Share This Page
Drug Sales Trends for gabapentin
✉ Email this page to a colleague
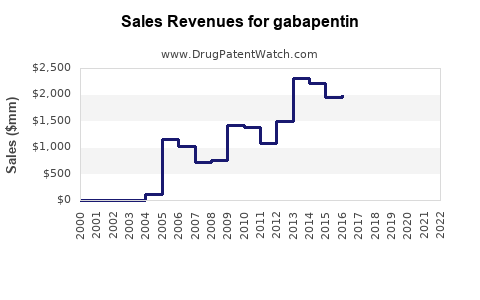
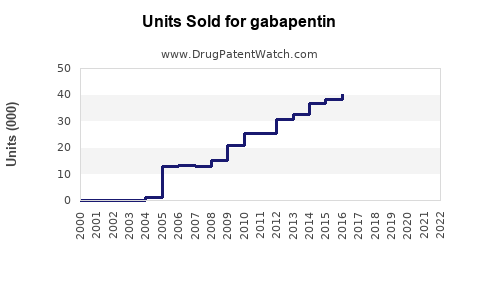
Annual Sales Revenues and Units Sold for gabapentin
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GABAPENTIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GABAPENTIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GABAPENTIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GABAPENTIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| GABAPENTIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Gabapentin
What is the Current Market Size and Key Drivers?
Gabapentin, marketed under brand names such as Neurontin, is a prescription medication primarily used to treat neuropathic pain, epilepsy, and off-label indications like anxiety disorders. The global gabapentin market size reached approximately $1.8 billion in 2022, with annual growth rates between 4% and 6%. The key drivers include rising prevalence of neuropathic pain, epilepsy, and expanding off-label uses.
Major markets include the United States, Europe, and China. The U.S. accounts for over 60% of global sales, driven by high prescription volume and >=65 years aging population.
What Are Market Segments and Growth Dynamics?
Indications:
- Neuropathic pain (~45% of sales): Characterized by chronicity and complex management needs.
- Epilepsy (~35%): Increasing awareness supports sustained demand.
- Off-label uses (~20%): Anxiety, sleep disorders, and migraine prevention.
Distribution Channels:
- Retail pharmacies (~70%) remain dominant.
- Hospital procurement (~20%) influences sales, especially in epilepsy treatment.
- Online pharmacies (~10%) show growing contribution.
Geographic Trends:
| Region | 2022 Market Share | Growth Rate (2022-2027) | Key Factors |
|---|---|---|---|
| United States | 60% | 3.8% | Prescribing guidelines, aging |
| Europe | 23% | 5.2% | Increased diagnosis of neuropathy |
| China | 9% | 7.0% | Expanding healthcare access |
| Rest of World | 8% | 5.5% | Market penetration initiatives |
What Are Projections for Future Sales?
Based on current trends and anticipated market developments, global gabapentin sales are projected to reach roughly $2.4 billion by 2027, with a compound annual growth rate (CAGR) of 4.5%. The growth outlook relies on rising demand for chronic pain therapies, aging populations, and increased off-label prescribing.
Projected annual sales (2023-2027):
| Year | Estimated Global sales (USD billions) | Growth Rate |
|---|---|---|
| 2023 | 2.0 | 4.2% |
| 2024 | 2.1 | 4.8% |
| 2025 | 2.2 | 4.5% |
| 2026 | 2.3 | 4.5% |
| 2027 | 2.4 | 4.5% |
How Will Patent Expirations and Generic Competition Impact Sales?
The original patents for gabapentin expired in the early 2000s in the U.S. and Europe. This led to widespread generic entry, which significantly reduced branded drug prices and margins.
Generic versions dominate the market, accounting for approximately 85% of sales volume globally. Price erosion is expected to continue, with estimates indicating a 10-15% annual decline in average selling price (ASP) in mature markets.
The entry of specialized formulations (e.g., extended-release capsules) or combination therapies could affect demand dynamics. Regulatory restrictions or new prescribing guidelines targeting off-label use may also influence future sales.
What Are Opportunities and Risks?
Opportunities:
- Expansion into emerging markets with increasing healthcare access.
- Development of extended-release formulations to improve compliance.
- Exploration of new indications, such as fibromyalgia or diabetic neuropathy.
Risks:
- Growing regulatory scrutiny over off-label prescriptions.
- Competition from newer therapies for neuropathic pain and epilepsy.
- Price competition from generics reducing profit margins.
Investment Considerations
Investors should monitor patent statuses across key markets, regulatory policies affecting off-label use, and developments in formulations or combination treatments. Companies with diversified portfolio strategies or authorized generics may better withstand market pressures. Market entrants leveraging biosimilar or novel delivery systems could reshape demand.
Key Takeaways
- The gabapentin market was valued at approximately $1.8 billion in 2022.
- Sales are projected to reach $2.4 billion by 2027, growing at 4.5% CAGR.
- The U.S. leads market share; Europe and China offer growth potential.
- Generic competition results in declining ASP; innovation is necessary to sustain revenue.
- Regulatory policies and emerging formulations influence future sales.
FAQs
Q1: How will patent expirations affect gabapentin sales?
A1: Patent expirations have led to widespread generic competition, reducing prices and profit margins. Future patent protections are unlikely, emphasizing reliance on generics for volume.
Q2: What growth opportunities exist for gabapentin?
A2: Expanding into emerging markets, developing novel formulations, and establishing new approved indications offer potential growth avenues.
Q3: What are the main risks facing gabapentin sales?
A3: Price erosion from generics, regulatory restrictions on off-label prescribing, and competition from newer drugs pose significant risks.
Q4: How significant is off-label use in sales projections?
A4: Off-label use accounts for about 20% of sales, with potential restrictions in some regions possibly impacting revenue.
Q5: What is the impact of new formulations on the market?
A5: Extended-release formulations could improve patient compliance and open new market segments, but they must demonstrate clear advantages over existing products.
References
[1] MarketWatch. (2023). Gabapentin Market Size, Share & Trends.
[2] Frost & Sullivan. (2022). Global Neuropathic Pain Therapy Market Analysis.
[3] European Medicines Agency. (2021). Gabapentin: Safety and Prescribing Guidelines.
[4] IQVIA. (2022). Prescription Drug Market Data.
[5] U.S. Food and Drug Administration. (2022). Patent and Exclusivity Data.
More… ↓
