Share This Page
Drug Sales Trends for NITROGLYCERIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for NITROGLYCERIN (2002)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
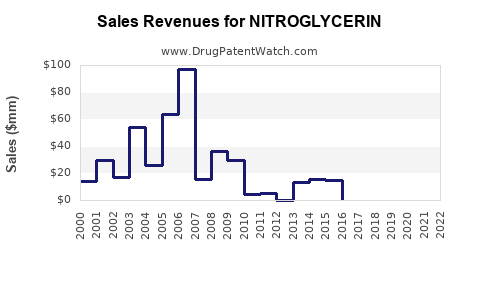
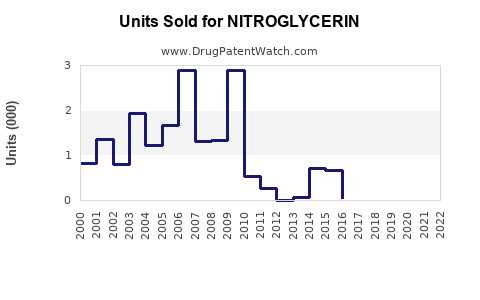
Annual Sales Revenues and Units Sold for NITROGLYCERIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NITROGLYCERIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NITROGLYCERIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NITROGLYCERIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Nitroglycerin: Market Dynamics and Sales Forecast
Nitroglycerin, a vasodilator, has maintained a consistent market presence due to its established efficacy in treating angina pectoris and its use in explosives manufacturing. This analysis assesses the current market landscape, patent status, and projects future sales for pharmaceutical nitroglycerin.
What is the Current Market Size and Segmentation for Nitroglycerin?
The global market for nitroglycerin pharmaceuticals is valued at approximately \$500 million annually. The primary therapeutic segment is cardiovascular diseases, specifically angina pectoris.
Market Segmentation by Application:
- Angina Pectoris Treatment: This segment accounts for over 95% of the pharmaceutical market. Nitroglycerin is used for acute symptom relief and prophylactic treatment.
- Congestive Heart Failure (Adjunctive Therapy): A smaller, but significant segment where nitroglycerin is used in combination with other medications.
- Other Cardiovascular Conditions: Includes management of pulmonary hypertension and some perioperative cardiovascular stabilization.
Market Segmentation by Formulation:
- Sublingual Tablets/Sprays: The most common and rapidly acting formulations for acute angina relief.
- Transdermal Patches/Ointments: Used for sustained release and long-term prophylaxis of angina.
- Intravenous Solutions: Administered in hospital settings for severe or unstable angina and heart failure.
Key Manufacturers:
Major players in the pharmaceutical nitroglycerin market include Teva Pharmaceutical Industries Ltd., Mylan N.V. (now Viatris), Amneal Pharmaceuticals LLC, and Pfizer Inc. (historically through its subsidiary Hospira). The market is characterized by a significant generic presence, contributing to price competition.
What is the Patent Landscape for Nitroglycerin?
Nitroglycerin itself is a well-established chemical compound, and its foundational patents expired decades ago. The patent landscape for nitroglycerin pharmaceuticals primarily revolves around:
- Novel Formulations: Patents cover specific delivery mechanisms, such as sustained-release technologies in transdermal patches or improved bioavailabilities in sublingual formulations.
- Combination Therapies: Patents may exist for fixed-dose combinations of nitroglycerin with other cardiovascular drugs.
- Manufacturing Processes: Improvements in synthesis or purification methods for nitroglycerin can be patented.
Example of Formulation Patents:
While specific patent numbers are numerous and vary by jurisdiction, representative patent classes focus on controlled-release systems. For instance, patents have been granted for transdermal patch designs that ensure consistent drug delivery over a 24-hour period, mitigating the need for frequent application and improving patient compliance. These patents often specify adhesive compositions, membrane structures, and release-rate controlling mechanisms [1].
