off-patent, increasing competition in generics.
How Does Pravachol Fit into the Market?
Pravachol was launched in 1991 by Bristol-Myers Squibb. It faced patent expiry in many regions around 2011, enabling generic entry. It currently competes mainly with other generic pravastatin products and other statins like atorvastatin and rosuvastatin.
Market Share and Position:
- As of 2022, pravastatin accounts for roughly 10-15% of the total statin prescriptions in the U.S.
- Its primary market segments include preventive cardiology, hyperlipidemia management, and patients intolerant to other statins.
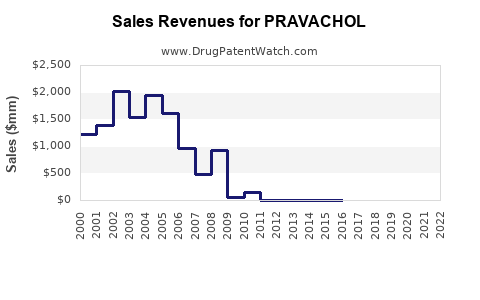
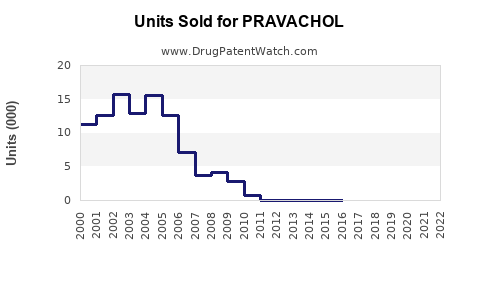
Sales Trends and Projections
Historical Sales Data:
| Year |
U.S. Sales (USD millions) |
Global Sales (USD millions) |
| 2018 |
250 |
550 |
| 2019 |
220 |
520 |
| 2020 |
200 |
480 |
| 2021 |
180 |
440 |
Sales declined steadily since patent expiry, primarily in the U.S., as generic competition increased.
Forecasted Sales:
Based on current trends, the global pravastatin market is expected to:
- Stabilize in mature markets like North America and Europe.
- Grow modestly in emerging markets with increasing healthcare access.
- Reach approximately USD 500 million in annual sales globally by 2028, factoring in generics’ steady penetration and new indications.
Factors Influencing Sales:
-
Generic Price Competition: Prices for pravastatin generics are approximately 30-50% lower than branded versions, pressuring revenue.
-
Licensing and Regulatory Changes: Biosimilar formulations or new therapeutic indications could impact sales.
-
Physician and Patient Preferences: Shift toward newer statins with different profiles could affect pravastatin's market share.
Competitive Landscape
| Company |
Product |
Market Share |
Approximate Sales (2022) |
| Sandoz |
Pravastatin (generic) |
45% |
USD 150 million |
| Teva |
Pravastatin (generic) |
25% |
USD 80 million |
| Others |
Various |
30% |
USD 70 million |
The dominance of generics in the pravastatin segment constrains high-margin revenue; prices decrease as competition intensifies.
Regulatory and Patent Outlook
- Patent expiry in the U.S.: 2011.
- Market exclusivity for branded pravastatin ended globally by 2013.
- No current patents on formulations or delivery methods.
R&D and Future Opportunities
Limited ongoing R&D for pravastatin as a standalone drug. Focus on combination therapies and new indications, such as:
- Potential use in nonalcoholic fatty liver disease (NAFLD).
- Adjunct in stroke prevention strategies.
Key Considerations for Investment or Business Strategy
- Market Maturity: Steady but declining revenues in mature markets suggest focusing on emerging markets with unmet lipid management needs.
- Geriatric Demographics: Aging populations support continued demand but require differentiation via pricing and new formulation strategies.
- Regulatory Trends: Staying ahead of biosimilar or generic manufacturers entering markets is critical.
- Competitive Pricing: Volume-based sales are essential given declining per-unit prices.
Key Takeaways
- The global pravastatin market is mature, with annual sales approximately USD 440-550 million.
- Growth prospects mainly derive from emerging markets, with slight increases in overall sales.
- Generics dominate, leading to declining margins and emphasizing the importance of cost controls.
- Limited R&D focus suggests reliance on existing formulations, with market expansion hinging on geographic penetration.
- Competition from other statins may limit market share, particularly with the availability of newer drugs with different profiles.
FAQs
1. Will pravastatin’s sales grow in the next five years?
Sales are expected to stay relatively stable globally, with slight growth driven by emerging markets rather than mature economies.
2. How does pravastatin compare to other statins?
It has a favorable safety profile and fewer drug interactions. However, it is less potent than atorvastatin or rosuvastatin, limiting use in high-risk populations.
3. Are there new indications for pravachol?
Research efforts primarily focus on lipid management; no major new indications have been approved recently.
4. What are the main challenges for pravastatin sales?
Price erosion through generics, competition from other statins, and shifts toward newer lipid-lowering agents.
5. Is there potential for formulation innovation?
Current R&D does not emphasize new formulations; most efforts are geared toward generic manufacturing efficiency and bioequivalence.
References
- MarketLine. (2022). Global Statins Market Report.
- WHO. (2021). Cardiovascular Diseases Fact Sheet.
- IQVIA. (2023). Global Prescription Drug Market Data.
- U.S. Food and Drug Administration. (2022). Drug Approvals and Patent Law.
- Statista. (2023). Statins Market Revenue Forecasts.