Last updated: February 14, 2026
What Is CARAFATE and Its Market Position?
CARAFATE (sodium alginate) is a gastric protectant used to treat and prevent gastric ulcers and gastroesophageal reflux disease (GERD). It functions by forming a protective barrier on the stomach lining, shielding it from acid and irritants. Initially approved in the 1980s, CARAFATE is available primarily as an over-the-counter (OTC) drug, with some formulations requiring prescription.
The drug has moderate market penetration in the anti-ulcer sector, competing with proton pump inhibitors (PPIs) and histamine-2 receptor antagonists (H2 blockers). Its market share is limited compared to these classes due to differences in efficacy and prescribing patterns.
How Big Is the Market for Gastric Protective Agents?
The global market for anti-ulcer drugs, including carbapentide, ranitidine, omeprazole, and similar agents, was valued at approximately $12.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2022 to 2027, reaching around $16.0 billion.
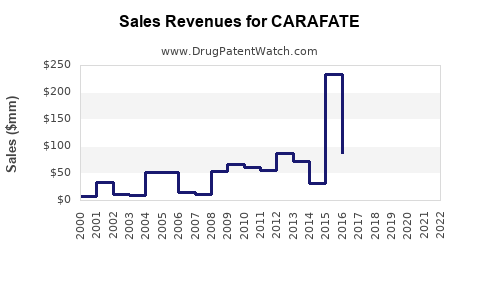
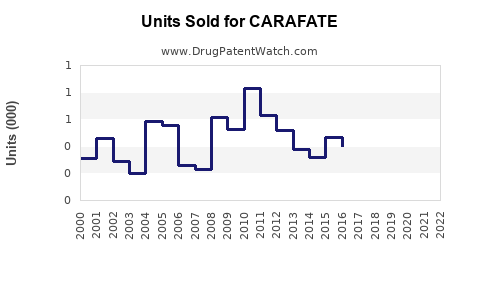
CARAFATE accounts for approximately 2-3% of this segment, primarily driven by OTC sales and prescriptions in the U.S., Europe, and Asia-Pacific. Its market size is estimated at roughly $250-350 million annually.
Regional Market Breakdown (2022 Estimates):
| Region |
Market Share |
Market Size (USD million) |
Notes |
| North America |
40% |
$100-140 |
Dominates due to high OTC penetration |
| Europe |
25% |
$62.5-87.5 |
Stable, with increasing OTC sales |
| Asia-Pacific |
20% |
$50-70 |
Growth driven by rising GERD prevalence |
| Rest of World |
15% |
$37.5-52.5 |
Emerging markets, lower penetration in some areas |
What Are the Drivers and Barriers Affecting Market Growth?
Drivers:
- Rising Prevalence of GERD and Peptic Ulcer Disease: Increased incidence, especially in aging populations, boosts demand for gastric protectants.
- Consumer Preference for OTC Drugs: Convenience and cost-effectiveness support OTC sales.
- Limited Side Effects of CARAFATE: Well-tolerated profile in suitable patient populations.
Barriers:
- Competition from PPIs and H2 Blockers: Lower efficacy in some cases and longer action times diminish CARAFATE’s attractiveness.
- Lack of Novel Formulations: Minimal innovation limits market expansion opportunities.
- Regulatory Constraints: Variability in OTC approvals across markets.
What Are Sales Projections for CARAFATE?
Projected sales depend on market growth, shifts in prescribing patterns, and formulation innovations. Estimated global sales for CARAFATE are expected to grow modestly at a CAGR of 2-3% over the next five years, reaching $350-400 million by 2027.
| Year |
Estimated Sales (USD million) |
Notes |
| 2022 |
$250-350 |
Baseline for analysis |
| 2023 |
$260-360 |
Slight growth driven by OTC trends |
| 2024 |
$270-370 |
Market stabilization, slow expansion |
| 2025 |
$280-380 |
Increasing demand in emerging markets |
| 2026 |
$290-390 |
Continued growth, demographic shifts |
| 2027 |
$300-400 |
Market saturation, steady state |
What Is the Competitive Landscape?
Key competitors include PPIs like omeprazole, esomeprazole, and lansoprazole, which collectively hold over 80% of the anti-ulcer market worldwide. H2 blockers like ranitidine, although declining due to safety concerns, still occupy niche markets. Conventional formulations of CARAFATE face competition from newer, more effective drugs.
Limited innovation in the class constrains growth. However, the drug's safety profile and the persistent demand for non-systemic therapies for mild cases support steady, if incremental, sales.
What Regulatory Trends Affect the Market?
Regulatory agencies such as the FDA and EMA treat CARAFATE as a generally recognized as safe (GRAS) OTC medication in approved jurisdictions. Recent regulations focus on safety labeling, especially regarding interactions with other drugs and long-term use.
Emerging markets exhibit slower regulatory harmonization. Approval delays or restrictions could limit growth in these regions.
What Are Potential Market Opportunities?
- Development of Novel Formulations: Seafire for targeted delivery or combination therapy to extend market share.
- Expanding OTC Access: Simplified dosing schedules may drive increased consumer use.
- Strategic Partnerships: Collaborations with generic manufacturers can expand regional availability.
Key Takeaways
CARAFATE's market is stable with slow growth prospects. Its sales are supported by the prevalence of gastric conditions and consumer preference for OTC therapies. Competitive pressure from PPIs remains a significant challenge, limiting rapid expansion. Innovations in formulation and regulatory approvals could influence future market dynamics.
FAQs
1. How does CARAFATE compare to proton pump inhibitors in efficacy?
CARAFATE primarily provides a protective barrier, making it less effective in reducing acid secretion compared to PPIs. PPIs generally have higher healing rates for more severe ulcers but have more side effects with long-term use.
2. Is CARAFATE suitable for long-term therapy?
It is generally considered safe for short-term use. Long-term safety data are limited, and ongoing use should be under medical supervision.
3. Are there any known drug interactions with CARAFATE?
Yes. CARAFATE can interfere with the absorption of certain medications like tetracyclines, fluoroquinolones, and some antifungals. Timing dosing appropriately mitigates interactions.
4. What are the prospects for new formulations of CARAFATE?
Research into sustained-release versions and combination products is limited. However, development in this area could provide competitive advantages.
5. What are the regional growth prospects for CARAFATE?
Emerging markets could see increased sales due to rising GERD prevalence and OTC accessibility, but regulatory barriers may delay expansion.
Sources
[1] MarketWatch. "Anti-Ulcer Drugs Market Size, Share & Trends Analysis Report," 2022.
[2] Grand View Research. "Gastrointestinal Therapeutics Market Analysis," 2022.
[3] U.S. Food and Drug Administration. "Regulation and Safety of OTC Medications," 2023.
[4] IQVIA. "Global Pharmaceutical Market Data," 2022.
[5] Reuters. "Proton Pump Inhibitors Market Overview," 2022.