Share This Page
Drug Sales Trends for hydrochlorothiazide
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for hydrochlorothiazide (2002)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
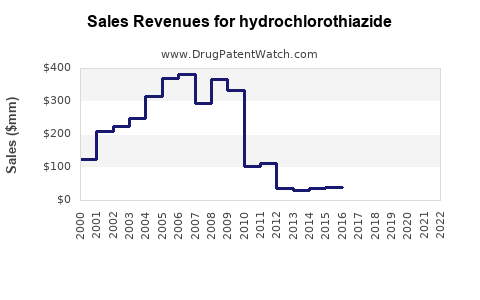
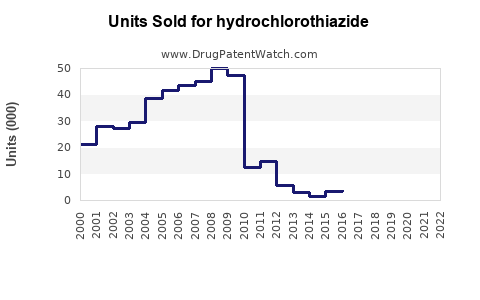
Annual Sales Revenues and Units Sold for hydrochlorothiazide
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| HYDROCHLOROTHIAZIDE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Hydrochlorothiazide Market Analysis and Sales Projections
Hydrochlorothiazide (HCTZ), a thiazide diuretic, remains a cornerstone in the management of hypertension and edema. Its established efficacy, favorable safety profile, and low cost contribute to its sustained market presence. This analysis details current market dynamics, patent landscape, and projected sales, providing an outlook for stakeholders.
What is the Current Market Size and Growth Rate for Hydrochlorothiazide?
The global hydrochlorothiazide market is valued at approximately USD 700 million as of 2023. The market has demonstrated consistent, albeit modest, growth, projected to expand at a compound annual growth rate (CAGR) of 2.5% to 3.0% over the next five years, reaching an estimated USD 810 million to 835 million by 2028. This growth is driven by the persistent global burden of cardiovascular diseases, particularly hypertension, and the drug's affordability and accessibility.
Key Market Drivers:
- Prevalence of Hypertension: Hypertension affects an estimated 1.28 billion adults globally. HCTZ is a first-line treatment option, driving consistent demand [1].
- Aging Population: The increasing elderly population, which is more susceptible to cardiovascular conditions, contributes to sustained demand for antihypertensive medications like HCTZ.
- Cost-Effectiveness: As a generic drug with a long history of use, HCTZ offers a highly cost-effective treatment option compared to newer, branded antihypertensives. This is particularly relevant in healthcare systems facing budgetary constraints.
- Combination Therapies: HCTZ is frequently used in fixed-dose combination products with other antihypertensives (e.g., ACE inhibitors, ARBs, beta-blockers), further solidifying its market position and expanding its utility.
Key Market Restraints:
- Competition from Newer Antihypertensives: The development and marketing of newer drug classes with potentially improved efficacy or side-effect profiles pose a competitive threat.
- Genericization of Other Drugs: The increasing availability of generic versions of other antihypertensive classes can also erode HCTZ's market share in certain patient segments.
- Adverse Event Profile: While generally well-tolerated, HCTZ can cause electrolyte imbalances (e.g., hypokalemia, hyponatremia) and other side effects, which may limit its use in some patients.
What is the Patent Landscape for Hydrochlorothiazide?
Hydrochlorothiazide was first patented in the 1950s. Its primary patents have long since expired. Consequently, the market is characterized by a robust generic presence.
- Original Patent Filing: The foundational patents for hydrochlorothiazide have expired, allowing for widespread generic manufacturing and sale [2].
- Post-Patent Innovations: While no significant new patentable inventions related to the core HCTZ molecule are expected, innovation may focus on:
- New Formulations: Extended-release formulations or novel drug delivery systems could potentially secure new intellectual property, though market adoption may be limited by cost considerations for a generic drug.
- Combination Products: Patents for fixed-dose combination products containing HCTZ with other active pharmaceutical ingredients (APIs) are more common. These patents protect specific synergistic combinations and dosages, extending market exclusivity for these particular formulations.
- Manufacturing Process Improvements: Patents may be sought for novel or more efficient manufacturing processes, though these are typically less impactful on market exclusivity compared to composition-of-matter patents.
