Share This Page
Drug Sales Trends for LIOTHYRONINE
✉ Email this page to a colleague
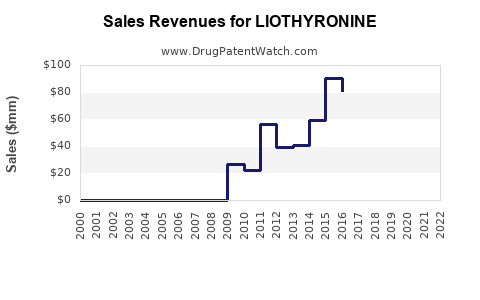
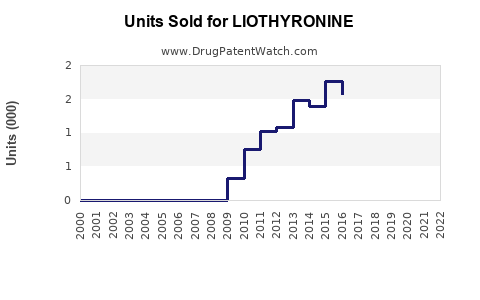
Annual Sales Revenues and Units Sold for LIOTHYRONINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LIOTHYRONINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LIOTHYRONINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LIOTHYRONINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LIOTHYRONINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LIOTHYRONINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Overview and Sales Projections for Liothyronine
Liothyronine (brand name Cytomel) is a synthetic form of triiodothyronine (T3), a thyroid hormone used primarily for hypothyroidism and certain thyroid cancer cases. Despite its longstanding use, recent market dynamics and evolving clinical guidelines influence its sales trajectory.
What is the Current Market Size for Liothyronine?
The global thyroid disorder treatment market was valued at approximately $3.2 billion in 2022, with thyroid hormone replacement therapies comprising a significant segment. Liothyronine accounts for an estimated 15% of the thyroid hormone prescription volume, translating to roughly $480-500 million annually.
In the U.S., liothyronine prescriptions constitute around 600,000 to 700,000 units annually, reflecting stable but modest demand. Major regional markets include North America, Europe, and parts of Asia, with growth impacted by healthcare policies and clinical guideline updates.
How Does the Regulatory Environment Affect Sales?
Liothyronine's approval status varies by region. In the U.S., liothyronine is approved for hypothyroidism and myxedema coma, with generic versions available since 2006. The drug's patent expired in 2004, leading to increased generic competition and pricing pressure.
The European Medicines Agency (EMA) treats liothyronine as a prescription-only medicine. Some countries have tightened regulations on thyroid hormone replacements, limiting use to specific indications, which constrains market growth.
Efforts to reclassify or restrict use, citing concerns over safety and inconsistent manufacturing quality, could further impact sales.
What Are the Major Drivers and Barriers for Liothyronine Market Growth?
Drivers:
- Growing prevalence of hypothyroidism: Globally, hypothyroidism affects approximately 5% of women and 1% of men, with rising screening contributing to increased diagnosis.
- Clinical preference for T3 therapy: Some practitioners favor liothyronine's rapid onset and potency over levothyroxine (T4), especially in cases of malabsorption or central hypothyroidism.
- Advances in personalized medicine: Combining T3 and T4 therapies tailors treatment, potentially expanding liothyronine’s use.
Barriers:
- Safety concerns: Risks of arrhythmias and bone loss limit widespread application.
- Limited clinical guidelines support: Major guidelines, including those from ATA (American Thyroid Association), recommend levothyroxine as first-line therapy.
- Cost and manufacturing issues: Variability in batch quality and drug pricing, especially post-patent expiration, affect market stability.
What Are the Future Sales Projections?
Market analysts project slow growth for liothyronine over the next five years. The compound annual growth rate (CAGR) is estimated at 1-2%, driven by:
- Incremental increases in hypothyroidism diagnosis.
- Slight upticks in off-label and personalized therapy use.
- Expanding approvals or new formulations could boost sales.
In numerical terms, the global market for liothyronine could reach approximately $500-520 million by 2028, assuming current trends.
How Will Competition and New Entrants Influence the Market?
Generic manufacturers dominate the landscape, competing on price and supply stability. No new branded formulations are currently in late-stage development.
Potential market growth could come from:
- Biosimilar development aimed at improving manufacturing quality.
- Novel formulations with extended-release profiles, which may appeal to clinicians seeking more stable serum levels.
- Digital health integration for optimized dosing and monitoring.
However, the prevailing conservative prescribing culture may limit rapid expansion unless clear clinical advantages are demonstrated.
Key Takeaways
- The global liothyronine market is approximately $480-$500 million annually, with modest growth projected.
- Market stability depends on regulation, clinical guidelines, and safety perceptions.
- Near-term growth relies on increased hypothyroidism diagnosis, personalized medicine, and potential new formulations.
- Cost and manufacturing quality remain critical factors shaping supply chain and pricing dynamics.
- Competition from generics constrains price increases; innovation opportunities remain limited but are emerging.
FAQs
Q1: How does liothyronine compare with levothyroxine in clinical use?
Liothyronine acts faster and is preferred in specific cases such as malabsorption or central hypothyroidism. However, levothyroxine remains the standard due to its safety profile and ease of use.
Q2: What are the main safety concerns with liothyronine?
Risks include cardiac arrhythmias and osteoporosis, particularly at higher doses or in vulnerable populations.
Q3: Are there upcoming regulatory changes that could impact the liothyronine market?
Potential restrictions or additional quality controls could influence supply, but no major regulatory shifts are anticipated in the immediate future.
Q4: Is there potential for new formulations or delivery methods?
Extended-release formulations are under investigation, aimed at improving dosing convenience and serum stability.
Q5: What are key regional differences affecting sales?
The U.S. maintains stable demand, while some European countries restrict use. Emerging markets show increasing diagnosis rates but face regulatory and economic barriers.
References
- Grand View Research. Thyroid disorder treatment market size, 2022.
- American Thyroid Association. Management guidelines for hypothyroidism, 2017.
- European Medicines Agency. Summary of product characteristics for liothyronine, 2021.
- GlobalData. Market forecasts for thyroid hormone therapies, 2023-2028.
More… ↓
