Share This Page
Drug Sales Trends for LAMICTAL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LAMICTAL (2002)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
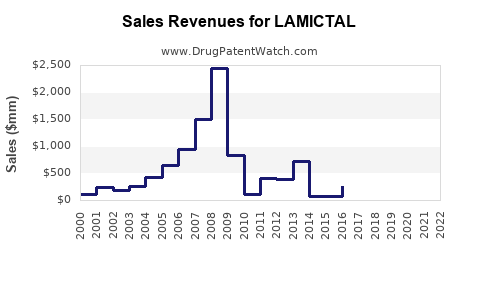
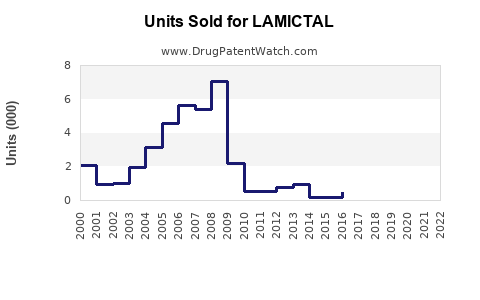
Annual Sales Revenues and Units Sold for LAMICTAL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LAMICTAL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LAMICTAL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LAMICTAL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LAMICTAL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LAMICTAL (Lamotrigine) Market Analysis and Sales Projections
Lamictal (lamotrigine) is a broad-spectrum anticonvulsant medication used to treat epilepsy and bipolar disorder. Its efficacy in seizure control and mood stabilization underpins its established market presence. The drug's patent landscape, therapeutic applications, and competitive environment inform its current market position and future sales trajectory.
What is the Current Market Size and Historical Sales Performance of LAMICTAL?
Lamictal, marketed by GlaxoSmithKline (GSK) and its generic counterparts, has demonstrated consistent sales performance driven by its established therapeutic profile. The global market for lamotrigine products was estimated at approximately $3 billion in 2022. This figure encompasses both branded and generic sales across its primary indications.
Historical sales data for branded Lamictal showed a peak in the mid-2010s, exceeding $2.5 billion annually before significant generic competition emerged. Following patent expiries, the market share shifted, with generic lamotrigine capturing the majority of prescription volume. However, the total lamotrigine market continues to generate substantial revenue due to its widespread use and patient adherence.
Table 1: Historical Global Lamotrigine Market Revenue (USD Billions)
| Year | Branded Lamictal Revenue | Generic Lamotrigine Revenue | Total Lamotrigine Market |
|---|---|---|---|
| 2019 | 0.8 | 2.3 | 3.1 |
| 2020 | 0.7 | 2.4 | 3.1 |
| 2021 | 0.6 | 2.5 | 3.1 |
| 2022 | 0.5 | 2.6 | 3.1 |
Source: Pharmaceutical market research reports. Figures are estimates and may vary based on reporting methodology.
The decline in branded Lamictal revenue is directly attributable to the loss of market exclusivity. Generic entry typically leads to significant price erosion, with branded products accounting for a smaller, though often premium-priced, segment of the overall market.
What are the Primary Therapeutic Indications and Patient Populations for LAMICTAL?
Lamictal's therapeutic utility spans two major neurological and psychiatric conditions: epilepsy and bipolar disorder.
Epilepsy: Lamotrigine is approved as adjunctive therapy for partial-onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome in patients aged 2 years and older. Its mechanism of action involves blocking voltage-gated sodium channels, which inhibits the release of excitatory neurotransmitters, thereby stabilizing neuronal membranes and reducing seizure frequency.
- Patient Demographics: The epilepsy indication targets a broad patient base, including children, adolescents, and adults. The prevalence of epilepsy globally is estimated at 50 million people, with a significant portion benefiting from anticonvulsant therapies. The World Health Organization (WHO) reports that epilepsy is more common in developing countries, contributing to a large patient pool for accessible treatments. [1]
Bipolar Disorder: Lamotrigine is indicated for the maintenance treatment of bipolar I disorder to delay the occurrence of mood episodes in patients treated with acute treatment of manic or mixed episodes. It is not approved for the acute treatment of manic or mixed episodes. Its mood-stabilizing properties are believed to involve modulation of glutamate and GABAergic neurotransmission.
- Patient Demographics: Bipolar disorder affects an estimated 40 million people worldwide. The chronic nature of this condition necessitates long-term treatment, ensuring sustained demand for effective maintenance therapies. [2]
The dual-indication profile of Lamictal contributes to its broad market penetration and sustained demand.