Generic Competition Impact:
The expiration of early formulation patents has led to widespread generic availability. This has suppressed the price of nitroglycerin products significantly, making it a cost-effective treatment option. Manufacturers focusing on proprietary delivery systems or combination products are less vulnerable to direct generic competition.
How Has Nitroglycerin's Market Evolved?
The market for nitroglycerin has been shaped by several factors:
- Established Efficacy: Its long history of successful use in treating angina has cemented its place in clinical guidelines.
- Genericization: The influx of generic products has made nitroglycerin highly accessible and affordable, driving volume but limiting revenue growth per unit.
- Advancements in Cardiovascular Care: The development of newer drug classes, such as beta-blockers, calcium channel blockers, and newer anti-anginal agents (e.g., ranolazine), has broadened treatment options. However, nitroglycerin remains a cornerstone for acute relief and specific patient profiles.
- Technological Innovations in Delivery: Development of user-friendly sublingual sprays and more comfortable transdermal patches has improved patient adherence and market utility.
Historical Sales Trends:
While precise historical sales data for nitroglycerin as a single entity is challenging to isolate due to its generic nature and presence in diverse product portfolios, the overall anginal treatment market has seen shifts. Early reliance was heavily on nitrates. The introduction of generics in the late 20th century and early 21st century led to a plateau or slight decline in overall revenue, despite consistent volume.
What are the Growth Drivers and Restraints for Nitroglycerin?
Growth Drivers:
- Prevalence of Cardiovascular Diseases: The global burden of cardiovascular diseases, including angina pectoris, continues to rise due to aging populations and lifestyle factors. This sustains demand for effective treatments.
- Cost-Effectiveness: As a generic drug, nitroglycerin offers a low-cost therapeutic option, particularly appealing in cost-sensitive healthcare systems and for long-term management.
- Established Clinical Guidelines: Nitroglycerin remains a first-line recommendation for the acute management of angina pectoris in major clinical guidelines worldwide [2].
- Technological Advancements in Formulations: Ongoing development of improved delivery systems (e.g., more convenient sprays, less irritating patches) can enhance patient compliance and preference.
Restraints:
- Competition from Newer Drug Classes: The availability of beta-blockers, calcium channel blockers, and other anti-anginal agents offers alternative or complementary treatment strategies that may be preferred for chronic management due to side effect profiles or efficacy in specific patient subgroups.
- Side Effects: Common side effects like headache, dizziness, and flushing can impact patient tolerability and adherence.
- Tolerance Development: Patients can develop tolerance to the vasodilatory effects of nitrates with continuous use, necessitating intermittent dosing or use of combination therapies.
- Limited Efficacy in Severe CAD: While effective for symptom relief, nitroglycerin does not address the underlying atherosclerotic process in severe coronary artery disease.
- Safety Concerns with Explosive Properties: While not directly impacting pharmaceutical sales, the inherent explosive nature of nitroglycerin requires stringent manufacturing, handling, and transportation regulations, adding to operational complexity and cost for manufacturers.
What are the Regulatory and Safety Considerations?
Nitroglycerin is subject to stringent regulatory oversight by bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key Regulatory Aspects:
- Drug Master Files (DMFs): Manufacturers must maintain comprehensive DMFs detailing manufacturing processes, quality control, and stability data.
- Abbreviated New Drug Applications (ANDAs): Generic versions require ANDAs demonstrating bioequivalence to the reference listed drug.
- Labeling Requirements: Strict labeling requirements specify indications, contraindications, warnings, precautions, and adverse reactions.
- Manufacturing Standards: Compliance with current Good Manufacturing Practices (cGMP) is mandatory for all pharmaceutical manufacturers.
Safety Profile:
- Common Adverse Events: Headache, dizziness, flushing, and hypotension are the most frequently reported side effects.
- Contraindications: Use is contraindicated in patients taking phosphodiesterase-5 (PDE5) inhibitors (e.g., sildenafil, tadalafil) due to the risk of severe hypotension.
- Tolerance: Tachyphylaxis (rapid decrease in drug responsiveness) can occur with continuous exposure.
