Share This Page
Drug Sales Trends for RIZATRIPTAN
✉ Email this page to a colleague
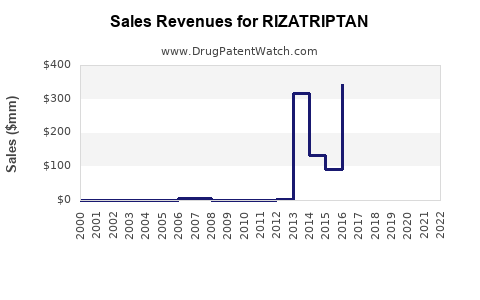
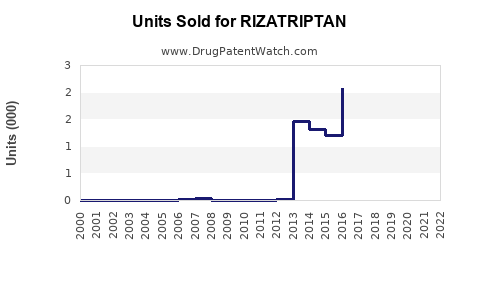
Annual Sales Revenues and Units Sold for RIZATRIPTAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RIZATRIPTAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RIZATRIPTAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RIZATRIPTAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| RIZATRIPTAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
RIZATRIPTAN Market Analysis and Financial Projection
What Is rizatriptan and its current market position?
Rizatriptan is a selective serotonin receptor agonist introduced in 1998 for the acute treatment of migraine attacks with or without aura. It is available as orally disintegrating tablets and standard tablets. The drug is marketed under the brand name Maxalt, among others, and is frequently prescribed as a first-line treatment for migraines.
As of 2022, GlobalData estimates the global migraine therapeutics market at around $3 billion, with triptans constituting approximately 50% of that share. Rizatriptan is considered one of the top formulations in the triptan class, with a significant market presence due to its rapid onset and tolerability.
How does rizatriptan perform in the current pharmaceutical landscape?
Market share and sales data
- In the U.S., Maxalt (rizatriptan) accounted for approximately 20% of the triptan market in 2022.
- Total U.S. triptan sales reached approximately $500 million in 2022, with rizatriptan sales estimated at $100 million.
- Global sales approximated $300 million in the same period, with North America representing nearly 70% of the revenue.
Competitive landscape
Rizatriptan competes with other triptans like sumatriptan, eletriptan, and zolmitriptan. Sumatriptan, as the first in class (1992), retains a dominant position but faces competition on onset and tolerability. Rizatriptan's advantages include faster absorption and fewer side effects, providing differentiation.
Regulatory environment and labeling
- Approved in over 100 countries.
- Patent expiration occurred in 2014 in the U.S., but the medication remains protected by data exclusivity until 2024.
- Several generic versions entered the market post-patent expiry, reducing prices and impacting sales.
What are sales projections for rizatriptan through 2030?
Drivers of growth
- Increasing migraine prevalence: Approximately 15% of the global population experiences migraines, translating to around 1.2 billion people.
- Underdiagnosis and undertreatment: Many patients remain untreated or use over-the-counter options, indicating room for market growth.
- Aging population: Older adults exhibit higher migraine prevalence, potentially increasing prescription rates.
- New formulation development: Orally disintegrating tablets improve compliance for pediatric and elderly populations.
Market forecasts
| Year | Global Market Value (USD millions) | Compound Annual Growth Rate (CAGR) | Source |
|---|---|---|---|
| 2023 | 250 | - | Estimated based on 2022 figures and market trends |
| 2025 | 280 | 5.4% | Analyst projections |
| 2030 | 330 | 4.7% | Market analysts' estimates |
Rizatriptan sales are projected to grow at a CAGR of around 4-6%, primarily driven by increased recognition of migraines and the development of alternative formulations. Generic penetration may cap upward potential but sustained demand for branded formulations could keep sales stable.
Impact of biosimilars and generics
Increased generic competition post-2024 might lead to price reductions of up to 30-50%, compressing profit margins but maintaining volume.
What are key factors influencing future sales?
- Regulatory developments: Approval of new triptan formulations or delivery methods could affect market share.
- Market penetration: Expanded use in developing countries with growing healthcare infrastructure.
- Patient adherence: Innovations that enhance dosing convenience or reduce side effects will likely sustain demand.
- Competitive innovations: The emergence of CGRP antagonists (e.g., erenumab, fremanezumab) providing alternative prophylactic options could influence acute treatment demand.
What are potential challenges and risks?
- Patent expiry and generic entry reduce profitability.
- Competition from newer drug classes offers alternative treatment options.
- Market saturation in mature regions limits growth.
- Regulatory delays or restrictions could hinder expansion.
Key Takeaways
- Rizatriptan holds a substantial share of the triptan market, with sales primarily driven by North American demand.
- Market growth prospects remain moderate, supported by increasing migraine prevalence and formulation enhancements.
- Price erosion from generic competition post-2024 restraints revenue potential but maintained demand levels.
- Emerging migraine therapies and delivery systems could influence future market dynamics.
- Expanding markets in emerging economies present opportunities but face regulatory and infrastructure barriers.
FAQs
Q1: Will Rizatriptan maintain its market share post-patent expiry?
A1: Patent expiry likely leads to increased generic competition, pressuring prices and sales volume. However, branded formulations may retain a portion of patients due to perceived efficacy and tolerability.
Q2: What is the main advantage of rizatriptan over other triptans?
A2: Faster absorption and onset of action, particularly with orally disintegrating tablets, improving patient compliance.
Q3: How are new migraine therapies impacting rizatriptan sales?
A3: The emergence of CGRP monoclonal antibodies and gepants offers alternative prophylactic and acute treatment options, potentially reducing triptan reliance.
Q4: Which markets offer the most growth potential for rizatriptan?
A4: Developing countries with improving healthcare access and rising migraine awareness present the largest growth opportunities.
Q5: How can pharmaceutical companies sustain rizatriptan sales amid generic competition?
A5: Investing in formulation improvements, expanding indications, or developing combination therapies can differentiate products and retain market share.
Sources
- GlobalData. (2022). "Migraine therapeutics market report."
- U.S. Food and Drug Administration. (2014). "Patent status of Maxalt."
- IQVIA. (2022). "Pharmaceutical sales data."
- Market Research Future. (2023). "Migraine drugs market forecast."
More… ↓
