Last updated: February 16, 2026
What Is LANOXIN and How Is It Used?
Lanoxin, with the generic name digoxin, is a cardiac glycoside approved primarily for heart failure, atrial fibrillation, and atrial flutter. It enhances cardiac contractility and controls heart rate. The drug has been on the market since the 1950s, with a well-established safety and efficacy profile.
Market Overview
Global Sales and Market Size
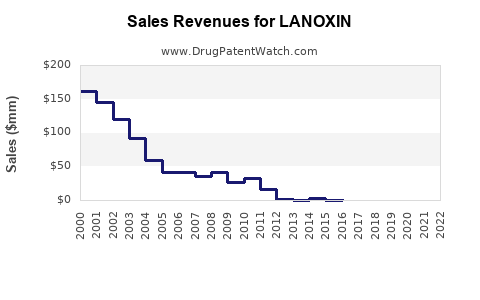
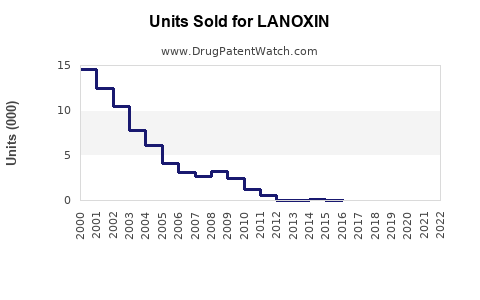
The global digoxin market was valued at approximately $200 million in 2022. It primarily supplies chronic heart failure and arrhythmia treatments. The market has experienced slow growth due to a declining preference for digoxin in favor of newer medications.
Regional Breakdown
- North America: 55% of sales, driven by high prevalence of cardiovascular diseases and established prescribing habits.
- Europe: 30%, with sustained demand but increasing competition from novel agents.
- Asia-Pacific: 10%, with modest demand growth owing to speed of adoption and regulatory differences.
- Rest of World: 5%
Market Trends
The use of digoxin is decreasing due to the availability of newer drugs like beta-blockers, ACE inhibitors, and amiodarone. However, digoxin remains relevant in specific cases where alternatives are contraindicated or ineffective.
Competitive Landscape
Major Players
- Pfizer (brand: Lanoxin)
- Teva Pharmaceuticals
-KT Corporation (generic formulations)
Alternatives and Competition
While digoxin maintains a niche, drugs such as amiodarone and sotalol are used for arrhythmia management. For heart failure, newer agents like sacubitril/valsartan (Entresto) are preferred.
Sales Projections
Short-Term Outlook (Next 2 Years)
Projected sales are expected to decline at an annual rate of approximately 3-5%. This decline stems from reduced prescribing and regulatory constraints on old-generation drugs.
| Year |
Estimated Market Size (USD Millions) |
Growth Rate |
| 2023 |
190 |
-5% |
| 2024 |
180-185 |
-3% to -5% |
Long-Term Outlook (Next 5–10 Years)
The market might stabilize at around $150 million due to its niche application but will continue to decline due to replacements by newer therapies and patent expirations in some regions.
Factors Influencing Sales
- Patent status: No patent protections remaining, leading to generic competition.
- Regulatory policies: Reimbursement policies favoring newer drugs.
- Prescribing habits: Shifts toward newer therapies and away from digoxin.
Regulatory Environment
No recent major regulatory changes impact digoxin. However, ongoing reassessment of safety profiles might influence prescribing in some regions. FDA warnings in 2010 highlighted toxicity risks, leading to more conservative use.
Key Takeaways
- The global digoxin market was valued at approximately $200 million in 2022.
- Sales are declining at about 3-5% annually due to competition from newer therapies.
- North America remains the largest market, followed by Europe.
- Long-term, the market is expected to contract further with driven decline, reaching approximately $150 million in 5–10 years.
- The drug’s niche use and established safety profile preserve limited demand despite overall market contraction.
FAQs
Q1. What factors contribute to the decline in digoxin sales?
The decline results from the availability of more effective, safer, and easier-to-manage medications, along with changing clinical guidelines that favor newer therapies.
Q2. Are there upcoming patent protections or formulations that could influence sales?
No, generic manufacturing dominates, eroding patent protections and significantly reducing prices, which further diminishes sales.
Q3. What regions are expected to experience the most significant decline?
Developed regions, particularly North America and Europe, will see the most decline due to regulatory pressures and increased adoption of alternative treatments.
Q4. How does digoxin compare with newer heart failure drugs?
Newer drugs like sacubitril/valsartan provide superior mortality benefits and safety margins compared to digoxin but are more costly.
Q5. Will genericization impact the profitability of existing manufacturers?
Yes, generic competition leads to price erosion and reduced profit margins for manufacturers of digoxin.
Sources
- Market research reports (e.g., GlobalData, MarketsandMarkets)
- US FDA prescribing information and warnings
- Scholarly articles on cardiac drug management and market trends