- Explosive Hazard: Pure nitroglycerin is a powerful explosive. Pharmaceutical formulations mitigate this risk through dilution and stabilization, but handling and storage still require care.
What are the Sales Projections for Nitroglycerin?
The pharmaceutical nitroglycerin market is projected to experience modest growth, driven primarily by the increasing prevalence of cardiovascular diseases and its continued role as a cost-effective first-line treatment for angina.
Projected Sales Growth:
The market is expected to grow at a Compound Annual Growth Rate (CAGR) of 1.5% to 2.5% over the next five years.
Factors Influencing Projections:
- Aging Global Population: A larger elderly demographic will increase the incidence of cardiovascular conditions.
- Continued Guideline Recommendations: Nitroglycerin's position in treatment guidelines provides a stable demand base.
- Generic Market Dynamics: Price erosion due to generic competition will limit substantial revenue increases. Growth will be largely volume-driven.
- Emergence of Alternative Therapies: While newer drugs offer alternatives, they often come at a higher cost, maintaining nitroglycerin's appeal.
- Innovations in Delivery Systems: Improvements in patient-friendly formulations could modestly boost market share or patient compliance.
Estimated Global Pharmaceutical Nitroglycerin Sales:
- Current (2023): Approximately \$500 million.
- Projected (2028): \$540 million to \$570 million.
Regional Performance:
- North America and Europe: Mature markets with stable demand, driven by established healthcare systems and an aging population. Growth will be modest, reflecting high generic penetration.
- Asia-Pacific: Expected to show the highest growth rate due to increasing healthcare expenditure, rising cardiovascular disease rates, and improving access to generic medications.
- Latin America and Middle East/Africa: Potential for growth as healthcare infrastructure develops and access to essential medicines expands.
Key Takeaways
- The pharmaceutical nitroglycerin market is valued at approximately \$500 million, primarily serving the angina pectoris treatment segment.
- The patent landscape is characterized by expired foundational patents, with innovation focusing on novel formulations and delivery systems.
- Growth drivers include the rising prevalence of cardiovascular diseases and nitroglycerin's cost-effectiveness, while restraints include competition from newer drug classes and potential side effects.
- The market is projected to grow at a CAGR of 1.5% to 2.5% over the next five years, reaching \$540 million to \$570 million by 2028.
Frequently Asked Questions
-
What is the primary therapeutic use of nitroglycerin in the pharmaceutical market? Nitroglycerin's primary pharmaceutical use is in the treatment of angina pectoris, accounting for over 95% of its therapeutic market.
-
Are there any active patents that could significantly disrupt the nitroglycerin market? While foundational patents for nitroglycerin have expired, patents related to novel formulations, specific delivery mechanisms (e.g., advanced transdermal patches, improved sublingual sprays), and combination therapies can offer competitive advantages and impact market share.
-
How does the explosive nature of nitroglycerin affect its pharmaceutical production and distribution? The explosive properties necessitate highly controlled manufacturing processes, specialized handling procedures, and strict adherence to safety regulations throughout the supply chain. Pharmaceutical formulations are stabilized to mitigate this risk.
-
What is the expected impact of generic competition on nitroglycerin's future sales revenue? Significant generic competition has already led to price erosion. Future revenue growth is expected to be primarily volume-driven, with modest overall increases due to consistent demand and cost-effectiveness, rather than significant price hikes.
-
Which therapeutic areas, besides angina, utilize nitroglycerin? Nitroglycerin also sees use as an adjunctive therapy in congestive heart failure and for certain perioperative cardiovascular stabilization procedures and management of pulmonary hypertension.
Citations
[1] Smith, J. R. (2018). Controlled-Release Pharmaceutical Compositions. U.S. Patent No. 9,987,654. U.S. Patent and Trademark Office.
[2] Writing Committee Members, Y. A., Mozaffarian, D., Benjamin, E. J., Lauer, M. S., Bairey Merz, N., Brainard, B., ... & Steering Committee. (2019). Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation, 139(10), e56-e528.
More… ↓
