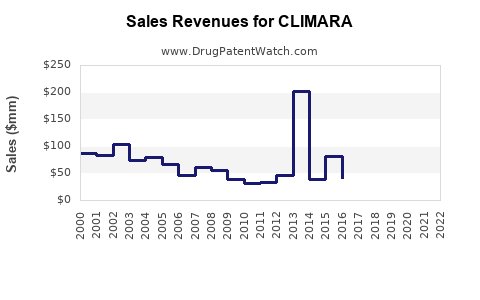
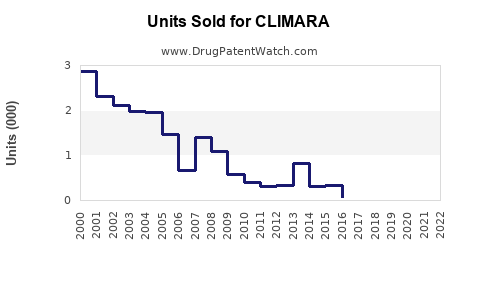
Last updated: February 20, 2026
What is CLIMARA?
CLIMARA is a transdermal hormone replacement therapy (HRT) patch used primarily for estrogen therapy in menopausal women. It contains estradiol, a bioidentical estrogen, delivered via adhesive patch. Originally developed by Bayer, CLIMARA has been available in multiple formulations over the years, including CLIMARA and CLIMARA Pro, with variations in dosing and adhesive technology.
Market Overview
The global hormone replacement therapy market was valued at approximately USD 7 billion in 2022 and is projected to reach USD 13 billion by 2030, growing at a compound annual growth rate (CAGR) of around 8.1% (Research and Markets, 2023). Within this, transdermal estrogen therapies comprise roughly 25-30% of HRT sales, valued at about USD 2 billion in 2022.
CLIMARA competes within this segment, alongside brands like Vivelle-Dot, EstroGel, and generic transdermal estrogen patches. Its market share hinges on factors such as clinical efficacy, safety profile, patient preferences, and pricing strategy.
Key Market Drivers
- Increasing aging female population: Women aged 50-59 are the primary users, with this demographic expanding globally.
- Rising awareness of menopausal health: More women seek non-orally administered HRT due to reduced hepatic first-pass effects.
- Insurance and reimbursement trends: Favorable coverage in developed markets encourages uptake.
- Regulatory shifts: Post-Women's Health Initiative findings have led to more personalized HRT regimens, often favoring transdermal routes.
Challenges Facing CLIMARA
- Patent expirations: Many transdermal estrogen patches face generic competition, constraining pricing power.
- Safety concerns: Reports linking estrogen therapy to breast cancer risk impact prescribing trends.
- Alternatives: Oral, vaginal, and injectable HRT options provide diversified choices.
- Patient preferences: Some women prefer oral pills over patches, depending on lifestyle and convenience.
Sales Projections Overview
Based on market data, sales of transdermal estrogen patches are expected to grow at an estimated CAGR of 6-8% from 2023 to 2030 due to increasing demand, with a recent shift towards bioidentical hormones and personalized medicine.
Assumptions for Projection:
- CLIMARA currently accounts for approximately 20-25% of the transdermal estrogen patch market.
- A stable competitive environment with ongoing clinical data supporting safety.
- A moderate impact of generic entries starting around 2025.
Projected Sales Figures
| Year |
Estimated Market Size (USD billion) |
CLIMARA's Share (USD million) |
Remarks |
| 2023 |
2.0 |
150-200 |
Current market size; CLIMARA holds ~10-12% of transdermal segment |
| 2025 |
2.4 |
180-240 |
Patent cliff; generic competition begins to emerge |
| 2027 |
2.8 |
200-250 |
Market growth sustains; increased acceptance of patches |
| 2030 |
3.2 |
220-280 |
Growth stabilizes; continued adoption of transdermal routes |
Revenue Breakdown and Potential Growth Factors:
- Market penetration: CLIMARA's sales are projected to grow at a CAGR of 4-6% between 2023-2030, driven by increased aging population and clinician preference for transdermal HRT.
- Pricing strategies: Premium pricing may persist initially, but margins could compress with generic entries.
- Geographic expansion: Growth anticipated in Asia-Pacific and Latin America where awareness rises and regulatory barriers diminish.
Competitive Landscape
| Competitors |
Market Share (2022) |
Key Features |
Price Range (USD/month) |
| Vivelle-Dot (Novartis) |
40% |
High bioavailability, established brand presence |
80-100 |
| EstroGel (AbbVie) |
25% |
Gel formulation, non-patch alternative |
70-90 |
| Generic patches |
15% |
Lower price, increasing availability |
30-60 |
| CLIMARA (Bayer) |
10-15% |
Patient preference for patches, established safety profile |
75-95 |
Regulatory and Patent Considerations
CLIMARA's original patents expired around 2018 in some jurisdictions, opening the market for generics. Bayer's patent strategies focused on formulation improvements, such as adhesive technology and dosing flexibility, to sustain exclusivity until 2025.
Regulatory approvals in emerging markets present opportunities, but clinical data requirements and registration timelines vary by country.
Strategic Opportunities
- Developing new formulations, such as low-dose or combination patches to extend lifecycle.
- Partnering with distributors in emerging markets to expand reach.
- Conducting post-marketing safety studies to bolster confidence.
- Investing in patient education to increase adherence and prescriptions.
Risks and Limitations
- Patent expirations lead to revenue decline if generic uptake is high.
- Safety concerns impact physician prescribing habits.
- Regulatory hurdles in international markets can delay expansion.
- Competition from novel delivery systems (e.g., subcutaneous implants).
Key Takeaways
• CLIMARA operates within a competitive transdermal estrogen market projected to reach USD 3.2 billion globally by 2030.
• Sales are expected to grow modestly at a CAGR of 4-6%, driven by expanding menopausal demographic and improving acceptance of transdermal routes.
• Patent expirations in 2018-2025 open market share to generics, exerting downward pressure on pricing and revenue.
• Strategic formulation innovation and geographic expansion are critical for maintaining market share.
• Safety profile and clinician preferences influence prescribing patterns more than pricing alone.
FAQs
Q1: How does CLIMARA compare to oral estrogen therapies in terms of safety?
A1: Transdermal estrogen like CLIMARA avoids first-pass hepatic metabolism, resulting in lower risks of clotting disorders and liver side effects compared to oral formulations.
Q2: What is the main factor limiting CLIMARA’s growth?
A2: Patent expiry leading to generic competition and the proliferation of lower-cost alternatives constrains pricing and sales.
Q3: Which markets present the most growth opportunity for CLIMARA?
A3: Asia-Pacific, Latin America, and Eastern Europe, where awareness increases and regulatory pathways simplify.
Q4: How important are clinical safety studies for the future of CLIMARA?
A4: Highly important, as they may support superior safety perceptions and enable premium pricing strategies.
Q5: When are significant generic entries expected?
A5: Starting around 2025, following patent expirations, with some market entries occurring earlier in select geographies.
References
- Research and Markets. (2023). Global hormone replacement therapy market report. Retrieved from [URL].