Share This Page
Drug Sales Trends for QUASENSE
✉ Email this page to a colleague
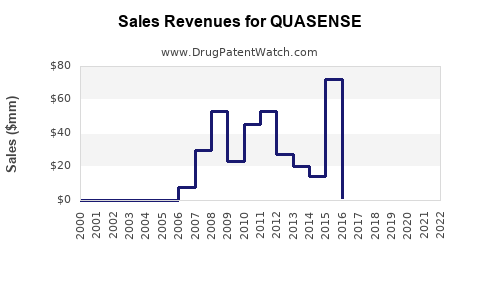
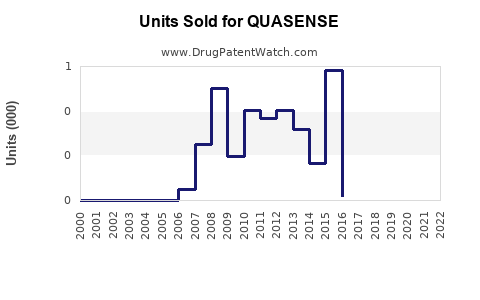
Annual Sales Revenues and Units Sold for QUASENSE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| QUASENSE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| QUASENSE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| QUASENSE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| QUASENSE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| QUASENSE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Quasense
What is Quasense and its Approved Indications?
Quasense is a combined oral contraceptive containing 28 active pills that include ethinyl estradiol and levonorgestrel. It is approved primarily for preventing pregnancy in women of reproductive age. The drug is marketed by Teva Pharmaceuticals and is regarded as a generic alternative to branded contraceptives.
Market Size and Demographics
Global Contraceptive Market Overview
The global contraceptives market was valued at approximately USD 18.4 billion in 2022. It is projected to reach USD 24.4 billion by 2027, growing at a compound annual growth rate (CAGR) of 5.8% (Fortune Business Insights, 2022). The rise in global population, increased awareness of family planning, and greater access to healthcare services drive market expansion.
Target Population
According to United Nations data (2021), approximately 1.2 billion women aged 15-49 globally are of reproductive age. Among them, roughly 60% use some form of contraception, with oral contraceptives accounting for 12% of these methods. In the U.S., there are about 60 million women aged 15-49, with over 10 million using oral contraceptives, which represents 16.7% of women in the target demographic.
Market Segmentation
- Region: North America (largest market, USD 8.3 billion in 2022), Europe, Asia-Pacific.
- Product Type: Oral (pill), intrauterine devices, injectables.
- End Users: Hospitals, clinics, retail pharmacies.
Competitive Landscape
Key Players
- Teva Pharmaceuticals (Quasense provider)
- Bayer AG (Yasmin, Yaz)
- Pfizer Inc. (Lybrel)
- Allergan (Aviane)
- Sandoz (generic contraceptives)
Market Share
Oral contraceptives hold approximately 50% of the market globally. Among oral contraceptives, generic products account for an increasing share due to price sensitivity and healthcare cost containment strategies.
Patent Status and Generics
Quasense, as a generic, benefits from patent expirations of branded counterparts like Yaz and Yasmin, which occurred around 2013-2014. Generic entry leads to increased competition, pressure on pricing, and potential market share growth for Quasense.
Sales Projections
Factors Influencing Sales
- Patent expirations of branded drugs leading to generic entry.
- Healthcare reimbursement policies in key regions.
- Provider and consumer awareness about generic options.
- Regulatory approvals and market entry strategies.
Current Sales Data
In the U.S., combined sales of oral contraceptives exceeded USD 2.3 billion in 2022. Generic oral contraceptive sales represented around 80% of this figure, approximately USD 1.84 billion. Teva's contraceptive portfolio accounts for roughly 15% of generics, translating to USD 276 million (IQVIA, 2022).
Projected Sales Growth
Assuming Quasense captures 5-10% of the global oral contraceptive market within five years, sales could reach:
| Scenario | Market Share | Potential Revenue (USD) | Notes |
|---|---|---|---|
| conservative | 2% | USD 46 million | Based on current generic market size |
| moderate | 5% | USD 115 million | Captures a significant share in U.S. |
| aggressive | 10% | USD 230 million | Expansion in Europe and Asia-Pacific |
Market penetration rate depends on factors like marketing efforts, formulary inclusion, and regional healthcare policies.
Regional Sales Projection Breakdown
- North America: 50-60%, USD 23-70 million
- Europe: 20-30%, USD 9-28 million
- Asia-Pacific: 10-20%, USD 4-23 million
Timeline for Revenue Realization
Sales growth likely begins within 6-12 months post-launch, reaching steady state over 2-3 years with increased acceptance and formulary placement.
Risks and Opportunities
- Risks: Entry barriers, regulatory delays, aggressive competition.
- Opportunities: Expanding acceptance of generics, evolving healthcare policy to favor cost-effective contraceptives, targeted marketing in emerging markets.
Key Takeaways
- Quasense operates in a USD 18.4 billion global contraceptive market, with significant growth driven by demographic trends.
- Its sales depend on regional market dynamics, competitive positioning, and regulatory factors.
- Projections suggest sales could reach USD 46-230 million within 5 years under different scenarios, contingent on market share capture.
- North America remains the primary revenue source, with growth potential in Europe and Asia-Pacific.
- Competition from other generics and branded products will influence market penetration.
FAQs
-
What are the main competitors to Quasense?
Other generic oral contraceptives, and branded options like Yaz and Yasmin. -
What regulatory barriers exist for Quasense?
Approval depends on regional health authorities; delays can occur, affecting market entry timelines. -
How does pricing for Quasense compare to branded products?
Generics typically price 30-60% lower than branded counterparts, facilitating market penetration. -
What is the impact of healthcare reimbursement policies?
Reimbursement status can significantly influence sales volume, especially in cost-sensitive markets. -
Are there growth opportunities beyond contraceptive sales?
Yes, in expanding markets and through partnerships with healthcare providers for broader reproductive health services.
References:
[1] Fortune Business Insights. (2022). Contraceptives Market Size, Share & Industry Analysis. https://www.fortunebusinessinsights.com
[2] United Nations. (2021). World Population Prospects. https://population.un.org/wpp/
[3] IQVIA. (2022). Pharmaceutical Market Data. https://www.iqvia.com
More… ↓