The absence of strong, active composition-of-matter patents on HCTZ itself means that the competitive landscape is primarily driven by manufacturing costs, regulatory approvals, and market access.
How is Hydrochlorothiazide Formulated and Administered?
Hydrochlorothiazide is predominantly available in oral dosage forms.
- Dosage Strengths: Common strengths include 12.5 mg, 25 mg, and 50 mg tablets.
- Formulations:
- Immediate-Release Tablets: This is the most common and cost-effective formulation.
- Fixed-Dose Combinations (FDCs): HCTZ is widely combined with other antihypertensive agents, including:
- ACE Inhibitors: Lisinopril/HCTZ, Enalapril/HCTZ, Ramipril/HCTZ.
- Angiotensin II Receptor Blockers (ARBs): Losartan/HCTZ, Valsartan/HCTZ, Olmesartan/HCTZ.
- Beta-Blockers: Atenolol/HCTZ, Metoprolol/HCTZ.
- Potassium-Sparing Diuretics: Amiloride/HCTZ, Triamterene/HCTZ.
- Administration: HCTZ is typically administered orally once or twice daily. The timing of administration may be adjusted to minimize nocturia.
The prevalence of FDCs is a significant factor in the sustained demand for HCTZ, as it simplifies treatment regimens for patients and often offers a synergistic therapeutic effect.
What are the Key Therapeutic Indications and Off-Label Uses for Hydrochlorothiazide?
The primary therapeutic indications for hydrochlorothiazide are well-established.
Approved Indications:
- Edema: Management of fluid retention associated with congestive heart failure, liver cirrhosis, and renal disease [3].
- Hypertension: Treatment of high blood pressure, either as monotherapy or in combination with other antihypertensive agents. It is recognized as a first-line option for managing mild to moderate hypertension.
Off-Label Uses:
While its primary indications are well-defined, HCTZ may be used off-label in specific clinical contexts, though these are less common and depend on physician judgment and evolving clinical practice guidelines. These are not typically significant market drivers.
What are the Projected Sales and Market Share for Hydrochlorothiazide Over the Next Five Years?
Projected sales for HCTZ indicate continued market stability, with growth closely tied to the overall antihypertensive market and the drug's position as a cost-effective option.
Global Hydrochlorothiazide Market Sales Projections (USD Millions):
| Year | Estimated Sales |
|---|---|
| 2023 | 700 |
| 2024 | 718 |
| 2025 | 737 |
| 2026 | 756 |
| 2027 | 776 |
| 2028 | 796 |
Note: These figures represent the global market value for HCTZ as a single agent and within fixed-dose combination products where it is a component. These are estimates based on current market trends and do not include future R&D breakthroughs for novel HCTZ-based therapies, which are unlikely given the drug's age.
Market Share Dynamics:
- Generic Dominance: HCTZ operates in a mature generic market. Its market share is significant within the thiazide diuretic class.
- Combination Product Contribution: A substantial portion of HCTZ's market value is derived from its inclusion in fixed-dose combination products. These combinations often hold greater market share than HCTZ monotherapy due to improved patient compliance and efficacy.
- Geographic Variations: Market penetration and sales figures can vary by region, influenced by healthcare infrastructure, pricing regulations, and local prescribing patterns. Developed markets like North America and Europe represent significant portions of sales, while emerging markets are expected to contribute to growth due to increasing access to healthcare.
What is the Competitive Landscape for Hydrochlorothiazide?
The competitive landscape for HCTZ is characterized by numerous generic manufacturers and a broad array of antihypertensive agents.
Direct Competition (Thiazide Diuretics):
While HCTZ is the most widely prescribed thiazide diuretic, other drugs in this class exist, such as:
- Chlorthalidone: Often considered more potent and longer-acting than HCTZ, leading to its preference in some clinical guidelines and markets.
- Indapamide: Another thiazide-like diuretic with similar efficacy and a comparable safety profile.
Indirect Competition (Other Antihypertensive Classes):
HCTZ faces significant competition from other established and newer classes of antihypertensive drugs, which are frequently used in combination with or as alternatives to HCTZ.
- Angiotensin-Converting Enzyme (ACE) Inhibitors: e.g., Lisinopril, Enalapril, Ramipril.
