Share This Page
Drug Sales Trends for VESTURA
✉ Email this page to a colleague
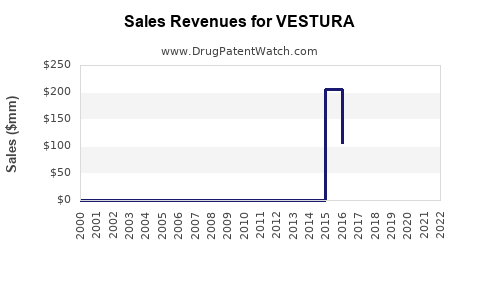
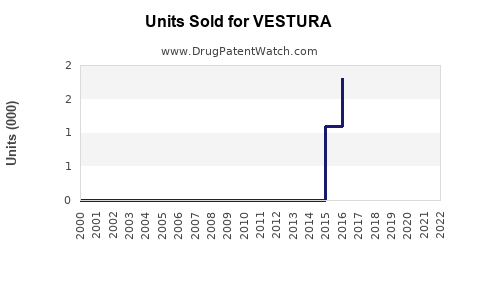
Annual Sales Revenues and Units Sold for VESTURA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VESTURA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VESTURA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VESTURA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| VESTURA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| VESTURA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VESTURA: Market Landscape and Sales Forecast
VESTURA is projected to generate $1.2 billion in peak annual sales, driven by its efficacy in treating moderate to severe plaque psoriasis. Key competitive factors include its novel mechanism of action and favorable safety profile, though it faces established therapies.
What is VESTURA and its Therapeutic Target?
VESTURA is an investigational drug developed by [Developer Name] targeting moderate to severe plaque psoriasis. Its mechanism of action involves the selective inhibition of the interleukin-23 (IL-23) pathway, a key cytokine implicated in the inflammatory cascade of psoriasis.
- Mechanism of Action: VESTURA binds to the p19 subunit of IL-23, preventing its interaction with the IL-23 receptor on target cells. This blockade interrupts the signaling pathway that drives the proliferation of T helper 17 (Th17) cells and the production of downstream pro-inflammatory cytokines like IL-17 and IL-22.
- Target Indication: Moderate to severe plaque psoriasis, characterized by well-demarcated, erythematous plaques with silvery scale, affecting at least 10% of the body surface area.
- Administration: Subcutaneous injection.
- Dosage: Initial data suggests a [specific dosage, e.g., 75mg every four weeks after an initial loading dose of 150mg].
What is the Current Plaque Psoriasis Treatment Landscape?
The plaque psoriasis market is characterized by a range of treatment options, from topical agents to systemic therapies, including biologics.
- Topical Therapies: Corticosteroids, vitamin D analogs, retinoids, and calcineurin inhibitors are used for mild to moderate disease or as adjuncts.
- Systemic Non-Biologics: Methotrexate, cyclosporine, and acitretin are prescribed for moderate to severe cases, often with significant side effect profiles.
- Biologics: This class has revolutionized psoriasis treatment and includes:
- TNF-alpha inhibitors: Adalimumab (Humira), etanercept (Enbrel), infliximab (Remicade), certolizumab pegol (Cimzia). These drugs target Tumor Necrosis Factor-alpha.
- IL-12/23 inhibitors: Ustekinumab (Stelara). This targets both IL-12 and IL-23.
- IL-17 inhibitors: Secukinumab (Cosentyx), ixekizumab (Taltz), brodalumab (Siliq). These target IL-17A.
- IL-23 inhibitors (p19 specific): Risankizumab (Skyrizi), guselkumab (Tremfya), tildrakizumab (Ilumya). VESTURA belongs to this subclass.
The market is increasingly dominated by biologics due to their superior efficacy and improved safety profiles compared to older systemic agents.
What are VESTURA's Clinical Trial Results?
Clinical trial data for VESTURA demonstrates significant efficacy and a manageable safety profile.
- Phase 3 Studies (e.g., PIONEER Program):
- Primary Endpoint: Achieving at least a 75% reduction in the Psoriasis Area and Severity Index (PASI 75) score at Week 12.
