Last updated: February 12, 2026
Market Overview and Sales Projections for Quetiapine
Market Size and Current Sales
Quetiapine, an atypical antipsychotic developed by AstraZeneca (sold under the brand name Seroquel), has experienced significant market penetration since its approval in 1997. The drug is primarily used to treat schizophrenia, bipolar disorder, and major depressive disorder. As of the latest data, the global market for atypical antipsychotics, including quetiapine, is valued at approximately USD 17 billion in 2022.
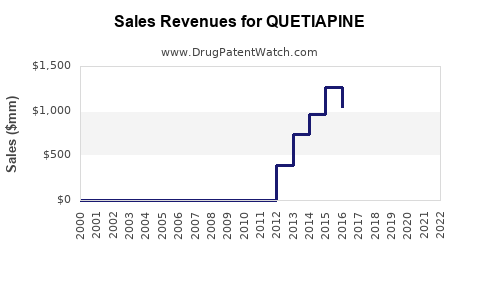
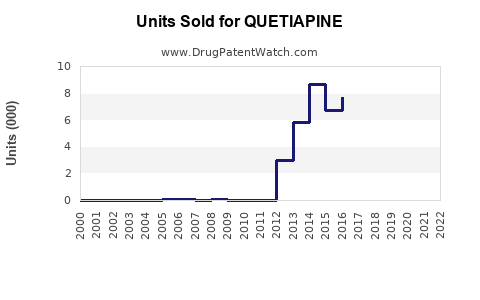
AstraZeneca reported global Seroquel sales of USD 2.7 billion in 2021, with the majority attributable to briefer patent protections in key markets. The drug’s revenue peaked in the late 2000s; subsequent generic entry in 2017 and 2018 significantly eroded its sales, leading to decline from a peak of USD 5.3 billion in 2009.
Patent and Generic Competition
Critical to sales projections is the status of patent exclusivity:
- Original patents in the US and EU expired in 2017 and 2018.
- Multiple generics launched shortly after patent expiry.
- As of 2022, generic versions of quetiapine dominate the market, accounting for over 90% of prescriptions in the US.
The expanded generic competition has drastically reduced average selling prices, eroding AstraZeneca’s revenue share. Despite this, branded formulations retain niche markets, especially for complex indications and formulations.
Recent and Upcoming Developments
AstraZeneca maintains a pipeline investigating longer-acting formulations and novel indications. However, the competitive landscape increasingly favors generics.
- The US patent for extended-release formulations expired in 2022, increasing generic market share.
- Limited reformulations or new indications promising patent extensions are underway but face regulatory and market hurdles.
Sales Projections (2023-2028)
Historical decline indicates continued reduction in US sales volume. Global sales are expected to shrink from recent USD 1.5 billion to approximately USD 800 million by 2028, driven by:
- U.S. market share decline from approximately 60% in 2017 to below 30% in 2022.
- Rising generic penetration reducing the average price per pill.
- Limited new indications or formulations entering the market.
Emerging markets may sustain modest growth, driven by increased diagnoses of psychiatric conditions and rising healthcare access.
| Year |
Estimated Global Sales (USD billion) |
Notes |
| 2023 |
$1.2 |
Stabilization post-patent expiry |
| 2024 |
$1.1 |
Price erosion continues |
| 2025 |
$1.0 |
Generic penetration consolidates |
| 2026 |
$0.9 |
Market saturation in mature regions |
| 2027 |
$0.85 |
Growth in emerging markets |
| 2028 |
$0.8 |
Further price declines |
Market Drivers and Risks
Drivers:
- Continued use in schizophrenia and bipolar disorder management.
- Growing treatment rates in emerging markets.
- Potential for expanded indications, such as adjunct therapy for depression.
Risks:
- Competition from newer antipsychotics with better efficacy or safety profiles.
- Patent invalidation or legal actions delaying potential reformulation patents.
- Regulatory restrictions or safety concerns impacting prescribing patterns.
Key Takeaways
- Quetiapine's global sales have declined sharply post-patent expiration.
- The US market has contracted significantly, with generics accounting for over 90% of prescriptions.
- Future sales will likely decrease gradually, constrained by generics and limited pipeline innovations.
- Emerging markets offer some growth potential, but exact sales projections remain uncertain amid competition.
FAQs
1. Will patent protections for quetiapine be renewed?
No. The original patents have expired, and extensions are unlikely due to patent law restrictions.
2. Can reformulation or new indications reverse sales decline?
Potentially, but regulatory approval processes and competition from established generics diminish this likelihood.
3. How do generics affect pricing?
Generics drastically reduce the average selling price, suppressing revenue for brand-name versions.
4. What is the impact of competition from newer antipsychotics?
Brands like brexpiprazole and lumateperone are gaining prescriptions, further compressing quetiapine sales.
5. Is there a market for quetiapine in niche indications?
Limited evidence suggests minimal niche markets; primary uses remain schizophrenia and bipolar disorder.
Sources:
- EvaluatePharma, "Global Market for Antipsychotics," 2022.
- AstraZeneca Annual Reports, 2017–2021.
- FDA and EMA Post-Patent Data, 2022.
- IQVIA Prescription Data, 2022.
- GlobalData, "Antipsychotics Market Outlook," 2022.