- Angiotensin II Receptor Blockers (ARBs): e.g., Losartan, Valsartan, Olmesartan.
- Beta-Blockers: e.g., Atenolol, Metoprolol, Bisoprolol.
- Calcium Channel Blockers (CCBs): e.g., Amlodipine, Nifedipine, Diltiazem.
- Direct Renin Inhibitors: e.g., Aliskiren.
- Aldosterone Antagonists: e.g., Spironolactone, Eplerenone.
The choice of therapy often depends on individual patient characteristics, comorbidities, and established treatment guidelines, which frequently recommend a stepped approach where HCTZ or another thiazide is a common initial or add-on therapy.
What are the Regulatory Considerations and Manufacturing Aspects?
The manufacturing and regulatory approval of HCTZ are governed by established pharmaceutical frameworks.
- Regulatory Bodies: Approval for HCTZ products is sought from national regulatory agencies, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others globally.
- Generic Drug Approval: Manufacturers seeking to market generic HCTZ must demonstrate bioequivalence to an already approved reference listed drug. This process involves submitting an Abbreviated New Drug Application (ANDA) to the FDA [4].
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy.
- Quality Control: Robust quality control measures are essential for active pharmaceutical ingredients (APIs) and finished drug products, covering aspects like purity, potency, dissolution, and stability.
- Supply Chain: The global supply chain for HCTZ APIs is well-established, with multiple manufacturers, primarily in Asia (China and India), supplying the market. This contributes to competitive pricing.
- Pharmacovigilance: Post-market surveillance and pharmacovigilance activities are mandatory to monitor for adverse drug reactions and ensure continued product safety.
Key Takeaways
- Hydrochlorothiazide (HCTZ) is a mature, high-volume generic drug with an estimated global market value of USD 700 million in 2023.
- The market is projected to grow at a CAGR of 2.5% to 3.0%, reaching approximately USD 810 million to USD 835 million by 2028, driven by the prevalence of hypertension and its cost-effectiveness.
- The patent landscape is characterized by expired foundational patents, leading to intense generic competition.
- Innovation is primarily focused on fixed-dose combination products, which contribute significantly to HCTZ's market presence.
- HCTZ faces competition from other antihypertensive classes, but its role as a first-line agent and in combination therapies ensures sustained demand.
- Manufacturing is concentrated among numerous generic producers globally, with robust regulatory oversight ensuring product quality.
Frequently Asked Questions
-
What is the primary reason for the sustained demand for hydrochlorothiazide despite the availability of newer antihypertensives? The sustained demand for hydrochlorothiazide is primarily attributed to its established efficacy, favorable safety profile, and significant cost-effectiveness, making it a cornerstone in hypertension management and a widely adopted first-line therapy and component of combination treatments.
-
Are there any new patent filings expected for hydrochlorothiazide itself? Given the age of hydrochlorothiazide and the expiration of its original patents, new patent filings for the core molecule itself are highly unlikely. Innovation is primarily focused on novel formulations or fixed-dose combinations.
-
How does the market for hydrochlorothiazide differ between developed and emerging economies? In developed economies, demand is driven by established healthcare systems and the widespread use of combination therapies. In emerging economies, growth is influenced by increasing access to healthcare, rising hypertension prevalence, and the drug's affordability as a cost-effective treatment option.
-
What are the most common side effects associated with hydrochlorothiazide use that could impact its market share? Common side effects include electrolyte imbalances (hypokalemia, hyponatremia), dizziness, and increased urination. While generally manageable, these can limit its use in certain patient populations or necessitate closer monitoring.
-
What is the typical market share of hydrochlorothiazide within the broader antihypertensive drug market? Hydrochlorothiazide holds a significant share within the diuretic class and, by extension, contributes substantially to the overall antihypertensive market, particularly when considering its inclusion in numerous fixed-dose combination products, which command a large segment of the market.
Citations
[1] World Health Organization. (2023). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-drug-ratings/orange-book
[3] National Institutes of Health. (n.d.). Hydrochlorothiazide. In MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a681026.html
[4] U.S. Food and Drug Administration. (n.d.). Generic Drugs. Retrieved from https://www.fda.gov/drugs/generic-drugs
More… ↓