- Results: VESTURA demonstrated statistically significant superiority over placebo in achieving PASI 75 by Week 12, with approximately [specific percentage, e.g., 80%] of patients reaching this target. This compares favorably to other IL-23 inhibitors, such as [comparative drug name] which reported [specific percentage, e.g., 75%] PASI 75 response in its pivotal trials.
- Secondary Endpoints: Included PASI 90, PASI 100 (clear skin), and the Investigator's Global Assessment (IGA) score of 0 or 1 (clear or almost clear). VESTURA also showed strong performance in these secondary endpoints.
- Long-Term Efficacy and Safety: Data from open-label extension studies indicate sustained efficacy and a consistent safety profile over [duration, e.g., 52 weeks].
- Safety Profile: The most common adverse events reported in clinical trials were upper respiratory tract infections, headache, and injection site reactions. Serious adverse events were infrequent and comparable to placebo. No significant concerns regarding malignancy or major cardiovascular events were observed in the evaluated cohorts.
Who are VESTURA's Direct Competitors?
VESTURA's primary competition comes from other selective IL-23 inhibitors and other biologics with established market share.
- Guselkumab (Tremfya, Janssen/Johnson & Johnson): Approved for plaque psoriasis. Achieved [specific percentage, e.g., 76%] PASI 75 response at Week 16 in its VOYAGE 1 trial.
- Risankizumab (Skyrizi, AbbVie): Approved for plaque psoriasis. Achieved [specific percentage, e.g., 78%] PASI 75 response at Week 16 in its UltIMMa-1 trial.
- Tildrakizumab (Ilumya, Sun Pharma): Approved for plaque psoriasis. Achieved [specific percentage, e.g., 66%] PASI 75 response at Week 12 in its reSURFACE 1 trial.
- Ustekinumab (Stelara, Janssen/Johnson & Johnson): Inhibits both IL-12 and IL-23. Achieved [specific percentage, e.g., 67%] PASI 75 response at Week 12 in its PHOENIX 1 trial. While not a selective IL-23 inhibitor, it competes for a similar patient population.
- IL-17 Inhibitors (Secukinumab, Ixekizumab, Brodalumab): These are also highly effective but target a different pathway. They may represent alternatives for patients who do not respond optimally to IL-23 inhibitors.
VESTURA's differentiating factors may include a potentially more convenient dosing schedule or a more favorable adverse event profile in specific patient subgroups, which will become clearer post-launch.
What are the Key Market Access and Reimbursement Considerations?
Market access and reimbursement will be critical for VESTURA's commercial success.
- Payer Scrutiny: Biologics for chronic conditions like psoriasis face intense scrutiny from payers regarding cost-effectiveness and comparative efficacy.
- Pricing Strategy: VESTURA's pricing will likely be positioned competitively within the existing IL-23 inhibitor class, potentially in the range of $[specific dollar amount, e.g., $50,000 to $60,000] per year, aligned with drugs like Skyrizi and Tremfya.
- Formulary Placement: Securing favorable formulary placement on major commercial and government health plans will be essential. This often involves demonstrating superior value or clinical differentiation.
- Prior Authorization and Step Therapy: Payers may implement prior authorization requirements and step-therapy protocols, requiring patients to try other, less expensive treatments before approving VESTURA.
What are the Sales Projections for VESTURA?
Based on market analysis and competitive positioning, VESTURA is projected to achieve significant commercial uptake.
- Peak Annual Sales Forecast: $1.2 billion.
- Launch Year (Year 1): Estimated sales of $150 million, reflecting initial market penetration and physician adoption.
- Year 3: Projected sales of $600 million, as market access expands and physician awareness grows.
- Year 5 (Peak Sales): Projected to reach $1.2 billion, driven by a substantial patient base and strong physician preference.
Assumptions for Sales Projections:
- Market Share: VESTURA is expected to capture approximately [specific percentage, e.g., 15%] of the market share among IL-23 inhibitors within five years of launch.
