Last updated: March 22, 2026
What Is COZAAR's Current Market Position?
COZAAR (losartan potassium) is an angiotensin II receptor blocker (ARB) approved for managing hypertension and reducing the risk of stroke. It is marketed by Merck & Co., with approval dating back to 1995. The drug targets a broad patient population, including those with hypertension, diabetic nephropathy, and high cardiovascular risk.
Market penetration is well-established in developed economies, with significant presence in the U.S., Europe, and Japan. Its main competitors are other ARBs such as Valsartan (Diovan) and Telmisartan (Micardis).
What Are the Key Market Dynamics Affecting COZAAR?
-
Prevalence of Hypertension: Estimated 1.28 billion adults globally have hypertension (WHO, 2021). The global hypertension market was valued at approximately USD 15 billion in 2020, expected to grow at a CAGR of 3–5% through 2027[1].
-
Generic Competition: Patent expiration in many markets has increased generic entry, reducing COZAAR's market share and pricing power. Generic losartan is available in most regions since around 2010.
-
Prescribing Trends: Shift toward combination therapies (e.g., losartan with hydrochlorothiazide) impacts sales volume. Growing focus on cardiovascular risk reduction influences prescribing behaviors.
-
Regulatory and Market Access Factors: Reimbursement policies and formulary inclusion affect drug utilization. COZAAR’s approval in numerous countries limits market entry barriers but competition remains rigorous.
What Are the Sales Trends for COZAAR?
-
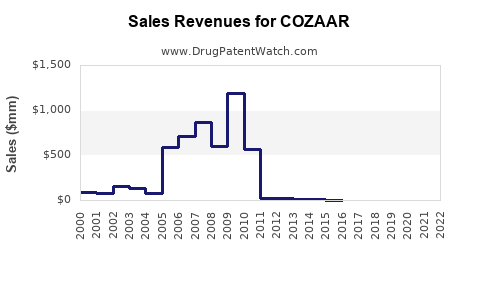
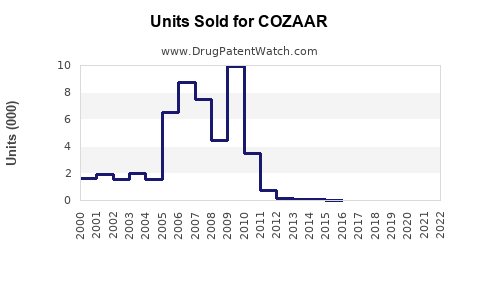
Historical Sales Data: In 2020, Merck reported approximately USD 830 million in sales globally for COZAAR[2].
-
Sales Decline Trend: Since 2010, global sales have declined from a peak of over USD 1.2 billion, driven by patent expirations and generic competition.
-
Geographical Distribution: U.S. sales account for a significant portion (~50%) of total revenue, followed by Europe (30%) and other markets (20%).
What Are Projected Sales Figures?
Short-term (Next 3 Years)
-
Assumptions: Continued generic competition, moderate growth in hypertensive and cardiovascular populations, and increased use of fixed-dose combinations.
-
Forecast: Estimated USD 600–700 million annually by 2025, reflecting a compound annual decline rate (CADR) of approximately 4–6% from 2020 levels.
Mid to Long-term (Next 5–10 Years)
Potential Impact of Pandemic and Healthcare Trends
-
Disruptions in healthcare access and delays in diagnosis might temporarily suppress demand.
-
Growth in telemedicine and electronic health record-driven prescribing could influence medication management patterns.
Competitive Landscape
| Drug Name |
Class |
Patent Status |
2020 Global Sales |
Key Markets |
| Valsartan |
ARB |
Patent expired |
Over USD 4B |
U.S., Europe |
| Telmisartan |
ARB |
Patent expired |
USD 1.8B |
U.S., Europe |
| Olmesartan |
ARB |
Patent expired |
USD 620M |
U.S., Europe |
Strategic Considerations for Stakeholders
-
Engage in market share retention via medication adherence programs.
-
Invest in developing fixed-dose combinations to enhance patient compliance.
-
Consider early adoption strategies for next-generation ARBs or formulations.
Key Takeaways
-
COZAAR’s global sales peaked in 2010, then declined due to patent expiry and generics.
-
2020 sales approximately USD 830 million; projected to decline further to USD 600–700 million by 2025.
-
Universal hypertension prevalence sustains demand, but increased competition pressures pricing and market share.
-
Future growth depends on innovation, market access, and shifting treatment paradigms.
FAQs
1. How does COZAAR compare to its competitors in sales and market share?
COZAAR's global sales are lower than Valsartan, which surpasses USD 4 billion annually. It maintains a significant share in its market segment but faces aggressive competition from generic ARBs.
2. What factors threaten COZAAR’s market position?
Patent expiry, price erosion from generics, and emergence of new combination therapies diminish its market share.
3. Are there new indications or formulations in development for COZAAR?
There are no publicly disclosed new indications or formulations currently under development for COZAAR.
4. How has COVID-19 impacted COZAAR sales?
Disruptions in healthcare services and diagnosis may temporarily suppress sales, though long-term impacts are uncertain.
5. Will COZAAR regain market share through new innovations?
unlikely without new formulations, indication approvals, or significant pricing advantages, given the saturation with generics.
Citations
[1] World Health Organization. (2021). Hypertension. https://www.who.int/news-room/fact-sheets/detail/hypertension
[2] Merck & Co. (2020). Annual Report.