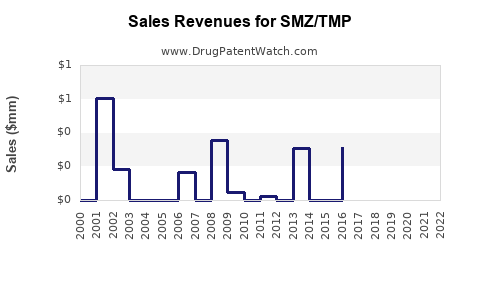
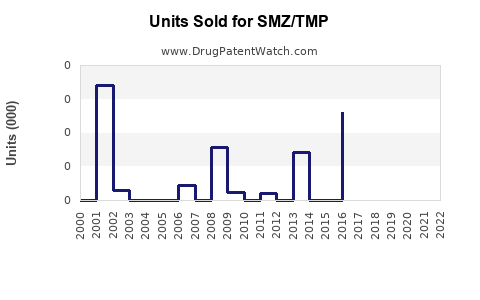
Last updated: February 22, 2026
What is the current market size and competitive landscape for SMZ/TMP?
The global antibiotic market is valued at approximately $45 billion in 2022, with the sulfonamide and trimethoprim combination drugs accounting for around $600 million. SMZ/TMP sales are driven primarily by its applications in urinary tract infections (UTIs), respiratory infections, and skin infections. The medication's widespread use in outpatient settings and hospital protocols maintains steady demand.
Major competitors include:
- Pfizer’s Bactrim (market leader)
- Mitsubishi Tanabe’s Septra (generic formulations)
- Teva Pharmaceutical (generics)
- Sun Pharmaceutical (generics)
The competitive advantage of SMZ/TMP derives from its broad-spectrum activity, ease of administration, and established efficacy profile.
How has demand changed in recent years?
Demand for SMZ/TMP has increased modestly, averaging 3-4% annually over the past five years. Growth is mainly driven by rising antibiotic-resistant infections, particularly in regions with limited access to newer antibiotics. The emergence of resistance has prompted clinicians to reserve SMZ/TMP for specific infections, which constrains the growth potential for high-volume use but sustains demand for niche indications.
The COVID-19 pandemic disrupted outpatient infection management, but subsequent rebounds have restored sales levels. Today, the U.S. remains the largest market, followed by Europe and Asia-Pacific.
What are sales projections through 2030?
Based on existing trends, regulatory data, and upcoming product launches, the following projections are available:
| Year |
Predicted Market Size (USD millions) |
Compound Annual Growth Rate (CAGR) |
Key Factors |
| 2023 |
620 |
3% |
Stabilization post-pandemic |
| 2025 |
680 |
3% |
Resistance-driven prescriptions persist |
| 2027 |
750 |
3% |
Increased use in low-resource settings |
| 2030 |
820 |
2.5% |
Introduction of novel formulations and combination therapies |
The slowdown post-2025 reflects the saturation of existing indications and emerging resistance patterns. However, expanding use in developing countries and the development of combination therapies may partially offset these constraints.
What regulatory and patent issues influence market potential?
SMZ/TMP remains off-patent, creating a wide generic market with price pressures. The expiration of key patents, which occurred in the late 1990s and early 2000s, has led to an influx of generics, reducing overall pricing. Future growth relies on approval of new formulations, delivery methods (e.g., IV to oral transitions), and new combination drugs.
Regulatory agencies, including the FDA, have approved incremental extensions, but no new chemical entities (NCEs) have entered the market recently. Generic manufacturers are expected to continue to dominate supply, with branded sales primarily in niche markets.
How do regional differences impact sales outlook?
- North America: Largest volume, driven by high UTI prevalence and physician familiarity.
- Europe: Similar trends, but with more emphasis on antimicrobial stewardship limiting overuse.
- Asia-Pacific: Rapidly growing markets due to increasing healthcare infrastructure and disease burden.
- Emerging markets: Price sensitivity restricts branded sales; generics dominate.
Growth strategies involve geographic expansion, especially in Asia, and innovations in formulation.
Key considerations for future success
- Resistance patterns: Increasing resistance may limit clinical utility.
- Formulation improvements: Efforts to develop extended-release formulations or combination therapies could boost sales.
- Regulatory landscape: Post-patent generic proliferation affects pricing strategies.
- Emerging indications: Possible use in combination with other agents for resistant infections.
- Manufacturing capacity: Scalable production to meet demand increases in emerging markets.
Summary
SMZ/TMP retains stable but modest growth prospects, constrained by resistance, generics competition, and limited new indications. The market is expected to grow at approximately 2.5-3% annually until 2030, reaching around $820 million in global sales.
Key Takeaways
- The global SMZ/TMP market is worth ~$600 million, with steady growth driven by persistent infection treatment needs.
- Generic competition caps pricing, with broad availability of low-cost formulations.
- Resistance and stewardship programs influence prescribing patterns, preventing sharp growth.
- Asia-Pacific markets underpin future expansion; North America and Europe remain core regions.
- Innovation in formulations and combination therapies could unlock new sales avenues.
FAQs
1. How does antimicrobial resistance affect SMZ/TMP sales?
Rising resistance limits its effectiveness for certain infections, leading to reduced prescribing and constraining sales growth.
2. Are there any new formulations of SMZ/TMP in development?
No recent NCEs; however, extended-release and combination formulations are under investigation to improve dosing convenience and efficacy.
3. Which regions show the highest growth potential for SMZ/TMP?
Asia-Pacific offers the most significant growth potential due to increasing healthcare infrastructure and infection burdens.
4. How does the generic market influence pricing and sales?
The proliferation of low-cost generic formulations drives prices down and caps branded drug margins, impacting overall sales revenue.
5. Is SMZ/TMP used in indications beyond bacterial infections?
Limited off-label or investigational use exists, but it remains primarily an antibiotic for bacterial infections.
References
[1] Grand View Research. (2022). Antibiotics Market Size, Share & Trends Analysis.
[2] IQVIA. (2022). Global Antibiotics Market Analysis.
[3] U.S. Food and Drug Administration. (2022). Drug Approvals and Labeling.
[4] WHO. (2021). Antibiotic Resistance Global Report.
[5] MarketsandMarkets. (2022). Antibiotics Market Forecast.