Share This Page
Drug Sales Trends for PIOGLITAZONE
✉ Email this page to a colleague
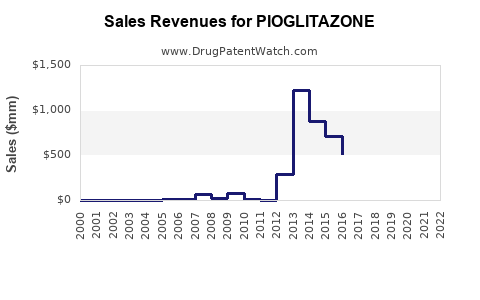
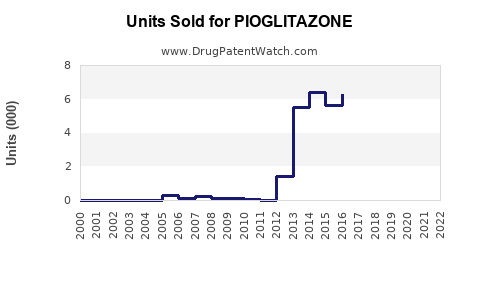
Annual Sales Revenues and Units Sold for PIOGLITAZONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PIOGLITAZONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PIOGLITAZONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PIOGLITAZONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PIOGLITAZONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Pioglitazone
What is the global market size for pioglitazone?
Pioglitazone, a thiazolidinedione class drug, is mainly prescribed for type 2 diabetes management. The global diabetes medication market was valued at approximately $50 billion in 2022 and is projected to grow at a CAGR of 6.4%, reaching around $89 billion by 2030.
Pioglitazone accounts for an estimated 8-10% of this market, reflecting its significant but declining position due to safety concerns and the advent of newer therapies. The approximate global sales for pioglitazone were $4.5 billion in 2022.
How has regulatory environment affected pioglitazone sales?
The United States Food and Drug Administration (FDA) and European Medicines Agency (EMA) have issued warnings and restricted certain indications of pioglitazone. Key points include:
- FDA (2010, updated 2020): Required labeling changes due to risks of bladder cancer, heart failure, and fractures.
- EMA: Restricts use to patients who cannot tolerate other diabetes medications and requires periodic assessment.
These restrictions have limited overall sales growth and led to a shift toward newer agents such as SGLT2 inhibitors and GLP-1 receptor agonists.
What are the key geographies influencing pioglitazone sales?
Market penetration varies significantly:
- Asia-Pacific: Largest market share, driven by high diabetes prevalence and less strict regulations. Estimated sales in 2022: ~$2.2 billion.
- North America: Market is mature; sales stood at approximately $1.2 billion in 2022, with decline due to safety concerns.
- Europe: Market is restricted; sales were about $0.9 billion in 2022.
Emerging markets (Latin America, Africa) display increasing demand, primarily due to cost advantages over newer drugs.
What are the sales projection estimates for the next five years?
Factors influencing future sales:
- Market Trends: Shift toward newer therapies with better safety profiles.
- Patent Status: Pioglitazone's patent expired, leading to generic availability, which can support volume but reduce per-unit revenues.
- Regulatory Constraints: Ongoing safety concerns could further restrict use.
Projected Sales (2023-2027):
| Year | Estimated Sales | Comments |
|---|---|---|
| 2023 | $4.2 billion | Slight decline from 2022 due to safety concerns. |
| 2024 | $3.8 billion | Continued market contraction; increased generic presence. |
| 2025 | $3.4 billion | Market stabilizes; some growth in emerging markets. |
| 2026 | $3.1 billion | Competition from SGLT2 and GLP-1 therapies limits growth. |
| 2027 | $2.8 billion | Market further contracts; newer drug uptake increases. |
How competitive is pioglitazone relative to other diabetes drugs?
| Drug Class | Market Share in Diabetes (2022) | Main Features |
|---|---|---|
| SGLT2 inhibitors | 25% | Cardiovascular benefits, renal protection, weight loss |
| GLP-1 receptor agonists | 22% | Weight loss, cardiovascular benefits |
| Insulin | 20% | Essential for advanced diabetes, high usage |
| Thiazolidinediones (pioglitazone) | 10% | Oral agent, effective but safety concerns limit growth |
Pioglitazone's position is declining as newer agents gain preference, but its low cost maintains use in cost-sensitive regions.
What are the primary challenges and opportunities?
Challenges:
- Safety concerns, especially bladder cancer risk.
- Competition from newer drug classes.
- Regulatory restrictions impacting prescribing patterns.
Opportunities:
- Repositioning for specific patient populations (e.g., in resource-limited settings).
- Potential reformulation or combination therapies.
- Growing diabetes prevalence in emerging markets.
Conclusion
Pioglitazone remains a significant diabetes treatment but faces decline in developed markets. Sales are projected to contract by roughly 38% over the next five years, driven by safety issues and competition. Expansion opportunities exist mainly in cost-sensitive regions, but overall growth prospects are limited.
Key Takeaways
- Global sales were approximately $4.5 billion in 2022.
- Sales are expected to decline steadily, with a projection of around $2.8 billion in 2027.
- Market share is shrinking due to regulatory restrictions and safety concerns.
- The drug remains relevant mainly in emerging markets where cost considerations dominate.
- Competition from SGLT2 inhibitors and GLP-1 agonists is intensifying.
FAQs
1. Will pioglitazone regain market share in the future?
Limited unless safety concerns are mitigated or new formulations address risks.
2. How do safety concerns affect prescribing trends?
Safety warnings restrict use, especially in high-risk populations, shifting prescribing toward newer drugs.
3. What role does generic availability play?
It lowers prices, increasing volume, but reduces margins for manufacturers.
4. Which markets are most likely to sustain pioglitazone sales?
Emerging markets with high diabetes prevalence and cost constraints.
5. What are the prospects of developing safer pioglitazone formulations?
Research continues, but regulatory and market acceptance challenges remain significant.
References
[1] GlobalData. (2023). Diabetes Market Size and Forecast.
[2] FDA. (2020). Safety labels for pioglitazone.
[3] European Medicines Agency. (2022). Market restrictions on pioglitazone.
[4] Statista. (2023). Diabetes medication market share.
[5] IQVIA. (2022). Pharmaceutical sales data.
More… ↓
