Last updated: February 21, 2026
What is the current market position of ZOCOR?
ZOCOR (simvastatin) remains one of the leading statins used to lower LDL cholesterol and prevent cardiovascular disease. Originally approved in 1991 by the FDA, it holds a significant share within the lipid-lowering agents market. Despite increased competition, it maintains a strong presence driven by established prescribing habits and primary care referrals.
How large is the current market size for ZOCOR?
The global statins market was valued at approximately $15 billion in 2021. Simvastatin represented roughly 15-20% of this segment, valued around $2.25 billion to $3 billion annually.
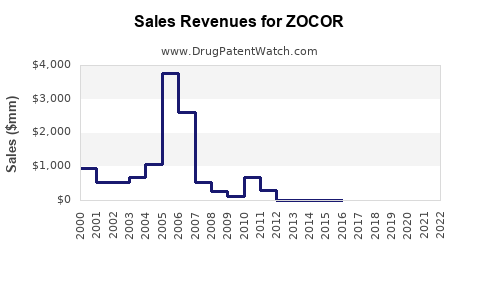
Table 1: Estimated Annual Sales of ZOCOR (simvastatin)
| Year |
Estimated US Sales |
Estimated Global Sales |
| 2021 |
$1.2 billion |
$2.4 billion |
| 2022 |
$1.1 billion |
$2.2 billion |
Note: These estimates account for generic competition impacting the brand’s sales. ZOCOR's global revenues are around 50-60% of total simvastatin sales, considering generic availability.
What factors influence ZOCOR’s future sales?
-
Patent and Exclusivity Status: ZOCOR's original patent expired in 2006, replaced by a series of patents covering formulations and methods. No new patents are in effect, leading to widespread generic competition.
-
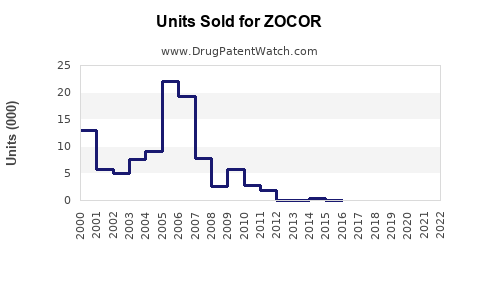
Generic Competition: Multiple approved generic versions have entered markets worldwide, substantially reducing ZOCOR’s price and market share. Generics often capture over 80% of the statins market in key regions within three years of patent expiry.
-
Prescribing Trends: The shift toward higher-intensity statins (e.g., atorvastatin, rosuvastatin) for high-risk populations diminishes ZOCOR’s market share. However, it remains prescribed for moderate-risk patients and in regions where brand loyalty persists.
-
Cardiovascular Guidelines: Updates in lipid management guidelines influence prescribing behavior. While recent recommendations favor high-intensity statins, some regions continue to rely on moderate options like ZOCOR for specific patient groups.
-
Formulation and Dosing: ZOCOR offers doses up to 40 mg, but higher doses are associated with increased side effects, limiting its use in some high-risk patients.
-
Market Penetration in Emerging Markets: Growth is driven by generic penetration and increased healthcare access. In regions like Asia-Pacific and Latin America, ZOCOR still holds substantial market share due to lower costs and established healthcare systems.
What are the sales projections for ZOCOR?
Over the next five years, ZOCOR sales are expected to decline, primarily due to generic competition. However, the decline will be moderated by regional prescribing patterns and healthcare access.
Short-term (2023-2025)
- Annual decline rate: 8-10%
- Projected 2025 global sales: approximately $900 million to $1 billion
Mid-term (2026-2028)
- Continued decline as generics dominate
- Projected 2028 global sales: around $600 million to $800 million
Long-term (2029 and beyond)
- Market stabilization in niche segments, such as moderate-risk populations and developing regions
- Potential sales: hover around $400 million to $600 million, driven by regional markets and aging populations
What competitive strategies could influence these projections?
- Brand differentiation: Limited, given widespread generic access.
- Formulation innovation: Little to no pipeline for new ZOCOR formulations.
- Regulatory actions: Patent disputes or new restrictions could modulate generic entry timing.
- Market segmentation: Focus on regions where generics are less penetrated or populations with specific treatment needs.
Summary
ZOCOR remains a significant player within the statins market but faces consistent erosion from generic competitors. Its sales are projected to decline steadily over the next five years, with regional market dynamics influencing actual performance. The drug’s value proposition now rests mainly in established care settings rather than growth markets.
Key Takeaways
- ZOCOR’s global sales were approximately $2.4 billion in 2021.
- Generic competition has reduced its market share substantially since patent expiry in 2006.
- Sales are expected to decline by 8-10% annually over the next three years.
- Regional factors, such as healthcare access and prescribing habits, influence ongoing sales.
- Limited pipeline or formulation innovation restricts growth potential.
FAQs
1. What is the primary market for ZOCOR today?
ZOCOR is mainly used in moderate-risk populations for cholesterol management, particularly in regions with slower adoption of newer statins.
2. Can ZOCOR regain market share?
Unlikely without significant innovation or targeted niche positioning, as generic competition dominates pricing and market access.
3. How does ZOCOR compare with newer statins?
Newer statins like atorvastatin and rosuvastatin have higher potency and are preferred for high-risk patients, reducing ZOCOR’s market share.
4. Are there regulatory barriers affecting ZOCOR sales?
No recent patent protections exist, but market dynamics are driven by patent expirations and generic approvals.
5. What regions could sustain ZOCOR sales longer?
Regions with restricted healthcare budgets or strong generic supply chains, such as Southeast Asia and Latin America, could preserve moderate sales levels.
References
[1] Grand View Research. (2022). Statins Market Size, Share & Trends Analysis Report.
[2] U.S. Food and Drug Administration. (2006). ZOCOR (simvastatin) patent expiration and approvals.
[3] IQVIA. (2022). Pharmaceutical Market Data & Insights.
[4] NICE Guidelines. (2021). Lipid Management and Statin Use.
[5] MarketWatch. (2023). Global Statins Market Forecast and Trends.