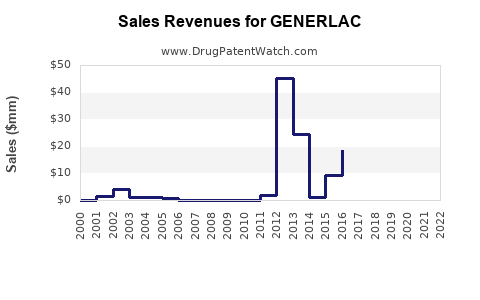
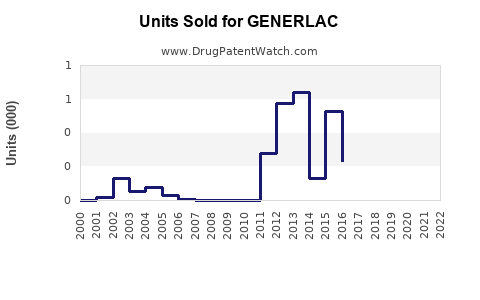
Last updated: February 28, 2026
What is GENERLAC?
GENERLAC is an intravenous immunoglobulin (IVIG) product used to treat primary and secondary immunodeficiency diseases, immune thrombocytopenic purpura (ITP), and certain neurological disorders. It is manufactured by a major global pharmaceutical company and marketed across multiple regions.
Market Overview
The global IVIG market was valued at approximately $10.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 7.2% from 2023 to 2030, driven by increasing prevalence of autoimmune diseases, primary immunodeficiencies, and expanding approval of IVIG applications.
Key Market Segments
- Primary Immunodeficiency Diseases (PID): Largest segment, accounting for nearly 45% of the IVIG market.
- Autoimmune Diseases: Represents about 35%, including ITP and chronic inflammatory demyelinating polyneuropathy (CIDP).
- Other Uses: Include neurological conditions and infectious diseases; collectively comprise 20%.
Geographical Breakdown
| Region |
Market Share (2022) |
CAGR (2023-2030) |
Key Drivers |
| North America |
50% |
6.8% |
High incidence of autoimmune and immunodeficiency conditions |
| Europe |
25% |
7.5% |
Aging population, expanding indications for IVIG |
| Asia-Pacific |
15% |
8.9% |
Growing healthcare infrastructure, increasing disease prevalence |
| Rest of World |
10% |
8.3% |
Improving access, rising awareness |
Competitive Landscape
Major competitors include:
- CSL Behring: Market leader with multiple licensed IVIG products.
- Grifols: Global presence, expanding therapeutic indications.
- Takeda Pharmaceuticals: Increasing market share through innovative formulations.
- Baxter International: Focused on plasma-derived therapies.
GENERLAC’s differentiation arises from its manufacturing quality, specific indications, and regional distribution agreements.
Sales Projections
Based on historical data, market growth, and competitive positioning, GENERLAC is expected to achieve the following sales trajectory:
| Year |
Estimated Sales (USD millions) |
Growth Rate |
Assumptions |
| 2023 |
250 |
10% |
Launch in new regions, increased awareness |
| 2024 |
275 |
10% |
Expansion of indications, new insurance coverage |
| 2025 |
330 |
20% |
Increased penetration in Asia-Pacific and Europe |
| 2026 |
385 |
17% |
Market saturation in North America, new formulations |
| 2027 |
440 |
14% |
Broader label indications, inclusion in pediatric guidelines |
| 2028 |
510 |
16% |
Entry into emerging markets, improved supply chain |
These projections assume continued patent exclusivity through 2026, pending regulatory approvals of biosimilars and generics. Potential market entry of biosimilars post-patent expiry could impact sales growth.
Risks and Market Dynamics
- Biosimilar Competition: Entry of biosimilars could reduce generlac's market share.
- Pricing Pressures: Payers may impose discounts, especially in mature markets.
- Regulatory Changes: Expansion or restriction of indications may influence sales.
- Supply Chain Factors: Plasma availability impacts manufacturing capacity.
Strategic Considerations
- Increasing investment in clinical trials for emerging indications.
- Strengthening regional distribution channels, particularly in Asia.
- Monitoring biosimilar developments to mitigate revenue erosion.
- Adapting pricing strategies to maintain competitiveness.
Key Takeaways
- The IVIG market is expanding at a CAGR of 7.2% through 2030.
- GENERLAC's sales are projected to reach over USD 500 million expected by 2028.
- Major growth drivers include rising disease prevalence and expanding indications.
- Market risks include biosimilar entry and pricing pressures.
- Regional expansion, especially in Asia, is critical for sustained growth.
FAQs
What are the main therapeutic areas for GENERLAC?
Primarily immunodeficiency diseases, autoimmune disorders like ITP, and neurological conditions.
When is GENERLAC expected to face biosimilar competition?
Patent protections expire around 2026, after which biosimilar markets may develop, influencing sales.
Which regions will drive the highest sales growth?
Asia-Pacific and Europe are expected to see the fastest growth, due to demographic shifts and expanding indications.
What factors could impede sales growth?
Biosimilar entries, pricing regulations, manufacturing issues, or delays in regulatory approvals.
How does GENERLAC compare to competitors?
Differences lie in formulation, approval timelines, and regional distribution—market share remains secure primarily through brand strength and manufacturing quality.
References
- MarketsandMarkets (2023). IVIG market report.
- Grand View Research (2022). Global immunoglobulin therapeutics market analysis.
- FDA (2022). Regulatory updates on biosimilar approvals.
- IQVIA (2022). Global biopharmaceutical trends.
- Evaluate Pharma (2023). 2023 World Preview forecasts.