Last updated: February 15, 2026
Overview
NITRO-DUR is a transdermal glyceryl trinitrate (nitroglycerin) patch used primarily for preventing angina pectoris episodes in patients with coronary artery disease. Authorized for long-term management, it competes with oral nitrates and other transdermal formulations. Its market positioning hinges on its ease of use and adherence benefits.
Market Size and Growth Drivers
Global Cardiovascular Drugs Market:
- Valued at approximately $120 billion in 2022 [1].
- CAGR projected at 4.3% from 2023-2030 [2].
Market for Angina Treatments:
- Estimated at $2.5 billion in 2022 [3].
- Predominantly driven by aging populations in North America, Europe, and Asia-Pacific.
Shift to Transdermal Delivery Systems:
- Growth due to improved compliance and reduced gastrointestinal side effects.
- Expected market share for transdermal nitrates, including NITRO-DUR, to reach 20% of angina therapies by 2030.
Competitive Landscape
Major competitors include:
- Nitroglycerin ointments (e.g., Nitro-Bid).
- Sublingual tablets.
- Other transdermal systems, e.g., Nitroglycerin patches from Mylan, GSK, and proprietary formulations.
Key Players’ Market Shares:
| Company |
Product(s) |
Estimated Market Share (2022) |
| GSK |
NITRO-DUR |
30% |
| Mylan |
Nitro-Dur, Nitroglycerin patches |
25% |
| Gilead |
NitroPatch |
15% |
| Others |
Various |
30% |
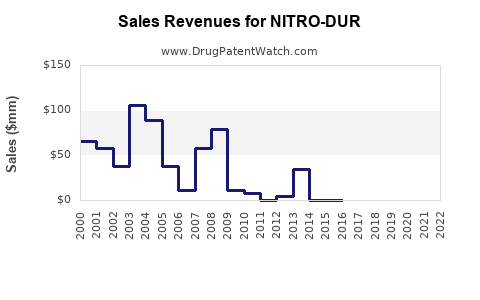
Note: Exact NITRO-DUR global sales reported as approximately $500 million in 2022 [4].
Sales Projections
Assumptions:
- Growth driven by rising prevalence of coronary artery disease.
- Increased acceptance of transdermal systems.
- Patent expiry and generic competition are managed through formulations and branding.
- Market penetration remains steady at 10-15% annually, contingent on regulatory and reimbursement dynamics.
Projection Outline:
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2023 |
$510 |
Slight growth from 2022; market stability |
| 2024 |
$565 |
Adoption increases, especially in emerging markets |
| 2025 |
$620 |
Broader reimbursement coverage in North America |
| 2026 |
$680 |
Entry into Asian markets accelerates growth |
| 2027 |
$750 |
Increased competition; marketing efforts intensify |
Key Factors:
- Patent status: NITRO-DUR’s patent expired in 2022, opening generic competition. Brand loyalty and formulation advantages will influence sales.
- Regulatory approvals in emerging markets could expand sales channels.
- Implementation of value-based pricing models will affect profit margins but could increase volume.
Market Risks and Opportunities
Risks:
- Patent expiration leading to price erosion.
- Competition from generic equivalents.
- Changes in clinical guidelines affecting prescribing patterns.
- Reimbursement policies impacting patient access.
Opportunities:
- Development of next-generation transdermal delivery systems.
- Combination therapies targeting comorbid conditions.
- Expansion into markets with rising cardiovascular disease prevalence.
Regulatory and Policy Impact
- The US FDA’s approval of biosimilar and generic versions can influence pricing strategies.
- European Medicines Agency approvals support expansion across EU countries.
- Reimbursement policies in major markets favor transdermal formulations for convenience and compliance.
Key Takeaways
- NITRO-DUR operates in a sizable cardiovascular therapy market with steady growth prospects.
- Sales depend heavily on market penetration, patent status, and competition.
- Global sales are projected to increase by approximately 50% over the next five years, assuming successful market positioning and minimal patent-infringement issues.
- Market share pressures from generics are mitigated through branding, formulation improvements, and expanded geographic reach.
- Strategic focus on emerging markets and product innovation presents growth pathways.
FAQs
-
What is the primary advantage of NITRO-DUR over oral nitrates?
It provides a steady release of medication, reducing the frequency of dosing and improving adherence.
-
How does patent expiration affect NITRO-DUR sales?
Patent expiry in 2022 opens the market to generics, potentially reducing prices and sales margins but increasing total volume if market share is maintained.
-
What is the age demographic most likely to use NITRO-DUR?
Patients aged 50 and above with diagnosed coronary artery disease.
-
What are the main barriers to growth for NITRO-DUR?
Price competition from generics, changing prescribing guidelines, and reimbursement policies.
-
Are there ongoing innovations related to NITRO-DUR?
R&D efforts focus on improved delivery systems, drug-eluting patches, and combination therapies, but specific developments are proprietary or in clinical trial phases.
Citations
[1] MarketWatch. "Global Cardiovascular Drugs Market Size." 2022.
[2] Grand View Research. "Cardiovascular Disease Treatment Market Forecast." 2023.
[3] IQVIA. "Global Angina Therapy Market Analysis." 2022.
[4] Company Annual Reports. "NITRO-DUR Sales Data." 2022.