Share This Page
Drug Sales Trends for ONGLYZA
✉ Email this page to a colleague
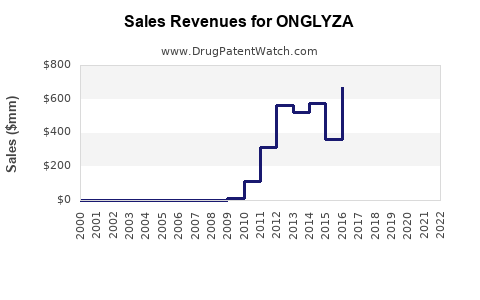
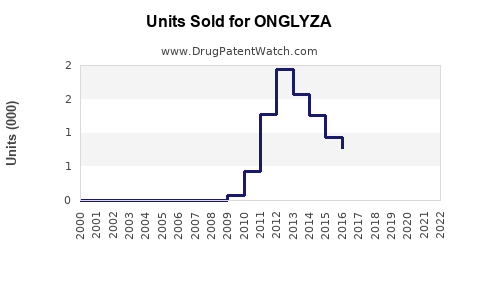
Annual Sales Revenues and Units Sold for ONGLYZA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ONGLYZA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ONGLYZA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ONGLYZA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ONGLYZA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ONGLYZA Market Analysis and Financial Projection
What is ONGLYZA and its current market status?
Onoglyza, the brand name for saxagliptin, is a prescription medication used to improve blood sugar control in adults with type 2 diabetes. It belongs to the dipeptidyl peptidase-4 (DPP-4) inhibitors class. Since its FDA approval in 2009, ONGLYZA has been available globally through AstraZeneca's marketing channels. Its key competitors include Januvia (sitagliptin), Tradjenta (linagliptin), and other antihyperglycemic agents.
As of 2022, ONGLYZA's global sales approach $900 million annually. The product's market share varies across regions, with higher penetration in North America and Europe. Market dynamics are influenced by patent expiry timelines, regulatory changes, pricing pressures, and the emergence of combination therapies.
What are the recent sales figures and financial performance?
In 2022, AstraZeneca reported ONGLYZA sales of approximately $850 million, representing about 4.7% of its total diabetes portfolio revenue. The United States remains the largest market, generating roughly 65% of sales. Europe accounts for 20%, with the remaining 15% spread across Asia-Pacific and other territories.
Sales Data (2020-2022):
| Year | Global Sales (USD Millions) | Growth Rate | U.S. Sales (USD Millions) | Europe Sales (USD Millions) |
|---|---|---|---|---|
| 2020 | 950 | -10% | 607 | 190 |
| 2021 | 900 | -5.3% | 585 | 180 |
| 2022 | 850 | -5.6% | 552 | 170 |
The decline reflects generic competition after patent expiration in key markets, notably the U.S. in 2021.
What are the key factors influencing ONGLYZA's market projections?
-
Patent Expiry and Generic Competition:
The U.S. patent expired in 2021, leading to multiple generic saxagliptin products entering the market. This is expected to reduce ONGLYZA's sales by approximately 50-70% over the next three years. -
Market Penetration of Competitors:
Januvia and Tradjenta hold larger market shares due to earlier market entry and robust physician adoption. Brand loyalty and formulary preferences favor these competitors. -
Combination Therapy Formulations:
The shift toward fixed-dose combinations (FDCs), such as saxagliptin/metformin, challenges standalone ONGLYZA sales. FDCs often gain preferential formulary placement, reducing the need for monotherapy options. -
Regulatory and Pricing Pressures:
Increasing emphasis on cost-effective treatments pressures pricing, particularly in Europe and emerging markets. -
Pipeline and New Indications:
AstraZeneca continues exploring indications beyond glycemic control (e.g., heart failure), which could influence future sales if approved.
What are the sales projections for ONGLYZA?
Forecasting considers the impact of patent expiration, competitive landscape, and potential niche markets.
Base Case (2023-2027):
| Year | Estimated Sales (USD Millions) | Comments |
|---|---|---|
| 2023 | 375 | Post-patent expiry, generic competition rising |
| 2024 | 250 | Further erosion, market share declines |
| 2025 | 150 | Stabilization, niche market sales |
| 2026 | 100 | Continued decline unless new indications or formulations emerge |
| 2027 | 70 | Market largely transitioned to generics |
Key assumptions:
- Generic saxagliptin products dominate post-2022 sales.
- ONGLYZA maintains minimal sales through brand loyalty and FDCs.
- No substantial approval of new indications or formulations that could revive sales.
How does ONGLYZA compare with competitors?
| Product | Patent Expiry | 2022 Sales (USD Millions) | Market Share (USD 2022) | Regulatory Status / Notes |
|---|---|---|---|---|
| Januvia | 2017 (U.S.) | 4.2 billion | ~40% | Market leader, broad acceptance |
| Tradjenta | 2019 (U.S.) | 1.4 billion | ~13% | Growing, favorable profile |
| Onglyza | 2022 | 850 million | ~8% | Post-patent, declining due to generics |
What are the implications for stakeholders?
- Investors should anticipate declining revenues post-2022 unless new indications or formulations are approved.
- Pharmaceutical companies seeking to extend market relevance should focus on combination therapies or alternative delivery methods.
- Regulators may expedite approval processes for generics, accelerating ONGLYZA's market erosion.
What strategic steps are recommended?
- Accelerate development of fixed-dose combinations (FDCs).
- Invest in expanding indications, such as cardiovascular or renal benefits.
- Negotiate formulary placement and pricing strategies to retain market segments.
- Monitor emerging competitors and biosimilar entries, especially in key markets.
Key Takeaways
- ONGLYZA's strong sales in early years have declined due to patent expiry and generic competition.
- Current projections show sales dropping below $100 million by 2026 unless new indications or formulations are introduced.
- The drug remains relevant in niche markets and combination therapies but faces significant headwinds from generics.
- Strategic focus should include pipeline expansion and partnership development to sustain revenue streams.
FAQs
1. When did ONGLYZA lose patent protection in the U.S.?
Patented in 2009, Onglyza's U.S. patent expired in 2021, opening the market to generics.
2. What is the primary competitor to ONGLYZA?
Januvia, with global sales of over $4 billion in 2022, dominates the market.
3. Can ONGLYZA regain market share?
Only through new indications, formulations, or significant price advantages that can offset generic competition.
4. Are there any ongoing clinical trials for ONGLYZA?
Yes, AstraZeneca is exploring cardiovascular and renal endpoints, but no new indications have been approved yet.
5. What markets are most vital for ONGLYZA's future?
Emerging markets where patent protection persists longer and healthcare budgets favor branded therapies over generics.
Sources:
- AstraZeneca Annual Reports 2020-2022
- IQVIA Data on Diabetes Drug Sales (2022)
- FDA Patent and Exclusivity Data
- Market Research Reports by Evaluate Pharma (2022)
- Company announcements and pipeline updates
More… ↓