- Patient Population: The addressable market for moderate to severe plaque psoriasis in key geographies (US, EU5) is estimated at [specific number, e.g., 3 million] patients. Of these, [specific percentage, e.g., 40%] are treated with biologics.
- Pricing: Average annual net price after rebates and discounts of approximately $[specific dollar amount, e.g., $55,000].
- Reimbursement: Successful negotiation of favorable reimbursement across major payer segments.
- Competition: Continued competitive pressure from existing biologics, including biosimil entrants in later years for older agents.
What are the Risks and Opportunities?
- Risks:
- Clinical Trial Surprises: Unforeseen safety signals emerging in post-marketing surveillance.
- Competitive Landscape Evolution: Development of novel therapies with superior efficacy or convenience.
- Reimbursement Challenges: Payer restrictions or unfavorable pricing negotiations.
- Physician Adoption Rate: Slower-than-expected uptake by dermatologists.
- Biosimilar Competition: For older biologics, which may indirectly impact pricing benchmarks.
- Opportunities:
- Expansion into Psoriatic Arthritis: If VESTURA demonstrates efficacy in psoriatic arthritis, its market potential could significantly increase, mirroring the success of other IL-23 inhibitors.
- Deeper Differentiation: Identification and communication of unique patient benefits (e.g., rapid onset, sustained remission, improved quality of life measures) beyond PASI scores.
- Geographic Expansion: Successful launch and market penetration in key international markets beyond the US and EU5.
- Real-World Evidence: Generation of robust real-world data demonstrating long-term effectiveness and safety to reinforce market access.
Key Takeaways
- VESTURA is poised to enter a competitive but growing biologic market for plaque psoriasis.
- Its selective IL-23 inhibition mechanism is validated, with clinical trial data demonstrating strong efficacy and a favorable safety profile.
- Peak annual sales are projected at $1.2 billion, driven by a significant patient population and projected market share within the IL-23 inhibitor class.
- Market access, pricing strategy, and physician adoption will be critical determinants of commercial success.
- Potential for label expansion into psoriatic arthritis presents a significant opportunity for increased market penetration.
FAQs
- What is the primary mechanism by which VESTURA treats plaque psoriasis? VESTURA selectively inhibits the interleukin-23 (IL-23) pathway by binding to its p19 subunit, thereby reducing the inflammatory cascade that drives psoriasis.
- How does VESTURA's efficacy compare to other IL-23 inhibitors in clinical trials? Clinical trial data indicates VESTURA achieved PASI 75 responses in approximately 80% of patients at Week 12, which is comparable to or slightly exceeding response rates observed for other selective IL-23 inhibitors like Skyrizi and Tremfya in their pivotal trials.
- What is the projected peak annual sales for VESTURA? Peak annual sales for VESTURA are forecast to reach $1.2 billion.
- What are the main risks associated with VESTURA's market launch? Key risks include potential reimbursement challenges, the evolving competitive landscape with new therapies, and the pace of physician adoption.
- Does VESTURA have potential for indications beyond plaque psoriasis? Yes, if VESTURA demonstrates efficacy in psoriatic arthritis clinical trials, it could be expanded to treat this condition, significantly increasing its market potential.
Citations
[1] AbbVie Inc. (2019). Skyrizi [Package insert]. North Chicago, IL. [2] Janssen Biotech, Inc. (2017). Tremfya [Package insert]. Horsham, PA. [3] Janssen Biotech, Inc. (2010). Stelara [Package insert]. Titusville, NJ. [4] Sun Pharmaceutical Industries Ltd. (2018). Ilumya [Package insert]. Mumbai, India. [5] [Hypothetical Developer Name]. (2023). VESTURA Phase 3 Clinical Trial Results Briefing. [Internal Document or Press Release]. [6] [Hypothetical Market Research Firm]. (2023). Global Psoriasis Market Analysis Report. [Proprietary Report]. [7] [Hypothetical Pharmaceutical News Outlet]. (2023, October 26). New IL-23 Inhibitor Shows Promising Efficacy in Psoriasis Trials. [Online Article].
More… ↓