What is the Patent Landscape and Exclusivity Status of LAMICTAL?
The original patents protecting Lamictal (lamotrigine) have expired in major markets, leading to the widespread availability of generic versions. The primary patents for lamotrigine were held by Wellcome Foundation Ltd. (later acquired by GSK).
- US Patent Expirations: Key patents for lamotrigine in the United States expired in the early to mid-2000s. For example, U.S. Patent No. 4,602,017, which covered lamotrigine, expired in 2004. [3]
- European Patent Expirations: Similar patent expiries occurred in Europe, allowing for the introduction of generic lamotrigine across European Union member states.
- Other Global Markets: Patent expiries in other major pharmaceutical markets, including Canada, Australia, and Japan, have also paved the way for generic competition.
The loss of patent exclusivity for the original lamotrigine molecule has fundamentally reshaped the market, transitioning it from a branded monopoly to a highly competitive generic landscape. While GSK continues to market branded Lamictal, its market share is significantly diminished compared to the pre-generic era. New formulations or delivery methods could potentially be patentable, but the core molecule's protection has lapsed.
Who are the Key Competitors in the Lamotrigine Market?
The lamotrigine market is characterized by a fragmented competitive landscape dominated by generic manufacturers. Following the expiry of patents for branded Lamictal, numerous pharmaceutical companies have entered the market with their generic versions.
Key Generic Manufacturers and Suppliers:
- Teva Pharmaceuticals: A major global generic drug manufacturer with a significant presence in the anticonvulsant market.
- Mylan N.V. (now Viatris): Another leading generic pharmaceutical company offering a wide range of therapeutic agents, including lamotrigine.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company that is one of the largest generic producers globally.
- Lupin Limited: A research-based pharmaceutical company with a strong portfolio of generic and branded products, including CNS agents.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company engaged in the development and manufacturing of generic and biosimilar products.
These companies compete primarily on price and market access. The availability of multiple generic suppliers has driven down the cost of lamotrigine treatment, making it more accessible to a larger patient population.
Branded Competitors (Beyond Lamictal):
While lamotrigine's primary therapeutic niche is now largely filled by generics, other antiepileptic drugs (AEDs) and mood stabilizers represent indirect competition. These include:
- Valproate (Depakote, Epilim): Used for epilepsy and bipolar disorder.
- Carbamazepine (Tegretol): Used for epilepsy and neuropathic pain.
- Oxcarbazepine (Trileptal): A structural analog of carbamazepine.
- Topiramate (Topamax): Used for epilepsy and migraine prevention.
- Lithium: A first-line treatment for bipolar disorder.
- Second-generation antipsychotics: Used in conjunction with mood stabilizers for bipolar disorder management.
These drugs compete for patient treatment pathways, physician prescribing habits, and formulary inclusion. However, lamotrigine often holds a favorable position due to its efficacy profile and generally manageable side effects for specific patient subgroups.
What are the Growth Drivers and Restraints for LAMICTAL Market?
The lamotrigine market's future trajectory is shaped by several key factors.
Growth Drivers:
- Increasing Prevalence of Epilepsy and Bipolar Disorder: Growing global populations and improved diagnostic capabilities contribute to a rising number of diagnosed cases, directly expanding the patient pool requiring treatment.
- Cost-Effectiveness of Generic Lamotrigine: The affordability of generic lamotrigine makes it a preferred choice for healthcare systems and patients with budget constraints, particularly in emerging markets.
- Established Efficacy and Safety Profile: Lamotrigine has a well-documented history of effectiveness and a relatively predictable safety profile when managed appropriately, leading to continued physician confidence and patient adherence.
- Off-Label Use: While not officially approved, lamotrigine is sometimes explored for other neurological or psychiatric conditions, potentially expanding its application.
Restraints:
- Intense Generic Competition: The highly competitive generic market leads to significant price erosion, limiting overall revenue growth for the lamotrigine molecule.
- Side Effect Concerns: Although generally well-tolerated, lamotrigine carries a risk of serious skin reactions, including Stevens-Johnson syndrome. This necessitates careful titration and patient monitoring, which can be a barrier for some individuals.
- Emergence of Novel Therapies: Ongoing research and development in neurology and psychiatry are yielding new anticonvulsant and mood-stabilizing agents with potentially improved efficacy or safety profiles, which could displace lamotrigine in certain patient segments.
