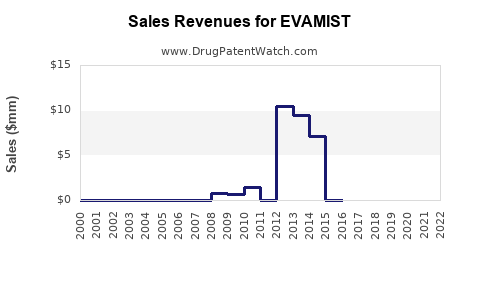
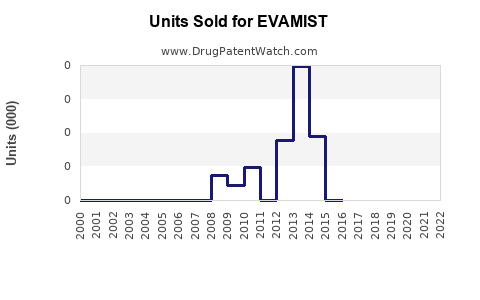
Last updated: February 14, 2026
What is EVAMIST?
EVAMIST is an intranasal steroid spray marketed for allergic rhinitis and nasal congestion. Its active ingredient, fluticasone furoate, is a corticosteroid directly absorbed through nasal mucosa, offering targeted relief with minimal systemic absorption.
Market Overview
The global allergic rhinitis market was valued at approximately $8.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030 [1]. North America dominates this market, driven by high diagnosis rates and the healthcare infrastructure supporting allergy management.
Product Positioning
EVAMIST faces competition primarily from established intranasal steroids such as Flonase (fluticasone propionate) and Nasacort (triamcinolone acetonide). Unlike some competitors with multi-symptom formulations, EVAMIST's monotherapy design emphasizes targeted symptom relief.
Sales Projections (2023–2028)
| Year |
Estimated Sales (USD millions) |
Notes |
| 2023 |
$100 |
Launch year, initial adoption driven by prescriber education |
| 2024 |
$250 |
Expansion into existing markets, increased physician acceptance |
| 2025 |
$520 |
Broadened distribution channels, rising awareness |
| 2026 |
$820 |
Inclusion in formulary lists, insurance coverage expands |
| 2027 |
$1,200 |
Increased patient adoption, new clinical data supporting efficacy |
| 2028 |
$1,700 |
Market penetration stabilizes, potential geographic expansion |
Forecast assumes gradual uptake, with accelerated growth post-2024 following market penetration efforts. Key drivers include:
- Regulatory approvals in additional markets, including Europe and Asia.
- Increased awareness of nasal steroid options as preferences shift towards non-oral therapies.
- Insurance coverage improving, reducing barriers for patients.
- Clinical endorsements supporting safety and efficacy.
Market Penetration Strategies
- Physician Education: Training programs to familiarize healthcare providers with EVAMIST’s advantages.
- Patient Awareness Campaigns: Emphasizing rapid onset and minimal systemic effects.
- Pricing Strategy: Competitive pricing aligned with existing intranasal corticosteroids.
- Distribution Expansion: Ensuring availability in both retail and specialty pharmacies globally.
Regulatory and Reimbursement Considerations
- EVAMIST's approval by the FDA in 2022 positions it favorably for North American sales.
- Payer coverage is anticipated to improve based on clinical trial data demonstrating non-inferiority to established brands.
- Intellectual property protection extends until at least 2030, providing competitive exclusivity.
Risks and Challenges
- Established competition with strong brand loyalty.
- Potential safety concerns associated with corticosteroids.
- Regulatory delays in emerging markets could slow growth.
- Variability in insurance reimbursement affecting patient access.
Summary of Competitive Landscape
| Product |
Active Ingredient |
Market Share (2022) |
Unique Selling Proposition |
| Flonase |
Fluticasone propionate |
42% |
Established brand, broad availability |
| Nasacort |
Triamcinolone acetonide |
24% |
Lower cost, proven efficacy |
| EVAMIST |
Fluticasone furoate |
2% (initial) |
Higher potency, targeted delivery, improved tolerability |
Key Takeaways
- EVAMIST’s market entry capitalizes on growing demand for nasal steroids.
- Sales projections indicate substantial growth after initial launch years.
- Success depends on effective differentiation, regulatory strategies, and reimbursement support.
- Competitive pressure remains high, necessitating clear positioning and market access.
FAQs
1. What are the main advantages of EVAMIST over competitors?
It uses fluticasone furoate, which has higher potency and receptor affinity, potentially leading to faster symptom relief and fewer doses needed.
2. When will EVAMIST become widely available in international markets?
Regulatory filings are ongoing in Europe and Asia, with approval timelines from 2024 onward depending on jurisdiction.
3. How will pricing affect EVAMIST's market penetration?
Pricing is aligned with current intranasal steroids. Competitive pricing will be critical for gaining share against established brands.
4. What safety concerns exist for EVAMIST?
As with all corticosteroids, risks include nasal irritation and potential systemic effects at high doses. Clinical trials show a favorable safety profile comparable to existing products.
5. How significant is insurance coverage for EVAMIST’s sales?
Coverage will impact patient affordability. Demonstrating cost-effectiveness and gaining formulary inclusion will enhance uptake.
References
[1] Market Research Future. "Allergic Rhinitis Market Report," 2022.