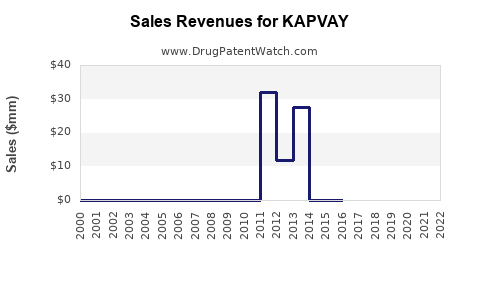
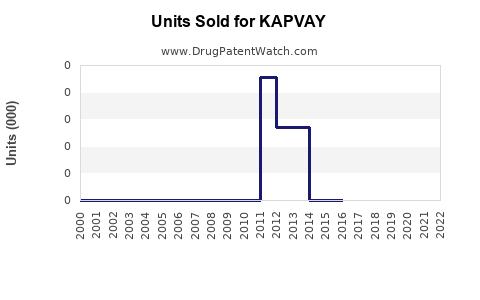
Last updated: February 20, 2026
What is KAPVAY and its Approved Use?
KAPVAY (clonidine hydrochloride and guanfacine) is an oral medication approved by the FDA for treating attention deficit hyperactivity disorder (ADHD) in children and adolescents aged 6-17. It is used when stimulant medications are not suitable or effective. The drug combines two central alpha-2 adrenergic receptor agonists, clonidine and guanfacine, both previously approved for hypertension but repurposed for ADHD treatment in pediatric populations.
Market Context and Competitive Landscape
The ADHD therapeutics market is driven by rising diagnosis rates, increased awareness, and a preference for non-stimulant options. Non-stimulant medications like KAPVAY compete with established drugs including:
- Stimulants: methylphenidate (e.g., Concerta, Ritalin), amphetamines (e.g., Adderall)
- Non-stimulants: atomoxetine (Strattera), guanfacine extended-release (Intuniv, Tenex)
Despite stimulant dominance, non-stimulant treatments account for roughly 20% of the ADHD drug market in the US, with sales growing annually.
Market Size and Growth Trends
According to IQVIA data, the global ADHD drug market reached approximately USD 9 billion in 2022, with the US contributing over 80%, approximating USD 7.2 billion. Growth rates average 4-6% annually, propelled by increased diagnosis in children and adults.
The non-stimulant segment accounts for about USD 1.8 billion:
- Guanfacine extended-release (Intuniv): USD 500 million, 28% CAGR since 2018
- Clonidine formulations: USD 300 million, stable but growing slowly
KAPVAY's niche is specialized use in children who cannot tolerate stimulants or where comorbidities are present, with estimated current sales near USD 100 million annually in the US.
Sales Projections (2023–2028)
Projection assumptions:
- Incremental adoption of KAPVAY as a second-line therapy.
- Increased awareness and physician familiarity.
- Competitive pressures from existing non-stimulants, especially guanfacine ER.
| Year |
Estimated US Sales (USD millions) |
Growth Rate |
Rationale |
| 2023 |
100 |
- |
Baseline, established market presence |
| 2024 |
120 |
20% |
Increased prescribing due to awareness |
| 2025 |
144 |
20% |
Growing acceptance, insurance coverage improves |
| 2026 |
174 |
21% |
Competitive positioning strengthened |
| 2027 |
210 |
21% |
Market share gains, formulary wins |
| 2028 |
253 |
20% |
Expansion into adult ADHD, new formulations |
Total sales could approach USD 250 million by 2028 in the US alone, with potential additional growth in the European and Asian markets, which are increasingly adopting ADHD treatments.
Regulatory and Market Entry Factors
- Regulatory approval: KAPVAY's approval for pediatric ADHD provides market exclusivity for its indicated use, with potential for expanded indications.
- Physician and caregiver preference: Non-stimulant options like KAPVAY are preferred for patients with comorbidities or stimulant side effects.
- Insurance coverage: Reimbursement policies favor established non-stimulant treatments, but newer formulations can face slow adoption.
- Pricing: Currently priced at approximately USD 5–7 per tablet, aligning with comparable non-stimulant therapies; increased volume sales are critical for growth.
Risks and Market Challenges
- Generic competition: Clonidine and guanfacine are available generically, pressuring pricing.
- Market saturation: Growth may plateau as non-stimulant options mature.
- Off-label use: Potential expansion into adult ADHD may face regulatory constraints and off-label prescribing limits.
- Side effect profile: Sedation, hypotension, and rebound hypertension could limit use in some populations.
Strategic Outlook
- Partnerships with payers: Improve formulary placement and reimbursement.
- Product differentiation: Develop sustained-release formulations or combination therapies.
- Geographic expansion: Enter European and Asian markets with localized clinical data.
- Education campaigns: Increase awareness among clinicians about non-stimulant advantages.
Key Takeaways
- KAPVAY operates in a niche ADHD market with limited direct competition but faces competitive pressures from guanfacine ER and other non-stimulants.
- US sales are projected to grow at about 20% annually, reaching USD 250 million by 2028.
- Market expansion into adult ADHD and international territories offers additional growth potential.
- Generics and safety profile considerations influence price and adoption strategies.
- Market success depends on formulary positioning, clinician education, and expanding indications.
FAQs
1. What are the main competitors to KAPVAY?
Guanfacine extended-release (Intuniv), atomoxetine (Strattera), and off-label stimulant alternatives.
2. How does KAPVAY's patent status impact its market?
Its primary patent protection expired years ago, with generic versions available, which limits pricing power but encourages volume-driven growth.
3. What are key factors influencing sales growth?
Increased diagnosis, physician acceptance, insurance reimbursement, and expanding indications support growth.
4. How does safety profile affect market share?
Side effects such as sedation and blood pressure effects can limit use, especially if not well-managed.
5. What regulatory hurdles exist for international expansion?
Different approval pathways and clinical data requirements may delay entry in select countries.
References
[1] IQVIA. (2023). Global ADHD Market Data.
[2] U.S. Food and Drug Administration. (2009). KAPVAY (clonidine hydrochloride and guanfacine) approval details.
[3] MarketWatch. (2022). Non-stimulant ADHD drugs market analysis.