Share This Page
Drug Sales Trends for TOLTERODINE
✉ Email this page to a colleague
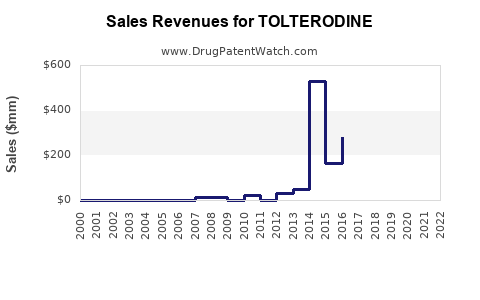
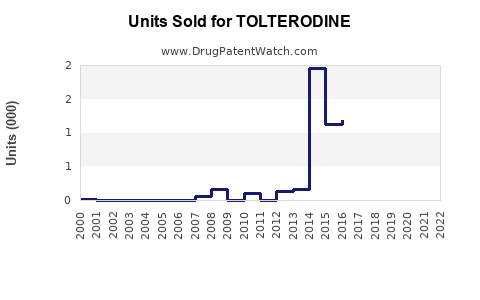
Annual Sales Revenues and Units Sold for TOLTERODINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TOLTERODINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Tolterodine
What is Tolterodine and How Is It Positioned in the Market?
Tolterodine is an antimuscarinic agent used primarily to treat overactive bladder (OAB). It was first approved by the FDA in 1998 as a prescription medication. The drug functions by reducing bladder muscle contractions, thereby alleviating urinary urgency, frequency, and incontinence.
Market positioning hinges on its efficacy and side effect profile, primarily compared to alternatives like oxybutynin, solifenacin, and darifenacin. Tolterodine comes in immediate-release (IR) and extended-release (ER) formulations, with the latter dominating the market due to better tolerability.
Current Market Size and Competitive Landscape
Global Market Size
- Estimated global overactive bladder medication market was worth approximately USD 2.8 billion in 2022.
- Compound annual growth rate (CAGR) from 2022 to 2028 projected at 7.4%[1].
Market Share Breakdown (2022)
| Drug Class | Market Share | Key Drugs |
|---|---|---|
| Antimuscarinics | 75% | Tolterodine, oxybutynin, solifenacin, darifenacin |
| Beta-3 Agonists | 20% | Mirabegron |
| Others | 5% | Botox, nerve stimulation devices |
Tolterodine holds roughly 15-20% of the antimuscarinic segment, with higher preference for its ER formulation.
Regional Distribution
- North America: 50%
- Europe: 30%
- Asia-Pacific: 15%
- Rest of the World: 5%
North American dominance derives from earlier adoption, extensive insurance coverage, and higher prevalence rates.
Sales Trends and Historical Data
In 2022, global sales of tolterodine approximated USD 450 million. North America accounted for over 70% of this revenue. Sales have plateaued since 2019, correlating with increased competition and the advent of beta-3 agonists.
Drivers of Growth
- Aging demographics increase OAB prevalence.
- Growing awareness and diagnosis rates.
- Introduction of ER formulations improve adherence.
Constraints on Sales Growth
- Competition from newer drugs with improved side effect profiles.
- Patent expirations for some formulations in 2015-2018.
- Regulatory challenges related to long-term safety data.
Future Sales Projections (2023-2028)
Assumptions
- Moderate market penetration maintained in North America and Europe.
- Incremental growth in Asia-Pacific due to increased healthcare access.
- Competition from beta-3 agonists limits market share expansion.
- Patent expirations for ER formulations will promote generic uptake.
Forecast Summary
| Year | Estimated Global Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 470 | Slight increase from prior year, driven by aging populations |
| 2024 | 505 | Market penetration stabilizes, generic entries increase |
| 2025 | 530 | Growth plateau, competition intensifies |
| 2026 | 560 | Slight growth due to increased diagnosis |
| 2027 | 590 | Market expansion in Asia-Pacific |
| 2028 | 620 | Continued growth, slight market share gains |
Cumulative sales over this period estimated at USD 3.325 billion, with North American sales representing roughly 70% of total.
Market Dynamics and Strategic Factors
Patent and Regulatory Trends
- Patent expiry for original formulations led to generic versions in late 2010s.
- Regulators focus on long-term safety, particularly regarding anticholinergic side effects.
- Emerging combination therapies may influence next-generation prescribing patterns.
Pricing Strategies
- Generic competition has driven prices downward.
- Pharmacoeconomic evaluations favor newer drugs, which may affect tolterodine’s pricing and sales.
R&D and Pipeline Developments
- No recent pipeline updates indicate significant new formulations.
- Focus remains on improving tolerability of existing drugs.
Implications for Stakeholders
For Manufacturers
- Focus on cost-effective generic production.
- Explore combination therapies or delivery mechanisms to differentiate.
For Investors
- Stable but declining market share expected due to generics ands newer therapies.
- Growth hinges on regional expansion and formulary inclusion.
For Healthcare Providers
- Tolterodine remains a first-line treatment in many regions.
- Shift towards beta-3 agonists may limit future prescribing.
Key Takeaways
- Tolterodine's global sales will likely grow modestly, reaching approximately USD 620 million by 2028.
- Market saturation leads to price pressures and increased generic competition.
- North America dominates sales, but Asian markets present growth opportunities.
- Patent expirations and the rise of beta-3 agonists impact market share.
- Innovation efforts are minimal, focusing mainly on improving existing formulations.
FAQs
Q1: How does tolterodine compare to newer overactive bladder treatments?
A1: Tolterodine offers comparable efficacy to newer antimuscarinics but has higher anticholinergic side effects, leading to lower adherence. Beta-3 agonists like mirabegron are increasingly preferred due to better tolerability.
Q2: What are the primary factors limiting tolterodine sales growth?
A2: Competition from beta-3 agonists, patent expirations leading to generics, and side effect profiles reduce its market appeal.
Q3: Are there any upcoming formulations or combinations in development?
A3: No significant pipeline developments are reported for tolterodine, with focus primarily on established formulations.
Q4: How significant is the impact of patent expiry on sales?
A4: Patent expiry around 2015-2018 triggered generic entry, leading to decreased unit prices and sales volumes.
Q5: What regional trends are expected for tolterodine sales?
A5: North American sales will remain dominant, but incremental growth is expected in Asia-Pacific regions with improved healthcare infrastructure.
References
[1] MarketsandMarkets. (2022). Overactive Bladder Market by Drug Class, Route of Administration, Distribution Channel, and Region – Global Forecast to 2028.
More… ↓