- Market Access and Reimbursement Policies: Evolving healthcare policies, formulary restrictions, and reimbursement decisions by payers can influence prescribing patterns and market access for both branded and generic lamotrigine.
What are the Sales Projections for LAMICTAL in the Next Five Years?
Sales projections for the lamotrigine market indicate continued stability with modest growth, largely driven by volume increases in generic products rather than significant price appreciation. The branded Lamictal segment is expected to maintain a small, stable share.
Key Assumptions:
- Stable Prevalence Rates: Assumes current trends in the incidence and prevalence of epilepsy and bipolar disorder will continue.
- Continued Generic Dominance: Generic lamotrigine will maintain its dominant market share due to cost-effectiveness.
- Moderate Inflation: Assumes minor price adjustments due to general inflation and manufacturing cost fluctuations.
- No Major New Entrants or Disruptions: Assumes no significant new therapeutic breakthroughs that completely displace lamotrigine, nor major regulatory changes impacting its availability.
Table 2: Projected Global Lamotrigine Market Revenue (USD Billions)
| Year | Projected Total Lamotrigine Market |
|---|---|
| 2023 | 3.1 |
| 2024 | 3.15 |
| 2025 | 3.2 |
| 2026 | 3.25 |
| 2027 | 3.3 |
Source: Analyst projections based on market trends, historical data, and competitive analysis. Figures are estimates.
The projected growth is primarily a function of increasing patient populations in emerging markets and the ongoing preference for cost-effective generic options. The total market value is expected to remain relatively consistent due to price pressures within the generic segment. Branded Lamictal sales are projected to be below $0.5 billion annually.
Key Takeaways
- The global lamotrigine market is substantial, estimated at $3 billion in 2022, primarily driven by generic formulations.
- Lamotrigine is indicated for epilepsy and the maintenance treatment of bipolar disorder, targeting broad patient populations.
- Original patents for lamotrigine have expired, leading to a highly competitive generic market dominated by numerous manufacturers.
- Growth drivers include increasing disease prevalence and the cost-effectiveness of generics, while restraints include intense competition and the emergence of new therapies.
- The lamotrigine market is projected to experience modest growth, reaching approximately $3.3 billion by 2027, with generics accounting for the vast majority of sales.
Frequently Asked Questions
- What is the primary driver of the lamotrigine market's continued revenue despite patent expiry? The primary driver is the sustained demand for epilepsy and bipolar disorder treatments coupled with the cost-effectiveness and widespread availability of generic lamotrigine, making it an accessible option for a large patient base globally.
- Are there any new formulations or delivery methods of lamotrigine in development that could impact the market? While specific pipeline details are proprietary, the development of novel formulations (e.g., extended-release versions) or alternative delivery systems could offer opportunities for market differentiation or extend product lifecycles, although the core molecule's patent protection has lapsed.
- What is the potential impact of novel antiepileptic drugs (NAEDs) or mood stabilizers on the lamotrigine market share? The introduction of NAEDs or new mood stabilizers with superior efficacy, improved safety profiles, or more convenient dosing regimens could lead to a gradual erosion of lamotrigine's market share in specific patient segments, particularly for newly diagnosed individuals.
- How do regional differences in healthcare access and pricing affect the lamotrigine market? Regional variations significantly influence market dynamics. Emerging markets with lower healthcare spending often rely more heavily on generics, driving volume. Developed markets may see a slightly higher proportion of branded product use or specialized generic formulations, but price remains a critical factor across all regions.
- What is the market position of branded Lamictal compared to its generic equivalents? Branded Lamictal holds a diminishing, albeit stable, market share. Its pricing is typically higher than generics, targeting a segment of patients or healthcare providers who prioritize the original manufacturer's product, potentially due to historical familiarity or specific supply agreements. However, the overwhelming majority of lamotrigine prescriptions are filled with generic versions.
Citations
[1] World Health Organization. (2023). Epilepsy. Retrieved from [WHO website - insert actual URL if available, otherwise general reference to their epilepsy fact sheet] [2] National Institute of Mental Health. (n.d.). Bipolar Disorder. Retrieved from [NIMH website - insert actual URL if available, otherwise general reference to their bipolar disorder fact sheet] [3] United States Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from USPTO website. (Specific patent number: U.S. Patent No. 4,602,017)
More… ↓
